Ethics and study approval
All experiments detailed in this manuscript were approved by the University of Pittsburgh Institutional Animal Care and Use Committee (20077737 and 23073380). The use of retroviral vectors was approved by the Institutional Biosafety Committee. Research involving human participants was approved by the institutional review board (21090049).
Animals
C57BL/6J inbred mice were purchased from Jackson Laboratories. Experimental mice were maintained under specific-pathogen-free conditions in accordance with the University of Pittsburgh Institutional Animal Care and Use Committee, certified by the Association for Assessment and Accreditation of Laboratory Animal Care. Procedures were performed under their guidelines. Male and female, 6- to 8-week-old mice were used in all the experiments. C57BL/6J mice were bred in-house, at a Charles Rivers facility (starting October 2020), or obtained from the Jackson Laboratory. NOD.Cg-Prkdc scid ll2rg tm1Wjl /SzJ/Arc (NSG), Rag2−/− and Ldlr−/− mice were purchased from the Jackson Laboratory.
Cell lines and cell culture
B16, B16-OVA, A549 and NALM6 cell lines were cultured in complete RPMI media (RPMI (Gibco, 11875-093), 10% fetal bovine serum (FBS), 2 mM l-glutamine, 100 U ml−1 penicillin–streptomycin, 1× non-essential amino acids, 1 mM sodium pyruvate, 5 mM HEPES, β-mercaptoethanol). Plat-E and 293 GP cell lines were cultured in complete Dulbecco’s modified Eagle medium (DMEM (Gibco, 11995-065), 10% FBS, 2 mM l-glutamine, 100 U ml−1 penicillin–streptomycin, 1× non-essential amino acids, 1 mM sodium pyruvate, 5 mM HEPES, β-mercaptoethanol). The NALM6 cell line was a gift from TCR2 therapeutics. Cell lines were free of mycoplasma contamination. Cell cultures were maintained at 37 °C with 5% CO2 in a humidified incubator.
Chemical reagents
Rapamycin (mTORC1 inhibitor; Sigma, 553210) was used at 5 mg kg−1 dose in vivo wherever mentioned. Puromycin (Sigma, P8833) was used at 10 mg ml−1 concentration in vitro. OVA peptide (257–264) (SIINFEKL) (Genscript, RP10611) was used at 10 μg ml−1. Itraconazole was used at 2 μM concentration in vitro. For blocking the insulin receptor in vivo, S961 (1 mg kg−1; MedChemExpress, HY-P2093) was intraperitoneally (i.p.) injected during the fasted and fed periods.
Mouse T cell isolation and culture
Mice were fasted (by removal of food without interruption of water supply) or fed ad libitum. To isolate CD8+ T cells from mice, the spleen and three lymph nodes (axillary, brachial and inguinal) from both the right and left sides were harvested. The spleen was minced, treated with ACK lysis buffer (0.15 M NH4Cl + 1.0 mM KHCO3 + 0.1 mM Na2-EDTA) for 2 min to lyse the erythrocytes and mixed with pooled and minced LN cells. Naive (CD62LhighCD44low) CD8+ T cells were then purified from total pooled lymphocytes according to the manufacturer’s instructions (Miltenyi Biotec, 130-096-543). For in vitro expansion, naive CD8+ T cells were stimulated with anti-CD3 and CD28 mAb-coated dynabeads (Thermo Fisher Scientific, Gibco, 11452D).
Heat-inactivation treatment of serum
Preprandial or postprandial serum was heat-inactivated at 56 °C for 30 min, followed by centrifugation at 15,000 rpm for 20 min. Supernatant was collected and stored at −20 °C.
Chylomicron isolation and in vitro treatment
Chylomicrons were isolated as described previously49. Triglyceride concentration and 3H radioactivity was measured using a triglyceride assay (Randox) and scintillation counter (Beckman Coulter). CD8+ T cells from fasted mice were obtained as described above. Before lipid uptake experiments, CD8+ T cells were deprived of FBS for 1 h to ensure minimal lipid occurrence, and all lipid uptake experiments were carried out in FBS-free RPMI medium. Chylomicrons containing 3H-triglyceride were added at a concentration of 100 μg ml−1 of triglyceride in FBS-free medium for 1.5 h at 37 °C. For in vitro treatment, chylomicrons were added before TCR stimulation for 6 h in complete medium.
