Animals
All animal experiments conformed to the Guide for the Care and Use of Laboratory Animals and were approved by the Institutional Committee of Laboratory Animal Experimentation of the RIKEN Centre for Biosystems Dynamics Research. C57BL/6 (B6), JF1/Ms (JF1), BDF1 (C57BL/6 × DBA/2) and R26R-H2B-EGFP+/− (C57BL/6 × DBA/2 background)43 mice, aged 8–10 weeks, were used to produce oocytes and sperm. Surrogate pseudopregnant females used as embryo transfer recipients (see ‘Embryo transfer’) were ICR strain mice mated with vasectomized males of the same strain. BDF1 mice were purchased from Japan SLC Inc.
Oocyte collection
Mature oocytes were collected from the oviducts of 8- to 10-week-old female mice that had been induced to superovulate with 5 IU of equine chorionic gonadotropin (ASKA Pharmaceutical) followed by 5 IU of human chorionic gonadotropin (ASKA Pharmaceutical) 48 h later. Cumulus-oocyte complexes were collected from the oviducts approximately 16 h after human chorionic gonadotropin injection. Cumulus-oocyte complexes were placed in M2 medium and treated with 0.1% (w/v) bovine testicular hyaluronidase. After several minutes, the cumulus-free oocytes were washed twice and then transferred to Chatot, Ziomek and Bavister medium (CZB). Mature MII oocytes were subjected to ICSI.
Micromanipulation
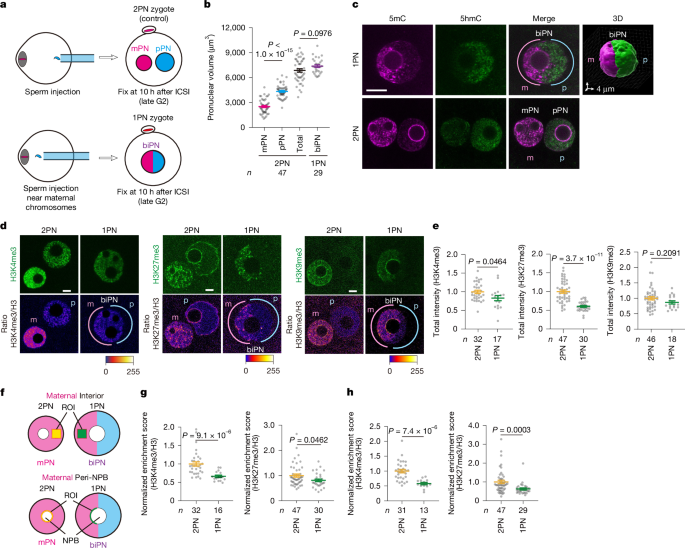
For cytoplasmic removal (Extended Data Fig. 3a,b), micromanipulation was performed as described previously44 with some modifications. Briefly, oocytes at the MII stage were transferred to M2 medium supplemented with 5 μg ml−1 cytochalasin B (Sigma-Aldrich) for 10 min. Then, the zona was cut using the LYKOS laser system (Hamilton Thorne) in a micromanipulation chamber placed on a warmed stage (37 °C) in an inverted microscope (Olympus). After cutting the zona, we held each oocyte with a holding pipette at the 9-o’clock position, and then rotated the oocyte until the hole of the zona was at the 3-o’clock position. After inserting a fire-polished injection pipette (inner diameter 30 µm) through the hole of the zona, we aspirated half the cytoplasmic volume and pinched off (Supplementary Video 3). The volume of the aspirated cytoplasm was controlled by an ocular micrometer. To obtain control oocytes, we aspirated half the total cytoplasmic volume and returned it to the oocyte.
