Cloning, protein expression and purification
The DNA encoding ThermoCas9 with a C-terminal His6 tag was integrated into the pML-1B vector and expressed in the Escherichia coli NiCo21(DE3) strain. Cells were grown in Luria–Bertani (LB) medium with 0.2% d-(+)-glucose at 37 °C until optical density at 600 nm reached 0.8, at which point addition, isopropyl-β-d-thiogalactopyranoside was added to 0.5 mM concentration. Cells were grown for an additional 16–18 h at 20 °C and harvested by centrifugation and stored in −80 °C. Previously frozen cells were lysed via sonication in a lysis buffer (500 mM NaCl, 50 mM phosphate buffer pH 8.0 (sodium phosphate dibasic and sodium phosphate monobasic), 5 mM imidazole and 1 mM β-mercaptoethanol) containing 1 tablet of cOmplete Mini Protease Inhibitor Cocktail (Sigma-Aldrich) per 100 ml. The lysate was centrifuged at a speed of 16,000 rpm for 60 min at 4 °C, after which the supernatant was loaded on a pre-equilibrated 5-ml HisTrap HP His tag protein purification column (Cytiva Life Sciences). The resin was subsequently washed with 200 ml wash buffer (500 mM NaCl, 50 mM phosphate buffer pH 8.0, 30 mM imidazole and 1 mM β-mercaptoethanol), before being eluted with elution buffer (500 mM NaCl, 50 mM phosphate buffer pH 8.0, 250 mM imidazole and 1 mM β-mercaptoethanol). The resultant eluate was transferred onto a pre-equilibrated HiTrap Heparin HP affinity column (Cytiva Life Sciences) and eluted with a 100 mM to 2 M NaCl gradient. The purified protein was then concentrated and stored at −80 °C until further use.
For purification of ThermoCas9 used in human gene-editing experiments, the DNA encoding 3×-nuclear localization sequence (2× SV40 NLS and 1× nucleoplasm NLS) fused with ThermoCas9 with a C-terminal His6 tag was integrated into the pML-1B vector and expressed in E. coli Rosetta (DE3) cells. The same purification method was used with the exception that the gel-filtration buffer was made with cytotoxin-free water.
In vitro RNA transcription
We used the T7 in vitro transcription method to produce the sgRNA for both ThermoCas9 and AceCas9. The sgRNA templates containing a T7 promotor were purchased from Eurofins Genomics. A 149 nt sgRNA for ThermoCas9 and a 106 nt sgRNA for AceCas9 (Supplementary Table 1), respectively, were transcribed by T7 RNA polymerase in a transcription buffer (5 mM NTPs, 50 mM Tris-HCl pH 7.5, 15 mM MgCl2, 5 mM dithiothreitol and 2 mM spermidine) and purified via the Monarch RNA Cleanup Kits (New England Biolabs). The DNA used in cryo-EM and biochemical assays was purchased from Eurofins Genomics.
Cryo-EM sample preparation, data collection and 3D reconstruction
The heparin-purified protein was incubated with sgRNA at a 1:1.5 molar ratio at 37 °C for 30 min, and the resulting RNP was further purified via size-exclusion chromatography with a Superdex 200 10/300 GL column (Cytiva Life Sciences) in gel-filtration buffer (300 mM NaCl, 30 mM HEPES pH 7.5 and 1 mM dithiothreitol). The Cas9–RNA–DNA ternary complex was assembled by adding pre-annealed substrate dsDNA into the RNP at a 2:1 molar ratio with the presence of 10 mM magnesium chloride. The reactive ternary complex was incubated at 37–50 °C for 15–30 min. Of the sample, 4 µl was added to glow-discharged Gold 300 mesh R1.2/1.3 grids, which was then allowed to adsorb for 30 s before blotting for 2.5 s under conditions of 20 °C and 100% humidity. These grids were rapidly frozen in liquid nitrogen cooled ethane within Vitrobot Mark IV.