Flow cytometry: staining and analysis
Viability staining with Zombie dyes (BioLegend) and surface staining were performed in phosphate-buffered saline (PBS) at 4 °C for 20 min. Samples were then washed, and flow sample events were immediately recorded on a BD LSRFortessa Flow Cytometer. For intracellular transcription factor staining, following viability and surface staining, cells were incubated in 4% paraformaldehyde at room temperature for 15 min, washed in PBS, permeabilized with an eBioscience Foxp3/Transcription Factor Staining Kit (Thermo Fisher Scientific, 00-5523-00) per the manufacturer’s protocol, washed in permeabilization buffer, then stained in permeabilization buffer at 4 °C overnight in the dark. For intracellular cytokine staining, following viability and surface staining, cells were fixed in BD Cytofix/Cytoperm Fixation per the manufacturer’s protocol, washed in permeabilization buffer, then stained in permeabilization buffer at 4 °C overnight in the dark, followed by washing with fluorescence-activated cell sorting (FACS) buffer (PBS, 0.5–1.0% bovine serum albumin (BSA) or 5–10% FBS, 0.1% NaN3 (sodium azide)). All flow sample events were recorded on a Fortessa (4-laser; BD Biosciences) and analysed using FlowJo v.10. Cell doublets were excluded by comparison of side-scatter and forward-scatter width to area.
For staining of surface proteins in murine CD8+ T cells, the following anti-mouse mAbs were used to detect the respective antigens during FACS staining: CD8 (clone 53-6.7), CD62L (clone MEL-14), CD44 (clone IM7), anti-CD4 (clone GK1.5), Thy1.1 (clone OX-7), Thy1.2 (clone 53-2.1), anti- CD69 (clone H1.2F3) from BioLegend. Anti-murine T-bet (4B10) and Ki67 (SolA15) were purchased from eBioscience. Anti-human/mouse TOX was purchased from eBioscience (clone TXRX10). Anti-TCF-1 was obtained from Cell Signaling (clone C63D9). mAbs to cytokines (anti-IFNγ (clone XMG1.2), anti-TNF (clone MP6-XT22), anti-IL-2 (clone JES6-5H4) and anti-granzyme B (clone GB11); all from BioLegend) were added (the concentration was preoptimized), followed by incubation for 20 min at 4 °C and washing with FACS buffer.
Staining for human proteins was performed with human-specific antibodies: anti-CD4 (clone OKT4), anti-CD8 (clone HIT8a), anti-CD44 (clone BJ18), anti-CD62L (clone DREG-56), anti-CD69 (clone FN50), anti-IFNγ (clone 4S.B3), anti-TNF (clone Mab11), anti-granzyme B (clone QA16A02), anti-IL-2 (MQ1-17H12), anti-CD45 (2D1), anti-CD45RA (HI100), anti-CD45RO (UCHL1) and anti-CCR7 (G043H7).
For intracellular cytokine staining, CD8+ T cells were restimulated with PMA + ionomycin and transport blocker brefeldin A (eBioscience, 4506-51; 5 μg ml−1) and monensin (eBioscience, 4506-51 and 4505-51; 2 μM) for 6 h at 37 °C in a 5% CO2 humidified incubator.
EdU flow cytometry
Cell proliferation was assessed using a Click-iT Plus EdU flow cytometry assay kit (Life Technologies, C10633). EdU (5-ethyl-2′-deoxyuridine) is a thymidine nucleoside analogue that is incorporated into DNA during DNA synthesis. Detection is based on a click reaction in which a copper-catalysed covalent reaction occurs between a picolyl azide (coupled to Alexa Fluor 488 dye) and an alkyne group (present in the ethynyl moiety of EdU). EdU was added to the cell culture for at least 4–6 h for detection, and cells were fixed and permeabilized following the instructions provided with the kit. The incorporated EdU was then detected using picolyl azide conjugated with fluorescent dye Alexa Fluor 488 in the presence of a copper protectant for 30 min at room temperature. Following incubation, cells were washed in Dulbecco’s PBS and resuspended in Dulbecco’s PBS before being run on a BD LSRFortessa Flow Cytometer.