To generate doubled oocytes (Extended Data Fig. 3a,b), we performed micromanipulation based on previously described methods45 with some modifications. Briefly, oocytes at the MII stage were transferred to M2 medium supplemented with 5 μg ml−1 cytochalasin B. A hole was made in the zona pellucida by applying several piezo pulses using an enucleation pipette. The MII spindle was aspirated into the pipette with a minimal volume of ooplasm. After enucleation, oocytes were cultured in CZB medium for at least 15 min for recovery. The enucleated oocytes and intact oocytes were treated with acid Tyrode’s solution (pH 2.1–2.5, Irvine Scientific) for 30 s to dissolve the zona. After several washes, the zona-free oocytes were cultured in CZB medium for at least 30 min, and then transferred to M2 medium containing 0.05 mg ml−1 phytohaemagglutinin (Wako Pure Chemical Industries) for 1 min. The oocytes were transferred to M2 medium on a micromanipulation chamber, in which an intact oocyte and an enucleated oocyte were attached to each other by micropipettes. The attached oocytes were cultured in CZB medium for at least 15 min, washed three times with fusion medium (0.3 M mannitol, 0.1 mM MgSO4 and 0.1% polyvinyl alcohol) then transferred to the same solution placed between parallel electrodes and separated by 0.5 mm in a chamber. Current was applied from an electric cell fuser (LF101; BEX Co.) (15 V alternate current at 1 MHz for 1 s to line up the attached oocytes, 30 V direct current pulse for 20 µsec to induce cell fusion). The oocytes were washed and then cultured in CZB medium for 30 min for recovery and cell rounding (Supplementary Video 4).
Fertilization
IVF was performed according to the manufacturer’s instructions using CARD MEDIUM (Cosmo Bio). To limit the time of fertilization, we set the insemination time to 1 h.
ICSI with sperm heads was carried out as described previously46. Briefly, the sperm head was separated from the tail by applying several piezo pulses to the neck region, and then injected into an oocyte. After 20 min of recovery at room temperature, injected oocytes were cultured in CZB.
To generate 1PN biparental zygotes, we injected the sperm head around the midpoint between the maternal spindle and the oocyte centre. Placing the sperm head away from the plasma membrane helped to prevent its extrusion into the polar body. The sperm head was still extruded occasionally into the polar body, resulting in the production of haploid parthenotes, which were distinguished by the immunostaining patterns of the polar body and pronucleus and excluded from further analysis. In rescue experiments, zygotes were treated with 50 nM TSA for 10 h after ICSI.
Parthenogenetic and androgenetic zygotes
To generate haploid parthenogenetic embryos, we activated oocytes using CZB supplemented with 10 mM SrCl2, 2 mM EGTA for 10 h. To generate androgenetic embryos, a sperm head was injected into the enucleated MII oocytes. Injected oocytes were cultured in CZB medium for 30 min for recovery, and then cultured in activation medium.
Embryo transfer
Embryo transfer was carried out as described previously47. Embryos at the two-cell stage were transferred at 0.5 days post-coitum into the oviducts of pseudopregnant female mice of the ICR strain that had been mated with a vasectomized male the night before transfer. At 18.5 days post-coitum, the offspring were delivered by Caesarean section because the mother sometimes eats the pups. Mice without implantation marks were excluded from the data. Surviving pups were fostered by an ICR foster mother that had given birth on the same day.
Live cell imaging
After linearization of the template plasmid, mRNA was synthesized using the mMESSAGE mMACHINE KIT (Ambion). Synthesized RNAs were stored at −80 °C until use. In-vitro-transcribed mRNAs (2.0 pl of 1,000 ng µl−1 EGFP-Nup153, 2.0 pl of 850 ng µl−1 importin β-binding domain of importin α (IBB)-DiHcRed and 1.2 pl of 1,000 ng µl−1 mCherry-methyl CpG binding domain (MBD)-nuclear localization signal (NLS)) were microinjected into MII oocytes. IBB-DiHcRed and mCherry-MBD-NLS serve as markers for nuclear import48 and post-fertilization paternal chromosomes49, respectively. The microinjected oocytes were cultured for 1 h, and then ICSI or IVF was performed.
Live cell imaging was performed as described previously32 with some modifications. Briefly, a Zeiss LSM710 or LSM880 confocal microscope, operated using ZEN software (ZEN 2011 SP3 (black) v.8.1.11.484 for LSM710 and ZEN 2.3 SP1 FP1 (black) v.14.0.13.201/v.14.0.9.201 for LSM880) and equipped with a ×40 C-Apochromat 1.2 numerical aperture water immersion objective lens (Carl Zeiss) was controlled by a multi-position autofocus macro50. For imaging, 29 confocal z-sections (every 3.0 µm) of 512 × 512 pixel xy images covering a total volume of 84.85 × 84.85 × 84.00 µm were acquired at 5-min intervals for at least 14 h after fertilization.
