Animals and housing conditions
Experiments were performed in accordance with the Animal Protection Law of the European Union after permission by the Governments of Upper Bavaria, Germany, or Copenhagen, Denmark, or by the Institutional Animal Care and Use Committees of the Universities of Texas Southwestern, Michigan, Duke or Yale, USA. Mice were double- or single-housed and unless otherwise indicated fed ad libitum with either a regular chow (1314, Altromin or 5L0D, LabDiet) or HFD (58% fat, D12331, Research Diets) under constant ambient conditions of 22 ± 2 °C with constant humidity (45–65%) and a 12 h:12 h light:dark cycle. Leptin receptor-deficient db/db mice were purchased from the Jacksons Laboratory (Strain 000697). Doxycyclin-inducible GIPR-overexpressing mice (TRE-GIPR mice) were generated in-house at The University of Texas Southwestern Medical Center as described previously41. C57BL/6J DIR-knockout mice were generated in-house at Helmholtz Munich (Supplementary Information 1).
Pharmacological studies
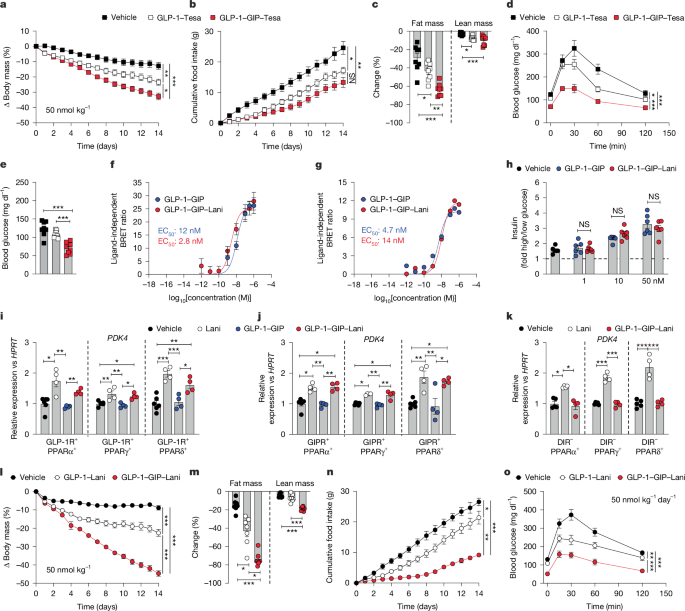
Indirect calorimetry and assessment of body composition was performed as described in Supplementary information 1. Drug effects were assessed in age-matched male single-, or double-housed C57BL6/J mice that were randomly assigned in groups matched for genotype, body weight and body composition (fat and lean tissue mass). Mice were treated subcutaneously at the indicated doses with 5 μl per g body weight of either Vehicle, Lani (CAS: 927961-18-0, MedChemExpress), semaglutide, GLP-1–Lani, GLP-1–Tesa, GLP-1–GIP, GLP-1–GIP–Tesa, GLP-1–GIP–Lani, or co-administration of either GLP-1–GIP plus Lani or GLP-1–Lani plus acyl-GIP (Extended Data Fig. 1a–f). All peptides were provided by the Novo Nordisk Research Center Indianapolis, IN, USA or the Indiana Biosciences Research Institute, IN, USA (for drug development see Supplementary Information 1). Assessment of drug effects on the cardiovascular system and on POMC neuronal activity was performed as described in Supplementary Information 1.
Glucose and lipid metabolism
Glucose and insulin tolerance was assessed in 6 h-fasted mice injected intraperitoneally with either 1.5–2 g kg−1 of glucose or 0.6–0.75 U kg−1 of insulin (Humalog; Eli Lilly). Glucose-induced insulin secretion was assessed in 6 h-fasted mice orally gavaged with 4 g kg−1 glucose. Pyruvate tolerance was assessed in 12 h-fasted mice injected intraperitoneally with 1.25 g kg−1 sodium pyruvate (11360070, Thermo Fisher). Commercially available ELISAs were used according to the manufacturer’s instruction to measure insulin (90080, Crystal Chem), triglycerides (94501, Fujifilm), cholesterol (293-93601, Fujifilm) and free fatty acids (434-91795, 436-91995, 270-77000, Fujifilm). Hyperinsulinaemic-euglycaemic clamps and assessment of tissue-selective glucose uptake were performed as described in Supplementary Information 1.