Phospho protein staining
Cells were surface stained and live/dead stained with Zombie. After surface staining, cells were washed once with PBS, followed by paraformaldehyde fixation for 1 h at 4 °C. Then, cells were resuspended in ice-cold methanol for 1 h, washed, and stained with anti-puromycin (BioLegend, clone 2A4, 381508), anti-p-S6 (BD Bioscience, clone N7-548, 560433), anti-p-AKT-S473 (Cell Signaling, clone D9E, 4071S) and anti-p-4E-BP1 (BD Bioscience, clone M89-61, 562599) for 30 min on ice.
Metabolic assays
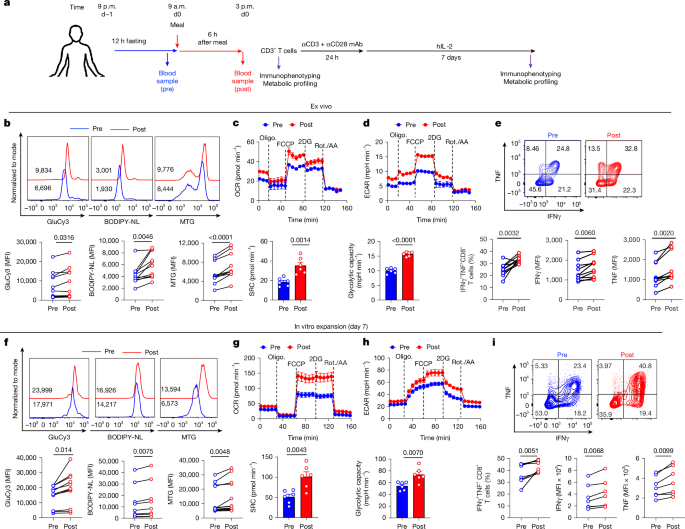
Cells were stained with MitoTracker Green (0.125 mM; Life Technologies) and BODIPY-NL (493/503) for assessment of mitochondrial mass (or size) and neutral lipid content, respectively. Glucose uptake was assessed by staining with GluCy3. Cells were washed twice with DPBS followed by addition of dye solution with preoptimized concentrations in serum-free RPMI medium and incubated at 37 °C in a 5% CO2 humidified incubator for 30 min. After incubation, cells were washed twice with DPBS followed by surface staining. Metabolic stains were assayed live on an BD LSRFortessa Flow Cytometer. To stain cholesterols specifically among lipids, Fillipin III (Millipore, F4767) was used after fixation and permeabilization of the cells for 30 min at room temperature.
Measurement of mitochondrial respiration
The OCR of treated cells was measured using an XFe96 Extracellular Flux analyser (Seahorse Biosciences). On the day of experiment, a Mito stress test assay medium was prepared by mixing Seahorse XF base medium (minimal DMEM) with l-glutamine (2 mM), sodium pyruvate (1 mM) and glucose (10 mM) and maintained at a pH of 7.4. All the inhibitors (including oligomycin (2 μM), carbonyl cyanide p-trifluoromethoxyphenylhydrazone (FCCP) (2 μM), 2-deoxy-d-glucose (10 mM) and a mix of rotenone (0.5 μM) and antimycin A (0.5 μM)) were prepared in assay media per the manufacturer’s recommendation, and the machine was calibrated using calibrant buffer in the calibrant plate before the experiment. Cells (150,000 per well) were seeded in poly-d-lysine precoated XFe96 plates, and the OCR was measured. Various parameters were calculated from the OCR graph: basal respiration, defined as (last rate measurement before oligomycin) − (non-mitochondrial respiration); ATP turnover, defined as (last rate measurement before oligomycin) − (minimum rate measurement after oligomycin injection); maximal respiration, defined as (maximum rate measurement after FCCP) − (non-mitochondrial respiration); and SRC, calculated by subtracting basal respiration from maximal respiration. To measure fuel dependency, inhibitors of specific nutrient pathways were used; these included UK5099 (2 μM), an inhibitor of the glucose oxidation pathway that blocks the mitochondrial pyruvate carrier; BPTES (3 μM), an inhibitor of the glutamine oxidation pathway that allosterically inhibits glutaminase GLS1; and etomoxir (4 μM), which inhibits long-chain fatty acid oxidation by inhibiting carnitine palmitoyl-transferase 1A.