Kdm5b knockdown
To knock down Kdm5b, 2.5 pl of 5 nM Kdm5b (catalogue no. 4390771, product ID s93702) and negative control (catalogue no. 4390843) Silencer Select siRNAs (Thermo Fisher Scientific) were microinjected into MII oocytes. The microinjected oocytes were cultured for 1 h then used for ICSI.
Three-dimensional analysis
To measure pronuclear volume (Fig. 3 and Extended Data Fig. 4), we performed three-dimensional surface rendering with the signals of mCherry-MBD-NLS or H2B-GFP with Imaris software (Bitplane, v.7.4.2). The volume was obtained after ellipsoid fitting.
Immunostaining
Zygotes were fixed 10 h after fertilization (PN5 stage) in 2% paraformaldehyde in PBS-polyvinyl alcohol (PVA) (pH 7.4) for 30 min. For the 5hmC and 5mC staining, fixed zygotes were incubated in 4 N HCl solution at room temperature for 10 min, followed by neutralization (1 M Tris-HCl, pH 8.0) for 20 min. After blocking and permeabilization in PBS-PVA containing 1 mg ml−1 bovine serum albumin (BSA) (PBS-PVA-BSA) and 0.1% Triton X-100, oocytes were incubated with appropriate primary antibodies overnight at 4 °C, washed several times in PBS-PVA-BSA and incubated with secondary antibodies for 120 min at room temperature. DNA was counterstained with 40 µg ml−1 of Hoechst 33342. Finally, the oocytes were washed and transferred to BSA-PVA for imaging with a Zeiss LSM780 confocal microscope, operated using ZEN software (ZEN 2011 SP3 (black) v8.1.11.484). Images were reconstructed in three dimensions with Imaris software.
The following primary antibodies were used: rabbit anti-histone H3K4me3 (1:200, Upstate Biotechnology, catalogue no. 07-473), rabbit anti-histone H3K9me3 (1:200, Upstate Biotechnology, catalogue no. 07-442), rabbit anti-histone H3K27me3 (1:200, Upstate Biotechnology, catalogue no. 07-449), rabbit anti-histone H3K27ac (1:200, Abcam, catalogue no. ab177178), mouse anti-histone H3 (1:200, Abcam, catalogue no. ab195277), mouse anti-nuclear pore complex (mab414) (1:200, Abcam, catalogue no. ab24609), rat anti-NUP98 (1:800, Abcam, catalogue no. ab50610), rabbit anti-5hmC (1:500, Active Motif, catalogue no. 39769), mouse anti-5mC (1:500, Eurogentec, catalogue no. BI-MECY-0100) and rabbit anti-KDM5b (1:200, Abcam, catalogue no. ab181089) antibodies. The secondary antibodies were Alexa Fluor 488 Goat Anti-Mouse IgG (H+L) (catalogue no. A11029), Alexa Fluor 488 Goat Anti-Rabbit IgG (H+L) (catalogue no. A11034), Alexa Fluor 488 Goat Anti-Rat IgG (H+L) (catalogue no. A11006) and Alexa Fluor 555 Goat Anti-Mouse IgG (H+L) (catalogue no. A21424) (1:400, Molecular Probes).
Quantification of fluorescence signals
Total fluorescence intensity throughout the entire pronucleus was measured using Imaris. The pronuclear volume was defined based on H3 signals in three dimensions, and the total signal intensity of the modified histone within the defined volume was obtained. Fluorescence intensity of a cytoplasmic region of equal volume was measured and subtracted to yield corrected total pronuclear intensity values.