Raw micrographs of ThermoCas9 bound with DNA containing 5′-NNNNCCA-3′ PAM and AceCas9 bound with DNA containing 5′-NNN5mCC-3′ PAM were collected at the Laboratory for Biomolecular Structure of the Brookhaven National Laboratory using a Titan Krios G3i cryo transmission electron microscope equipped with a Gatan K3 direct electron detector. Raw micrographs of ThermoCas9 bound with DNA containing 5′-NNNNCGA-3′ PAM were collected at the Pacific Northwester Center for Cryo-EM using a Titan Krios Electron Microscope equipped with a Gatan K3 direct electron detector (Thermo Fisher Scientific). Movies were recorded at a nominal magnification of 105,000 in a super-resolution mode with an energy filter of 15 eV, corresponding to a corrected physical pixel size of 0.82 Å per pixel. A total dose of 50–60 e− Å−2 was spread over 60 frames with random defocus set to −0.8 to −2.5 µm. Motion correction was executed in bin 2 via MotionCorr2 and contrast transfer function (CTF) estimation was carried out with Gctf61. A total of 6,080 micrographs were collected and 2,516,939 particles were picked using Topaz62, followed by multiple rounds of 2D classification using cryoSPARC63, resulting in 2,015,088 good particles for 3D classification. After heterogenous refinement in cryoSPARC, the dataset was classified into five classes. Several rounds of 3D refinement and 3D classification were then performed using Relion 4.0 (ref. 64) to obtain high-quality particles. Finally, several rounds of non-uniform refinement65 were performed using cryoSPARC to reach the final 3D structures. Structural models were built in COOT66 and refined in PHENIX67 to satisfactory stereochemistry and real-space map correlation parameters. Note that water molecules were only modelled based on both density and interaction chemistry in the two high-resolution structures.
Bacterial survival assay
The survival assay in bacterial cells followed a previously outlined procedure44 with minor modifications. In brief, electrocompetent E. coli BW25141 cells, harbouring the modified p11-LacY-wtx1 plasmid encoding toxic ccdB protein, were transformed with 60 ng of WT or mutant ThermoCas9 plasmids. Afterwards, the cells were recovered in LB for 30 min with shaking at 37 °C. Subsequently, 0.05 mM isopropyl-β-d-thiogalactopyranoside was introduced, and the recovery process continued for an additional 60 min. The recovered cells were then plated on LB agar plates containing either chloramphenicol (15 mg ml−1) or a combination of chloramphenicol and 10 mM arabinose. The plates were incubated at 37 °C for 16–20 h. Manual counting of colonies was performed on both plates, and survival rates were determined by dividing the CFUs on arabinose-containing plates by those on chloramphenicol-only plates. For directed evolution of ThermoCas9, a library of ThermoCas9 linker II variants were transformed into BW25141 cells harbouring a modified p11-LacY-wtx1 plasmid containing a PAM-distal truncated protospacer of 17 nucleotides (17-mer) in the same manner as stated above. CFUs that grew on arabinose in the 17-mer cells were selected for Sanger sequencing.
In vitro DNA cleavage assay and competition assay
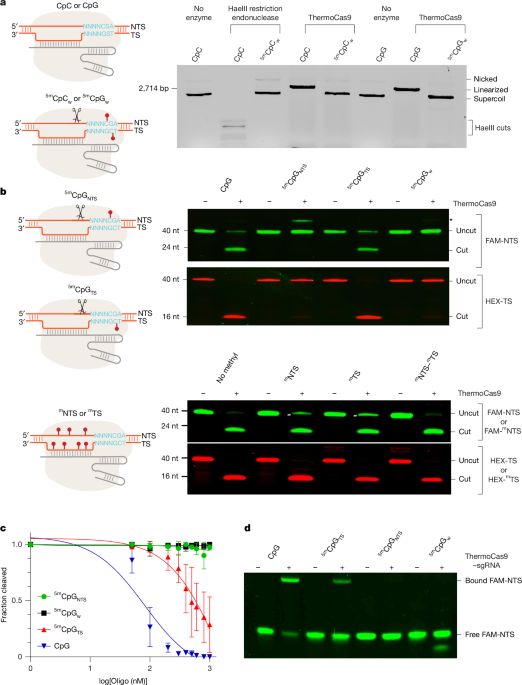
ThermoCas9 was combined with sgRNA at a 1:2 ratio and incubated at 37 °C for 30 min to form the RNP. The target plasmid at 6 nM was then added to the RNP at 1 µM and allowed to incubate for varying lengths of time. The reactions were stopped by adding a 5× stop buffer (25 mM Tris pH 7.5, 250 mM EDTA pH 8.0, 1% SDS, 0.05% w/v bromophenol blue and 30% glycerol). The reaction products were separated on a 0.8% agarose gel and stained by ethidium bromide.