For ATP rate measurements, we followed the guidelines mentioned in Agilent’s ATP Rate Assay kit (Agilent, Part Number 103592-100). In brief, the mitoATP production rate (basal) was calculated as (last OCR rate measurement before first injection) − (minimum OCR rate measurement after oligomycin but before rotenone and antimycin A injection) × 2 × (P/O)] where P/O (phosphate/oxygen) ratio indicates molecules of ADP phosphorylated to ATP per atom of oxygen reduced. An average value of 2.75 is often used for efficient cellular respiration; the GlycoATP production rate (basal) was calculated as the last glycoPER measurement before first injection; the total ATP production rate (basal) was calculated as (mitoATP production rate) + (glycoATP production rate); and the XF ATP rate index (basal) was calculated as (mitoATP production rate)/(glycoATP production rate).
Bone marrow chimera mice generation
C57BL/6J (Thy1.1 background) and LDLR-deficient (Thy1.2 background) mice were euthanized, and bone marrow cells were collected. ACK lysis buffer treatment was performed to lyse RBCs. Single-cell suspensions from WT and Ldlr−/− mice were mixed in equal numbers to yield a 50:50 ratio. The mixed bone marrow suspension was cotransferred intravenously by tail vein injection into preirradiated Rag2−/− mice (600 rad, whole-body radiation). After 1 month, immune T cell reconstitution was confirmed by staining conventional CD4 and CD8 T cell markers and congenic markers (Thy1.1 and Thy1.2).
Western blot
Cells were lysed in lysis buffer, and protein amounts were quantified by a colorimetric method using a BCA protein assay kit (Thermo Fisher, 23225). Then, 10–15 μg of total protein was loaded in each well for western blotting. Proteins were blotted on nitrocellulose membranes. First, the blots were blocked using 0.5% BSA in Tris-buffered saline solution. Then, bands were detected using the respective primary antibodies and secondary antibodies tagged with HRP and developed and imaged on x-ray films. For quantification of mitochondrial complex proteins, mitochondrial fractions of T cells were obtained using a mitochondria isolation kit (Thermo Scientific, 89874) following the manufacturer’s instructions. The following antibodies were used to blot proteins: anti-puromycin mAb (Cell Signaling, clone 12D10, MABE343); anti-mouse p27 (Kip1) mAb (Cell Signaling, clone D69C12, 3686T); anti-mouse β-actin (Santa Cruz Biotechnology, clone C4, sc-47778); anti-mouse HSP60 (Novus Biologicals, clone 1C7, NBP1-77397); and total OXPHOS rodent western blot antibody cocktail containing antibodies against all five complexes of electron transfer chain (Abcam, ab11013).
Murine tumour growth curve and analysis
Tumour cells were intradermally injected into the dorsal side anterior to the tail. Adoptive T cell transfer was performed on day 6, when tumour size was about 25–30 mm2. CD8+ T cells from fasted or fed Pmel-1 mice were TCR stimulated and in vitro expanded up to day 7. Five million expanded fasted or fed Pmel-1 CD8+ T cells were intravenously (through the tail vein) transferred into B16-tumour-bearing hosts in different groups. For untreated mice, a suitable negative control or PBS was injected. Tumour size was measured every third day using a digimatic calliper (Mitutoyo Europe GmbH), and tumour size (area) was calculated using the formula length × breadth. For murine tumour growth with lymphodepletion, tumour-bearing mice were injected with cyclophosphamide monohydrate (Cytoxan) (200 mg kg−1, i.p.; Sigma C0768) 1 day before adoptive T cell transfer (200,000 cells only per mouse).