To quantify the relative levels of H3K4me3, H3K27me3 or H3K27ac to histone H3, the mean signal intensity for each modified histone (Ime_pro) within ROIs in two defined categories: (1) the pronuclear interior away from the NPB and (2) the peri-NPB region. NPBs were defined as spherical areas of depleted H3 signal within the pronucleus. ROIs for the interior regions were placed more than 1 µm away from both the pronuclear and NPB rims and from the estimated maternal–paternal boundary within the maternal pronucleus or hemisphere. ROIs for the peri-NPB regions were generated using the ‘Make band’ (band = 1) function in Fiji (v.1.54p) after delineating the periphery of the NPB manually within the maternal pronucleus or hemisphere. For comparisons of fluorescence levels across samples, ROIs of identical size were applied consistently: fixed at 3.82 µm × 3.82 µm under normal conditions and adjusted to 3.32 µm × 3.32 µm for smaller pronuclei (for example, halved zygotes). The mean cytoplasmic signal intensity (Ime_cyto) from a region near the pronuclei was subtracted (Ime_pro − Ime_cyto). Similarly, the histone H3 intensity was determined as (IH3_pro − IH3_cyto). The ratio between the two values, (Ime_pro − Ime_cyto)/(IH3_pro − IH3_cyto) was then calculated.
Single-embryo RNA-seq
C57BL/6 × JF1/Ms zygotes were selected 10 h after fertilization (late G2 or PN5 stage) and the second polar body was removed using a micromanipulator. Two-cell embryos were collected 34 h after fertilization. Single-embryo RNA-seq libraries were prepared with SMART-Seq v.4 Ultra Low Input RNA Kit (Takara Bio) and Nextera XT DNA Library Preparation Kit (Illumina).
The RNA-seq reads were filtered using fastp (v.0.20.0)51 to remove Illumina adaptor sequences, and to eliminate low-quality bases with the ‘-3 -q 15 -l 15’ options. The filtered reads were mapped to the mouse genome (mm10) using Hisat2 with default parameters except for the ‘–sp 1000,1000’ option. The featureCounts v.2.0.052 was used to generate counts of reads aligned to annotated genes in the Ensembl GRCm38.99 with options ‘-p -O–fraction’.
Before conducting subsequent downstream analyses, samples were subjected to stringent quality filtering using the following threshold: embryos with a number of detected genes greater than 10,000, and a ratio of mitochondrial genes less than 0.2 were retained. This filtering excluded three samples. Samples were also subjected to parental allele classification (‘Parental allele classification in bioinformatics analysis’) to exclude parthenotes (paternal allele ratio of less than 0.1) that could not be distinguished from 1PN biparental zygotes by microscopic observation.
The amount of poly(A) RNA for each embryo was estimated from the inferred linear regression parameters based on the ratio of spike-in derived reads using the ‘relative2abs’ function implemented in the monocle package53. In each experiment, embryos derived from 2PN zygotes were used as a reference for data normalization. Differentially expressed genes (false discovery rate (FDR) < 0.05) between the PN types were explored using the edgeR (v.3.40.2)54 with the trimmed mean of M values normalization, using the dataset of genes expressed in at least ten samples.
Carrier-assisted chromatin immunoprecipitation followed by sequencing (CATCH-seq)
CATCH-seq is an improved method of the ultra-low-input native chromatin immunoprecipitation followed by sequencing (ChIP-seq) (ultra-low-input micrococcal nuclease-based native ChIP (ULI-NChIP))55; its original protocol was described in ref. 28 and further optimized in ref. 27. BDF1(C57BL/6×DBA2) × JF1/Ms zygotes were selected 10 h after fertilization (late G2 or PN5 stage) and the second polar body was removed using a micromanipulator. Zygotes were collected in 9 µl of Nuclei EZ lysis buffer (Sigma, NUC-101) supplemented with a complete EDTA-free protease inhibitor cocktail and 1 mM phenylmethanesulfonyl fluoride. The samples were snap-frozen in liquid nitrogen and stored at −80 °C until use. On the day of library construction, the samples were thawed on ice and added with 1 µl of the lysis buffer containing a constant number of Drosophila melanogaster S2 cells (Thermo Fisher Scientific, catalogue no. R69007) for spike-in normalization purposes. Then, 