Gene expression analysis
Total RNA was isolated using the RNeasy Kit (QIAGEN) according to the manufacturer’s instructions. cDNA synthesis was performed using the QuantiTect Reverse Transcription Kit (QIAGEN) or High-Capacity cDNA Reverse Transcription Kit (Thermo Fisher Scientific) according to manufacturer’s instructions. Gene expression was profiled using SYBR green (Thermo Fisher Scientific) and the Quantstudio 7 flex cycler (Applied Biosystems). The relative expression levels of each gene were normalized to the housekeeping gene HPRT. Primer sequences are listed in Supplementary Information 1.
Cell culture studies
HEK293T cells (CRL-3216, ATCC) were cultured in DMEM (11995073, Life Technologies) supplemented with 10% heat-inactivated FBS (10500064, Life Technologies), 100 IU ml−1 penicillin and 100 μg ml−1 streptomycin solution (Pen/Strep, P4333, Sigma Aldrich). Cells (700,000 per well) were seeded in 6-well plates and incubated to 70% confluency in DMEM (10% FBS, 1% Pen/Strep). Twenty-four hours following seeding, transient transfections were performed using Lipofectamine 2000 (11668019, Invitrogen) according to the manufacturer’s instructions without including additional transformation carrier DNA. BRET assays and in vitro quantification of PPAR-responsive genes were performed as described in Supplementary Information 1.
Proteomics, transcriptomics and histology
Proteomics, bulk RNA sequencing and immunofluorescence was performed as described in Supplementary Information 1. For histological analysis, excised samples were fixed in 4% (w/v) neutral buffered formalin, embedded in paraffin and cut into 3 µm slices for haematoxylin and eosin (H&E) staining or immunohistochemistry. Immunohistochemical detection of alpha and beta cells was performed using rabbit anti-insulin (3014, 1:800, Cell Signaling Technology) and mouse anti glucagon (G2654, 1:1,000, Merck) as primary antibodies and goat anti-rabbit AF750 (A21039, 1:100, Invitrogen) and donkey anti-mouse AF555 (A32773, 1:200, Invitrogen) as secondary antibodies. Nuclei were labelled with Hoechst33342 (H1399, Thermo Fischer). The stained tissue sections were scanned with an AxioScan 7 digital slide scanner (Zeiss) equipped with a 20× objective. Steatosis was graded semiquantitatively by the presence of fat vacuoles in liver cells according to the percentage of affected tissue (0, <5%; 1, 5–33%; 2, 33–66%; 3, >66%) and lobular inflammation was scored by overall assessment of inflammatory foci per 200× field (0, no foci; 1, <2 foci; 2, 2–4 foci; 3, >4 foci)56. Automated digital image analysis (Visiopharm) was used for determination of alpha and beta cell mass and mean islet size.
Glucose-stimulated insulin secretion
Mice were euthanized by cervical dislocation, followed immediately by clamping of the bile duct and perfusion with collagenase P (11249002001, Roche Diagnostics). Tissues were incubated in a 15 ml Falcon tube with 1 ml of collagenase P solution for 15 min at 37 °C, followed by addition of 12 ml of the cold G-solution (Sigma Aldrich) and centrifugation at 1,620 rpm at room temperature. The pellet was subsequently washed with 10 ml of the G-solution, which comprised of 500 ml HBSS (BE10-508F, Life Technologies) with 10% BSA (126615, Sigma Aldrich) and 1% Pen-Strep (15140122, Life Technologies), and re-suspended in 5.5 ml of gradient solution (15% Optiprep (5 ml 10% RPMI, Life Technologies) + 3 ml of 40% Optiprep, which was diluted from 60 % Optiprep with G-solution (D1556, Sigma Aldrich)) per sample, and placed on top of 2.5 ml of the gradient solution. To form a 3-layer gradient, 6 ml of the G-solution was added on the top. Samples were then incubated for 10 min at room temperature and centrifuged at 1,700 rpm. The interphase was then collected and filtered through a 70-μm nylon filter (352350, BD Falcon), before washing with G-solution. Islets were handpicked by a micropipette under the microscope and cultured in RPMI 1640 medium (11875093, Life Technologies) overnight.