Human T cell isolation
Human metabolic preprandial and postprandial status was studied under the guidelines of the Institution Review Board of the University of Pittsburgh/UPMC (IRB21090049). Informed consent was obtained from all participants included in the study. For the preprandial blood draw, blood was drawn from healthy participants between 8 a.m. and 9 a.m. after a fast of at least 12 h; this blood draw was followed by a meal. The next blood draw (postprandial blood draw) was performed after 5–6 h during which healthy participants were asked to eat normally. The blood samples were centrifuged at 1,500 rpm for 10 min. Supernatant (plasma) was collected and stored at −80 °C. The cellular precipitate was further resuspended in HBS medium. Ficoll gradient solution was added very slowly to the cellular resuspension, followed by centrifugation at 2,000 rpm for 20 min. Cells were collected carefully with a pipette from the middle cloudy layer and washed once with RPMI medium to remove any Ficoll or HBS solution. Total CD3+ (BioLegend, 480131) or CD8+ (BioLegend, 480129) T cells were enriched following the negative selection protocol.
Human CAR-T cell generation
X–Vivo 15 media (Lonza) supplemented with 5% human AB serum (Valley Biomedical), 10 mM HEPES buffer and 2 mM GlutaMAX (Gibco) were used for in vitro culture of human CAR-T cells. Purified human CD3+ T cells (as described above) were stimulated with TCR. Frozen peripheral blood mononuclear cells (PBMCs) were thawed, and CD3+ T cells were isolated by negative selection with a kit (BioLegend, 480131) following the manufacturer’s protocol. CD3+ T cells were then activated with TransAct (Miltenyi Biotec, 1:100; 130-111-160) in the presence of 200 IU ml−1 human IL-2 (Cellgenix) for 48 h, followed by retroviral transduction (with retroviruses encoding an hCD19-targeted CAR with a 4-1BB costimulatory domain)44. Briefly, the retrovirus was preloaded through spin-transduction (2 h, 2,000g) on a non-tissue-culture retronectin-coated plate. Then, CD3+ T cells were added and briefly spun down. The CD3+ T cells were incubated for 48 h and then FACS sorted, after which they were transferred to a G-Rex 6M Well Plate (Wilson Wolf) for expansion. Cells were collected on day 10 for further analysis or adoptive transfer. For human CAR-T cell activation cytokine assays, human CAR-T cells were incubated with target cells at a 1:1 ratio for 6 h at the presence of GolgiPlug (BD, 1:1,000) and then stained for cytokines.
Killing assays (by eSight)
For assessment of killing efficiency, we used a xCelligence RTCA DP eSight device (Agilent Technologies, 380601600), which measures impedance. Only adherent cells can be used for killing assays with this system. Five to ten thousand cancer cells were seeded in a plate in 50 ml complete RPMI medium and left to attach to the bottoms of wells. Pre- and post-CAR-T cells were seeded in a ratio gradient. Impedance was measured every 5 min, and an image was taken every 15 min by the machine. Killing efficiency was calculated relative to control wells to which no killer T cells were added.
In vivo therapeutic T cell experiments
For murine tumour growth curves, 125,000 B16 murine melanoma cells were injected intradermally into C57BL/6J mice. Once tumours became palpable or visible (1–4 mm2), cyclophosphamide 200 mg kg−1 was injected i.p. The following day, the mice were randomized on the basis of tumour size. Then, 0.5 million (unless reported otherwise) murine fasted or fed T cells were transferred intravenously by tail vein injection. Tumour growth was monitored 2–3 times per week. A survival event was recorded when a tumour ulcerated or surpassed 1.5 cm in diameter, or when a mouse died.