1 µl of a 1% Triton X-100 (Merck, catalogue no. 93443) and 1% deoxycholate (Nacalai, catalogue no. 10712-54) mixture solution was added to the samples, which then sat on ice for 5 min. The chromatin was fragmented by 2 U µl−1 micrococcal nuclease (MNase) (catalogue no. M0247S, NEB) in 1× MNase buffer supplemented with 1% PEG6000 (Hampton Research, catalogue no. HR2-533) and 2 mM dithiothreitol (Nacalai) at 21 °C for 7.5 min. The MNase reaction was stopped by adding 1/10 volume of 100 mM EDTA and 1/12 volume of the 1% Triton X-100 and 1% deoxycholate mixture, then the samples were rested on ice for 15 min. The chromatin lysates were then added to freshly prepared immunoprecipitation buffer55, and 5% volume was kept for input library construction. To the rest of the lysate, 30 ng of annealed I-SceI carrier DNA was added27. The forward and reverse strands of the carrier DNA were as follows: /5AmMC6/Gtagggataacagggtaattagggataacagggtaattagggataacagggtaattagggataacagggtaattagggataacagggtaattagggat aacagggtaat*c/3AmMO/ and /5AmMC6/Gattaccctgttatccctaattaccctgttatccctaattaccctgttatccctaattaccctgttatccctaattaccctgttatccctaattaccctgttatcc cta*c/3AmMO/, respectively, where asterisks represent phosphorothioate bonds. The oligonucleotides were synthesized by Integrated DNA Technologies. For each immunoprecipitation reaction, 0.25 µl of rabbit anti-H3K4me3 (Active Motif, catalogue no. 39159), 500 ng of rabbit anti-H3K27me3 (Diagenode, catalogue no. C15410069) conjugated to precleared Dynabeads Protein A (Thermo Fisher Scientific, catalogue no. 10006D) and G (Thermo Fisher Scientific, catalogue no. 10007D) mixture was used. The specificity of these antibodies was validated in previous studies (H3K4me3 (ref. 56), H3K27me3 (ref. 57)). After immunoprecipitation at 4 °C overnight, the chromatin-Dynabeads were washed twice each with low and high salt wash buffers, and the chromatin was eluted in the freshly prepared ChIP elution buffer at 65 °C for 1 h55. DNA was recovered by ChIP DNA Clean and Concentrator Kit (Zymo Research). Adaptor ligation was performed by NEBNext Ultra II DNA Library Prep Kit for Illumina (catalogue no. E7645, NEB) in a half scale of the manufacturer’s instruction, and the libraries were purified by 1.8× SPRIselect beads (catalogue no. B23318, Beckman Coulter). The DNA was amplified by KAPA Hifi 2× mater PCR mix (catalogue no. KK2605) for 13–15 PCR cycles with dual indexing primers (NEBNext Multiplex Oligos for Illumina, catalogue no. E6440). After purification with 0.9× SPRIselect beads, the samples were digested by I-SceI (5 U µl−1, NEB, catalogue no. R0694) at 37 °C for 2 h followed by heat inactivation at 65 °C for 20 min and purified by 0.9× SPRIselect beads. The second amplification was not performed. The libraries were sequenced on Nextseq500 (single-end reads) or Nextseq2000 (paired-end reads) (Illumina).
CATCH-seq data analysis
For CATCH-seq, all sequencing reads after trimming by fastp were aligned to the mouse (mm10) using Bowtie 2 v.2.3 (ref. 58) with ‘-N 1 -L 25’ options. For allele-specific analysis of CATCH-seq in the BDF1 × JF1 F1 hybrid embryos, parental allele classification was described below (‘Parental allele classification in bioinformatics analysis)’. For visualization of CATCH-seq using the Integrative Genomics Viewer (v.2.16.1)59, genome coverage tracks were generated using bamCoverage from deepTools (v.3.5.1)60 with the parameters ‘–binSize 50–scaleFactor 1–normalizeUsing RPKM–numberOfProcessors 28–extendReads 200–ignoreDuplicates–smoothLength 100’. SNPsplit (v.0.3.4)61 was applied to assign uniquely aligned reads to their parental origins. To compare replicates, read counts over each bin (10 kb) across the whole genome were calculated and the reads per million values for each bin were used to calculate the Spearman correlation coefficient. Averaged reads per kilobase million (RPKM) between replicates was calculated using bigwigAverage from the deepTools suite (v.3.5.3). To compare correlation between CATCH-seq and STAR ChIP-seq data23, read counts over each bin (10 kb) across the whole genome were calculated and the reads per million values for each bin were used to calculate the Spearman correlation coefficient using deepTools suite and visualized using the R function pheatmap.