Fluorescently labelled oligonucleotides were also used to prepare DNA substrates. Six-carboxyfluorescein (FAM)-labelled non-target strand DNA was annealed with an unlabelled target strand DNA at a 1:1 molar ratio. Separately, hexachlorofluorescein (HEX)-labelled target strand DNA was annealed with unlabelled non-target strand DNA at a 1:19 molar ratio. Annealing was performed by heating the DNA mixtures to 75 °C for 5 min, followed by a gradual cooling to room temperature. Pre-annealed dsDNA substrates were prepared at concentrations of 100–200 nM for the labelled strand. These substrates were then added to a ThermoCas9 RNP solution at 1 µM to initiate the cutting reaction. Divalent metal ions, specifically 10 mM of MgCl2, were also included in each reaction. The reaction mixtures were incubated at 37–50 °C for 1 h before adding 2× RNA loading buffer (97% formamide, 0.02% SDS and 1 mM EDTA). The reaction products were resolved using a 7 M urea 20% polyacrylamide denaturing gel. Gel electrophoresis was performed under denaturing conditions to ensure the separation of DNA fragments based on size. Following electrophoresis, the gel was visualized using a Bio-Rad ChemiDoc gel imaging system. Fluorescent labels were detected using excitation wavelengths of 488 nm for FAM and 580 nm for HEX.
For competition assays, ThermoCas9 RNP at 1 μM was mixed with the target plasmid at 10 nM, and a competing oligo DNA substrate at concentrations of 50 nM to 1 μM. The reactions were incubated at 50 °C for 15 min and stopped by adding the 5× stop buffer. The reaction products were separated on a 0.8% agarose gel and stained by ethidium bromide. The fraction of cleavage versus oligo concentration plots were fitted to a competitive one-site binding model in GraphPad to yield the estimated binding constant of each competing oligo (Ki).
Native gel-binding assay
FAM-labelled non-target strand DNA was annealed with an unlabelled target strand DNA at a 1:1 molar ratio. dsDNA (100 nM) was mixed with 1 μM ThermoCas9 RNP in the reaction buffer without MgCl2 for 1 h. The reaction product was then mixed with 6X purple gel loading dye (New England Biolabs) and loaded onto a 10% TBE gel (Invitrogen) for electrophoresis.
In vitro methylation screening
Genomic DNA from HEK293T and HCT116 cells was extracted with QuickDNA microprep kit (Zymo Research). The extracted genomic DNA was then incubated with 125–250 nM ThermoCas9 RNP in the reaction buffer containing 5 mM MgCl2 at 37 °C for 30–45 min. The reaction product was treated with E.Z.N.A. Plasmid DNA Mini Kit Solution I (Omega Bio-tek) for 10 min at a 1:1 volume ratio. DNA was subsequently cleaned up using the Monarch PCR & DNA Cleanup Kit (New England Biolabs). For PCR amplification, 1 µl of the reaction product was mixed with 0.25–1 µM primers and Q5 High-Fidelity 2X Master Mix (New England Biolabs). The PCR product was then mixed with 6X blue gel loading dye (New England Biolabs) and loaded onto a 2% agarose gel with a 100-bp DNA ladder (New England Biolabs) for electrophoresis.
In silico analysis of differentially methylated sites in human cells
Reduced representation bisulfite sequencing (RRBS) data were collected from the ENCODE functional genomics database for various cell lines68. We downloaded the call sets (bed files) from the ENCODE portal69 (https://www.encodeproject.org/) with the following identifiers: ENCFF001TMR, ENCFF001TMQ, ENCFF001TMS and ENCFF001TMT for the HEK293T cell line and ENCFF001TMM and ENCFF001TMN for the HCT116 cell line. An in-house program was used to compare the methylation profiles based on the methylation scores. The RRBS methylation profiles across various genetic loci in different cell lines were visualized using the Integrative Genomics Viewer70. An in-house program based on Python scripts and bed utilities was used to identify genes that are differentially methylated in different cell lines.