For human CAR-T cell experiments with NALM6, 0.5 million luciferase-expressing NALM6 cells were injected intravenously (through the tail vein) into NSG mice. The mice were then imaged 3 days afterward with IVIS and randomized into three groups. One group received 0.5 million human pre-CAR-T cells, another received post-CAR-T cells, and the third group was injected with untransduced T cells intravenously by tail vein injection. The number of CAR-T cells was always adjusted on the basis of the percentage of transduction of the product. The leukaemia burden was monitored with IVIS twice in a week, and a survival event was recorded when a mouse exhibited morbidity or died.
RNA and genomic sequencing and analysis
Cells were enriched by means of negative selection using magnetic-activated cell sorting or sorted using a Sony MA900 cell sorter from the single-cell suspension of splenocytes from experimental mice or from PBMCs of human donors in fasted and fed states. For bulk RNA-seq, total RNA was isolated from sorted CD8+ T cells using RNeasy Mini Kit (Qiagen). Purified RNA was sequenced at the Health Sciences Sequencing Core at Children’s Hospital of Pittsburgh. RNA-seq data were analysed performed using CLC Genomics and R. Only genes that had false discovery rate P < 0.05 were considered significant. A volcano plot was constructed using all genes with P < 0.05.
For ATAC-seq analysis, ex vivo cells and cells stimulated for 24 h with TCR in vitro were processed following the kit manual (Active Motif 53150). Cells (50,000 to 100,000) were placed in a 1.5-ml tube, washed with 1 ml cold PBS + 1% BSA to remove debris and contaminants, and lysed in 100 μl ATAC lysis buffer on ice for 10 min. Nuclei were isolated by centrifugation at 500g for 5 min at 4 °C. The supernatant was discarded, the pellet was washed gently with cold PBS + 1% BSA, and nuclei were resuspended in tagmentation reaction buffer containing 1% digitonin, 10% Tween-20 and assembled transposomes and incubated at 37 °C for 30 min. After incubation, 250 μl DNA purification binding buffer and 5 μl 3 M sodium acetate were added, and tagmented nuclei were purified using a column. Tagmented DNA was further PCR amplified with a combination of one i7 indexed primer and one i5 indexed primer in the presence of Q5 polymerase. The protocol was as follows: initial denaturation at 72 °C for 5 min and 98 °C for 30 s; followed by 10–15 cycles at 98 °C for 10 s, 63 °C for 30 s and 72 °C for 1 min; and final extension at 72 °C for 5 min. Amplified DNA was purified using SPRI magnetic beads, libraries were quantified using Qubit or Bioanalyzer, and fragment size was assessed (approximately 200–600 bp).
Proteome analysis by mass spectrometry
Cell pellets were lysed in 5% SDS, 100 mM triethylammonium bicarbonate (TEAB; Sigma-Aldrich), and 1× HALT Protease and Phosphatase inhibitor (Thermo Fisher Scientific). Protein content was measured using Pierce 660 nm protein assay reagent (Thermo Fisher Scientific) and adjusted to 10 mg per sample input. Proteins were reduced with 100 mM dithiothreitol (Sigma-Aldrich) for 10 min at 95 °C, followed by alkylation with 50 mM iodoacetamide (Sigma-Aldrich) for 30 min at room temperature. Proteins were acidified by addition of 12% phosphoric acid to a final concentration of 1.2%. The acidified proteins were diluted 1:7 in a binding buffer of 90% methanol in 100 mM TEAB. After dilution, the proteins were added to an S-trap micro column (ProtiFi) and washed three times with 90% methanol in 100 mM TEAB. Samples were digested using 1 mg of trypsin (Promega) in 50 mM TEAB at 55 °C for 1.5 h. Peptides were eluted sequentially in three steps with 50 mM TEAB, 0.2% (v/v) formic acid in water and 50% acetonitrile. Eluents were combined and desalted using C18 columns (Supelco) on a vacuum manifold. The resulting peptides were dried using a vacuum centrifuge, followed by resuspension in 0.1% (v/v) formic acid. Peptides were analysed by liquid chromatography tandem mass spectrometry using a nanoACQUITY HPLC (Waters) and Q Exactive HF mass spectrometer (Thermo Fisher Scientific).