For analyse the H3K27me3 allelic-biased region analysis, using the H3K27me3 ChIP-seq datasets of mourla stage embryos57, the 10-kb bins (no slide) were divided into ‘K27me3 Mat-biased’ and ‘K27me3 Pat-biased’ groups. Each 10-kb bin contained more than 50 SNPs. The average RPKM of H3K27me3 in each bin was calculated using ComputeMatrix in deeptools (v.3.5.1). Bins with RPKM > 2 were used as positive regions. Then, the allelic ratio of average RPKM in each group’s bin was calculated and fold change > 2 defined as allelic-biased regions.
Definition of H3K4me3 broad peaks
Previous H3K4me3 ChIP-seq data in fully grown oocytes62 were aligned to mm10 and the data were processed as described above.
H3K4me3 peaks were called MACS2 (v.2.2.9.1)63 with the parameters ‘-g mm –nomodel –nolambda –broad’. To identify broad H3K4me3 domains, those with a distance shorter than 5 kb were merged using the ‘merge’ function from BEDTools (v.2.26.0), as described previously24.
Parental allele classification in bioinformatics analysis
To distinguish the parental alleles of the read sequences of BDF1 × JF1/Ms and C57BL/6 × JF1/Ms embryos, diagnostic SNPs (dSNPs) were developed using the deposited deep sequence data obtained from JF1/Ms64. After mapping the JF1/Ms reads to the mouse genome (mm10) using BWA-MEM v.0.7 with the default parameters, SNPs were called using HaplotypeCaller implemented in GATK v.4.1.4.1 (ref. 65). After filtering out low-confidence SNPs with ‘QD < 2.0, QUAL < 30.0, SOR > 3.0, FS > 60.0, MQ < 40.0, MQRankSum < −12.5, and ReadPosRankSum < −8.0’, differently fixed 20,885,964 sites were used as dSNPs. For the reference of allele-specific analysis, dSNP sites on the mouse genome (mm10) were masked as N. Each aligned read (see ‘Single-embryo RNA-seq’ and ‘CATCH-seq data analysis’ above) was subjected to assign its parental origin using the dSNPs with SNPsplit v.0.3.4 (ref. 61).
Preparation of heavy-labelled peptides by cell-free synthesis for MS-QBiC
Stable isotope-labelled peptides were synthesized using an Escherichia coli reconstituted cell-free protein synthesis system, PURE system, containing 13C6 15N4 l-arginine and 13C6 l-lysine (Thermo Scientific). Details of the synthesis procedure are described previously66 with slight modifications. In brief, DNA templates for the PURE system were prepared by a two-step PCR using Taq DNA polymerase (PCR Master Mix, Promega) with forward primers containing the T7 promoter and the appropriate reverse primers listed in Supplementary Table 2. The amino acid sequences of the synthesized peptides are shown in Supplementary Table 2. Subsequently, synthetic peptides underwent reductive alkylation, trypsin digestion, and desalting using an in-house-made C18 stage-tip. The peptides were then dried with a Speedvac, dissolved in 0.1% trifluoroacetic acid/2% acetonitrile, and used for mass spectrometry.
Liquid chromatography–tandem mass spectrometry experiments
To perform liquid chromatography–tandem mass spectrometry (LC-MS/MS) analysis, pronuclei were isolated by micromanipulator (Supplementary Video 5). Approximately 170 maternal and paternal pronuclei were pooled for the analysis. Biological replicates were performed in triplicate. Each pronucleus was suspended in 50 mM ammonium bicarbonate and reduced and alkylated with tris(2-carboxyethyl)phosphine and iodoacetamide. Trypsin digestion was performed using the SP3 method67. For mass spectrometry, the tryptic digests of pronuclei were mixed with the heavy-labelled peptides and iRT-kit 11 peptides (Biognosys AG) as a retention time marker.