Transfections and gene editing in HEK293T and HCT116 cells using plasmid DNA
Human-codon-optimized thermocas9-sv40nls gene and its sgRNA module were expressed under the control of the constitutive cytomegalovirus (PCMV) and U6 RNA polymerase III (PU6) promoters, respectively (Supplementary Table 1). We co-expressed the EGFP reporter gene under the constitutive elongation factor 1α promoter (PEF1α) to allow for sorting of successfully transfected cells, as previously described27. We designed four spacers that target protospacers in the chromosomal genes VEGFA, EMX1 and PRDX4. All differentially methylated protospacers were flanked by a PAM of (5′-NNNNCGAA-3′) thus representing a potential CpG methylated PAM. The targeting spacers of EMX1 and PRDX4 are differentially methylated in the PAM sequence between HEK293T and HCT116; the negative and positive control targets are located on the VEGFA gene. HCT116 cells were maintained in McCoy’s 5A media and HEK293T cells were maintained in DMEM media supplemented with 10% fetal bovine serum and 1% penicillin–streptomycin at 37 °C with 5% CO2. Both HEK293T and HCT116 cells were seeded on physically surface-treated 24-well plates (Corning/Falcon) at a seeding density of 1.0 × 105 cells per well. After 24 h of incubation, 0.5 μg of genome-editing plasmid was transfected into the HEK293T and HCT116 cells using Lipofectamine 3000 Transfection reagent (L3000015, Thermo Fisher). For each well on the plate, transfection plasmids were combined with OptiMEM Reduced Serum Medium (31985062, Thermo Fisher) to a total volume of 25 µl and mixed with 1 µl P3000 reagent. Separately, 25 µl OptiMEM was combined with 1.1 µl Lipofectamine 3000 reagent. Plasmid and Lipofectamine solutions were then combined, incubated at room temperature for 10 min and pipetted on to cells. The transfected cells were cultured 72 h and further evaluated for the presence of GFP using fluorescence-activated cell sorting (FACS). For SpyCas9 gene-editing experiments, HEK293T cells were transfected with 0.5 μg of plasmid co-expressing SpyCas9 and sgRNA (Addgene #42230). The transfection methods were consistent with ThermoCas9, except cells were harvested 48 h post-transfection for genomic DNA isolation without FACS sorting.
FACS
After 72 h of incubation, the transfected HEK293T and HCT116 cells were harvested, centrifuged at 1,000 rpm for 5 min, resuspended in 250 μl DMEM (10% FBS and 1% penicillin–streptomycin), and filtered through Nylon Mesh 52 micron, 32% open area filter (Component Supply Co.). GFP+ fluorescent cells were bulk sorted using the BD FACSAria III cell sorter device (BD; 488 nm laser, FITC detection channel for GFP fluorescence). The cells were gated for ‘high-green’ to reduce the signal to noise of auto-fluorescent cells (Supplementary Fig. 4). Cells were transferred to a 96-well nucleon plate and centrifuged at 200 rpm for 2 min and cultured for approximately 1–2 weeks (37 °C; 5% CO2). When approximately 75% confluency was reached, the propagated cells of each experiment were steadily passaged to 24-well plates and further screened for indels.
ThermoCas9-mRNA production and nucleofection
In vitro transcription reactions for ThermoCas9-mRNA (Supplementary Table 1) were assembled with T7 buffer (NEB), 100 mM ATP (NEB), 100 mM GTP (NEB), 100 mM CTP (NEB), 100 mM pseudo-UTP (Trilink), CleanCap AG (Trilink), human-codon-optimized ThermoCas9-NLS (Gene Fragment with Adapters Twist Bioscience; 108612) and T7 RNA Polymerase (NEB) and incubated at 37 °C overnight. The following day, the reaction was further treated with DNase enzyme (NEB) followed by Monarch spin RNA cleanup kit (500 µg column) before transfection.