For assessment of glucose-induced insulin secretion, culture medium was removed and islet microtissues were equilibrated for 1 h with Krebs Ringer Hepes Buffer (KRHB; 131 mM NaCl, 4.8 mM KCl, 1.3 mM CaCl2, 25 mM HEPES, 1.2 mM KH2PO4, 1.2 mM MgSO4, 2% BSA) containing 2.8 mM glucose. The supernatant was collected as a sample under low glucose condition for 45 min incubation, and islets were incubated for another 45 min at 37 °C with KRHB containing 16.7 mM glucose and supplements as above. The supernatant was collected as a sample under high glucose condition and stored at −20 °C. For drug-induced insulin secretion, GLP-1–GIP and GLP-1–GIP–Lani were diluted in 1× KRHB buffer with 20 mM glucose to reach a concentration of 1, 10 or 50 nM. Cells were subsequently treated with either compound for 45 min. Insulin concentrations were determined using a Mouse Insulin ELISA (90082, Crystal Chem).
Conditioned taste avoidance
CTA experiments used either wild-type C57BL/6J mice purchased from the Jackson Laboratories or a glutamatergic neuron GLP-1R knockout mouse (a cross between a Glp1r-flox mouse and a Vglut2–ires–cre mouse, 035238 and 016963; Jackson Laboratory). Mice were acclimatized to the experimental conditions, then automated water systems were removed and replaced with two bottles full of water for three days. During this time, mice were handled and injected daily with 0.1 ml saline. On day 4, water bottles were removed for 22 h to induce thirst. On day 5, mice were given a bottle containing 0.15% saccharin in water for 2 h. Saccharin bottles were weighed before and after the 2 h period to ensure that all mice consumed the tastant. Then, mice were injected with either vehicle or the drugs as indicated, and the saccharin bottle was replaced by a water bottle. On day 8, water bottles were again removed for 22 h to induce thirst. On day 9, mice were given both a water bottle and a saccharin bottle, each of which were weighed at 0 and 24 h. The preference ratio was calculated as: saccharin intake/(saccharin + water intake).
Replicates, randomization and blinding
In vivo studies were performed in male mice that were randomly distributed in groups matched for genotype, age, body weight and body composition (fat and lean tissue mass). The number of independent biological samples per group is indicated in the figure legends and the Source Data files. For in vivo studies, drugs were aliquoted by a lead scientist in number-coded vials and most, but not all, handling investigators were blinded to the treatment condition. Tolerance tests (glucose, insulin, pyruvate and in vivo glucose-stimulated insulin secretion) were performed by experienced research assistants who were blinded to the treatment conditions.
Statistics and reproducibility
For animal studies, sample sizes were calculated based on a power analysis assuming that a body weight difference of ≥5 g between the treatment groups can be captured with a power of ≥75% when using a two-sided, two-tailed statistical test under the assumption of a s.d. of 3.5 and an alpha level of 0.05. Statistical analyses were performed using the statistical tools implemented in GraphPad Prism10 (v10.0.3), and after testing of data for normal distribution using the Kolmogorov–Smirnov test, D’Agostino and Pearson test, Anderson–Darling test or Shapiro–Wilk test implemented in GraphPad Prism (v10.0.3). Statistical tests and individual P values are presented in Supplementary Table 1 or Source Data. All results are given as mean ± s.e.m. P < 0.05 was considered statistically significant. Differences in energy expenditure and heart weight were calculated using ANCOVA with body weight or tibia lengths as covariate using SPSS (v31). No animals or data were excluded from the analysis unless for animal welfare reasons (for example, injury due to fighting) or identification of singular outliers using Grubbs test. Outliers are shown in the Source Data. The metabolic effects of GLP-1–GIP–Lani have been reproduced in several in vivo studies in the manuscript, and across several independent laboratories. In vitro studies in Fig. 1f,g and Extended Data Fig. 2a,b represent 3–6 independent biological replicates, each obtained in an independent study and calculated based on the average of 2–6 technical replicates. In vitro studies in Fig. 1i–k represent 4–6 independent biological replicates, each obtained in an independent study and calculated based on the average of 2 technical replicates. Histological data in Fig. 3l are representative examples out of n = 8 mice per group. Microscopic images and FOS quantification in Fig. 5a–f are representative examples from 3–4 mice per group. Electrophysiological recordings in Fig. 5p are representative examples from 6–7 mice per group. The original pictures from which representative examples are depicted are displayed in Supplementary Fig. 1. All in vivo data represent independent biological samples as indicated in the figure legends.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.