Mass spectra were obtained with an Orbitrap Eclipse (Thermo Fisher Scientific) coupled to a nanoflow UHPLC system (Vanquish; Thermo Fisher Scientific). The peptide mixtures were loaded onto a C18 trap column (PepMap Neo Trap Cartridge, ID 0.3 mm × 5 mm, particle size 5 μm, Thermo Fisher Scientific) and then fractionated by the C18 analytical column (Aurora, ID 0.075 × 250 mm, particle size 1.7 μm, IonOpticks). The peptides were eluted at a flow rate of 300 nl min−1 using the following gradient, with the percentage of solvent B indicated: a linear gradient from 0% to 2% over 1 min, followed by a linear increase from 2% to 5% over 2 min, a linear gradient from 5% to 16% over 58.5 min, another linear gradient from 16% to 25% over 22.5 min, a linear gradient from 25% to 35% over 13 min, a sharp increase to 95% over 1 min, holding at 95% for 5 min and finally re-equilibration at 5%. The composition of solvent A and solvent B is 100% H2O, 0.1% formic acid and 100% acetonitrile, 0.1% formic acid, respectively. The Orbitrap was operated in a data-dependent mode with a cycle time of 3 s. The MS1 scan was collected at 120,000 resolution with the mass range for 335–1,500 m/z using a standard automated gain control. Intensity threshold of 20,000 and charge states 2–7 were taken for MS/MS starting from high intensity precursors. Quadrupole was used for precursor isolation with 1.6 m/z isolation width and fragmented with normalized higher energy dissociation energy of 30% and resulting fragment ions were recorded in Orbitrap analyser. The MS2 scan was collected at 30,000 resolution using a standard automated gain control target and maximum injection time of 54 ms. Dynamic exclusion was set to 20 s.
The raw data files were searched against the Mus musculus dataset (Uniprot Proteome UP000005640, 20,230,207 downloaded) with the common Repository of Adventitious Proteins (cRAP, ftp://ftp.thegpm.org/fasta/cRAP) to recognize the contaminant proteins. The search was conducted through MASCOT v.2.8 (Matrix Science) using Proteome discoverer 2.5 (Thermo Fisher Scientific) with the FDR for peptide and protein identification set at 1%. The following settings were used for search parameters: enzyme, trypsin; FDR, 0.01; precursor mass tolerance, 10 ppm; product tolerance, 0.02 Da; fixed modification, carbamidomethylation of cysteine; variable modification, oxidation of methionine and acetylation of protein N-termini; maximum missed cleavages, 2. The peak area calculation was performed using Skyline (v.23.1.0.380) (MacCoss Lab Software)68 with setting MS1 filter to a count of three (M, M + 1 and M + 2). Extracted ion chromatograms processed using Savitzky-Golay filters provided by Skyline software are presented.
Theoretical model
We introduced our theoretical model for pronucleus growth controlled by a limiting factor, assuming pronucleus pores are active channels that selectively transport the limiting factor from the cytoplasm into pronucleus. The limiting factor transportation is associated with the volume flux, the rate of which is assumed to be dependent linearly on the number of the pronucleus pores. We assume that the number of pores \({A}_{i}\) of pronucleus \(i\) increases with the size of pronucleus as \({A}_{i}={a}_{i}{V}_{i}^{2/3}\), where \({a}_{i}\) is the production rate of the pronucleus pores and \({V}_{i}\) is the volume of the pronucleus and the exponent \(2/3\) converts the volume to the membrane area. Then, the time-evolution equation of the number of the limiting factor is given by