Briefly, solvent A (0.1% formic acid in water) and solvent B (0.1% formic acid in acetonitrile) were used for the mobile phase. Peptides were eluted on to a capillary column (75 µm ID × 360 µm OD × 15 cm length; Polymicro Technologies) containing C18 stationary phase (5 µm particle size, 125 Å pore size; Phenomenex) and resolved using a 100-min gradient with a flow rate of 200 nl per min (3–33% B for 90 min, 33–80% B for 2 min, constant at 80% B for 6 min, then 80–0% B for 2 min to equilibrate the column). Data were collected in positive mode, and PEAKS studio 11.5 (Bioinformatics Solutions) was used to search against the UniProt murine database. The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium through the PRIDE50 partner repository with dataset identifier PXD057581.
Untargeted liquid chromatography high-resolution mass spectrometry (13C enrichment in PPP flux)
Sample preparation
Metabolic quenching and polar metabolite pool extraction were performed by addition of ice-cold 80% methanol (aqueous) at a ratio of 500 µl buffer per 1 × 106 cells. Deuterated (D3)-creatinine and (D3)-alanine, (D4)-taurine and (D3)-lactate (Sigma-Aldrich) were added to the sample lysates as internal standards for a final concentration of 10 µM. Samples were further homogenized using a water bath sonicator at 25 °C, and the supernatant was then cleared of protein by centrifugation at 16,000g. Finally, 2 µl of cleared supernatant was subjected to online liquid chromatography mass spectrometry analysis.
Liquid chromatography high-resolution mass spectrometry method
Samples were analysed by untargeted liquid chromatography high-resolution mass spectrometry. Briefly, samples were injected via a Thermo Vanquish UHPLC and separated over a reversed-phase Thermo HyperCarb porous graphite column (2.1 mm × 100 mm, 3 μm particle size) maintained at 55 °C. For the 20-min liquid chromatography gradient, the mobile phase consisted of solvent A (water/0.1% formic acid) and solvent B (acetonitrile/0.1% formic acid). The gradient was as follows: 0–1 min 1% B, increased to 15% B over 5 min, with a continued increase to 98% B over 5 mins, followed by a hold at 98% B for 5 min and re-equilibration at 1% B for 5 minutes. A Thermo ID-X Tribrid mass spectrometer was operated in negative ion mode, scanning in ddMS2 mode (2 μscans) from 70 to 800 m/z at 120,000 resolution with an AGC target of 2e5 for full scan and 2e4 for ms2 scans using HCD fragmentation at stepped 15,35,50 collision energies. The source ionization setting was 2.4 kV spray voltage for negative mode. Source gas parameters were 35 sheath gas, 12 auxiliary gas at 320 °C, and 8 sweep gas. Calibration was performed before analysis using Pierce FlexMix Ion Calibration Solution (Thermo Fisher Scientific). Integrated peak areas for pentose phosphate intermediates were then extracted manually using Quan Browser (Thermo Fisher Xcalibur v.2.7). Purified standards and internal chemical libraires were then compared in retention time, m/z, along with ms2 fragmentation patterns to validate the identity of peaks.
Statistical analysis
The data presented in the figures are mean ± s.e.m. Wilcoxon matched-pairs signed-rank test and Wilcoxon rank-sum test were used for paired and unpaired data, respectively. Survival data are presented as Kaplan–Meier survival curves and were analysed by non-parametric log-rank test. Tumour growth curves were analysed with repeated-measures analysis of variance. All analysis was completed with Prism v.7 software (GraphPad) and confirmed with STATA v.17.0. P < 0.05 was considered to indicate statistical significance. In the figures, standard designations of significance are given: *P < 0.005, **P < 0.01, ***P < 0.001, ****P < 0.0001. The specific analysis used per figure can be found in the legends.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.