$$\frac{{\rm{d}}({V}_{i}{n}_{i})}{{\rm{d}}t}=k{A}_{i}{n}_{\mathrm{cyto}}$$
(1)
where \(k\) is the transportation rate of the limiting factor through a pronucleus pore and \({n}_{\mathrm{cyto}}\) is the density of the limiting factor in cytoplasm. As the limiting factors are transported to pronuclei, that in the cytoplasm decreases as
$$\frac{{\rm{d}}({V}_{\mathrm{cyto}}{n}_{\mathrm{cyto}})}{{\rm{d}}t}=-\sum _{i}k{A}_{i}{n}_{\mathrm{cyto}}$$
(2)
where \({V}_{\mathrm{cyto}}\) is the volume of the cytoplasm and the summation is calculated over the pronuclei that share the cytoplasmic limiting factor. By writing the volume flux rate associated with the transportation of a limiting factor as \(v\), the volume of the pronucleus \(i\) and the cytoplasm changes as
$$\frac{{\rm{d}}{V}_{i}}{{\rm{d}}t}=v{A}_{i}{n}_{{\rm{cyto}}},$$
(3)
$$\frac{{\rm{d}}{V}_{\mathrm{cyto}}}{{\rm{d}}t}=-\sum _{i}v{A}_{i}{n}_{\mathrm{cyto}}$$
(4)
where the summation is calculated over the pronuclei. Here we assume that the total number of the limiting factor and the total volume are conserved in zygote; \({\sum }_{i}{n}_{i}\,{V}_{i}+{n}_{\mathrm{cyto}}\,{V}_{\mathrm{cyto}}={N}_{\mathrm{total}}\) and \({\sum }_{i}{V}_{i}+{V}_{\mathrm{cyto}}={V}_{\mathrm{total}}\). By applying these conservation rules to equations (1) and (2), the time-evolution equations of the concentration of the limiting factor become
$$\frac{{\rm{d}}{n}_{i}}{{\rm{d}}t}=(k-v{n}_{i}){a}_{i}{V}_{i}^{-\frac{1}{3}}{n}_{\mathrm{cyto}}$$
(5)
$$\frac{{\rm{d}}{n}_{\mathrm{cyto}}}{{\rm{d}}t}=-\sum _{i}(k-v{n}_{\mathrm{cyto}}){a}_{i}{V}_{i}^{\frac{2}{3}}\frac{1}{{V}_{\mathrm{cyto}}}{n}_{\mathrm{cyto}}$$
(6)
To solve equations (3)–(6) numerically, we fixed the parameters as \(k=5\) and \(v=0.1\), and the time increment is set to \({\rm{d}}t=0.01\). The number of pronuclei and the pore production rate \({a}_{i}\) are varied to reproduce the experimental conditions. The maternal and paternal pronuclei are distinguished by the pore production rate: \({a}_{\mathrm{mat}}=100\) for the maternal pronucleus and \({a}_{{pat}}=150\) for the paternal pronucleus. For the control case, the total volume and limiting factor are set as \({V}_{\mathrm{total}}=100\) and \({N}_{\mathrm{total}}=1\), and the initial condition is set as \({V}_{\mathrm{mat}}(t=0)={V}_{\mathrm{pat}}(t=0)\,=\,\)\(0.1,\,{n}_{\mathrm{mat}}(t=0)={n}_{\mathrm{pat}}(t=0)=0\), \({V}_{\mathrm{cyto}}(t=0)={V}_{\mathrm{total}}-{V}_{\mathrm{mat}}(t=0)-{V}_{\mathrm{pat}}(t=0)\) and \({n}_{\mathrm{cyto}}(t=0)={N}_{\mathrm{total}}\,/{V}_{\mathrm{cyto}}(t=0)\). For the parthenote, the paternal pronucleus is omitted, and as a consequence the initial condition of the cytoplasmic volume is modified to \({V}_{\mathrm{cyto}}(t=0)={V}_{\mathrm{total}}-{V}_{\mathrm{mat}}(t=0)\). For the doubled and halved zygotes, the total volume and limiting factor are replaced by \({V}_{\mathrm{total}}=200\) and \({N}_{\mathrm{total}}=2\) and by \({V}_{\mathrm{total}}=50\) and \({N}_{\mathrm{total}}=1/2\), respectively.
Statistical analysis
Statistical analyses were performed using GraphPad Prism (v.7.02). Statistical methods used are described in the figure legends. Two-sided tests were performed for all analyses, except for Extended Data Fig. 2h where one-sided Fisher’s exact test was used. No statistical methods were used to predetermine sample size. Oocytes collected from several donor mice were pooled and allocated randomly to experimental groups. Investigators were not blinded to group allocation during experiments or analysis.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.