For ThermoCas9-mRNA delivery, all transfections were performed with a 4D Lonza nucleofector. Before the addition of nucleofection buffers, cells were detached with TrypLE and washed with PBS pH 7.2 1X (Gibco) to remove potential RNases. The ThermoCas9-mRNA nucleofection conditions were as follows: 16.4 µl SF or SE nucleofection buffer supplemented with 3.6 µl Supplement 1; 1.0 × 105 cells; 1 µl of 100 µM µl−1 custom sgRNA (SC1518-CRISPR Oligo, Genscript) and 3.8 µg µl−1 CleanCap ThermoCas9 mRNA. Pulse codes for nucleofections were DS-150, EN-113, DS-137 and EN-150 for HEK293T (CRL-3216, American Type Culture Collection (ATCC)), HCT116 (CCL-247, ATCC), MCF-7 (HTB-22, ATCC) and MCF-10A (CRL-10317, ATCC), respectively. MCF-7 cells were maintained in EMEM media supplemented with a final concentration of 2 mM L-glutamine, 0.01 mg ml−1 insulin and 10% fetal bovine serum. MCF-10A cells were maintained in Lonza MEGM Mammary Epithelial cell Growth Medium BulletKit supplemented with 100 ng ml−1 cholera toxin and grown at 37 °C with 5% CO2. For RNP, all conditions are the same as above but RNP conditions were 1 µl of 100 µM µl−1 custom sgRNA (SC1518-CRISPR Oligo, Genscript), and 1 µl of 3 mg ml−1 ThermoCas9 protein. All nucleofections were conducted with a 16-well nucleocuvette strip within the 4D-Nucelofector X Unit. After applying the electroporation pulse, cells were allowed to rest within the nucleocuvette strip for approximately 10 min before adding 80 µl of respective media to transfer to a 24-well plate.
Screening for genome editing
HEK293T and HCT116 genomic DNA was isolated from the bulk population of propagated cells grown approximately 2–3 weeks post-FACS sorting. Genomic DNA was extracted using the Zymo Research Quick-DNA MicroPrep kit. Genomic target regions (VEGFA, EMX1 and PRDX4) were PCR amplified with Q5 High-Fidelity 2X Master Mix (New England Biolabs). The PCR products were verified on a 2% DNA agarose gel, and they were subsequently gel purified with the E.Z.N.A. gel extraction kit (Omega-BioTek). To detect indel formation, the gel-purified PCR products were subjected to Sanger sequencing (FSU sequencing facility). The sequencing results of the genome-editing assays were analysed using the Inference of CRISPR Edits (ICE) tool70 (EditCo) (Supplementary Figs. 7, 8 and 12–17). For ThermoCas9-mRNA and RNP editing experiments: HEK293T, HCT116, MCF-7 and MCF-10A genomic DNA was isolated from cells 72 h post-nucleofection. All downstream sample processing is the same as mentioned above. To detect modified reads from mRNA-treated or RNP-treated samples, the gel-purified PCR products were subjected to premium PCR sequencing by Plasmidsaurus using Oxford Nanopore Technology with custom analysis and annotation. The ThermoCas9-mRNA and RNP genome-editing sequence analysis was performed using CRISPResso2 by uploading FASTQ files as single-end reads and using the standard settings for Cas9.
Bisulfite sequencing
Genomic DNA of both HEK293T and HCT116 were bisulfite treated via the EpiJET Bisulfite Conversion Kit (K1461, Thermo Scientific) following the manufacturer’s instructions. The MethPrimer online tool was utilized to design primers to amplify bisulfite-converted samples flanking the regions of gene-editing targets followed by Sanger sequencing (FSU sequencing facility).
5mC interrogation by Infinium Methylation EPIC array
DNA was quantified by Qubit fluorometry (Promega) and 250 ng of DNA from each sample was bisulfite converted using the Zymo EZ DNA Methylation Kit (Zymo Research) following the manufacturer’s protocol using the specified modifications for the Illumina Infinium methylation assay. After conversion, all bisulfite reactions were cleaned using the Zymo-Spin binding columns and eluted in 12 µl of Tris buffer. Following elution, bisulfite-converted DNA was processed through the Infinium Methylation EPIC array v2.0 protocol (Illumina). The EPIC array v2.0 contains more than 930,000 probes querying methylation sites including CpG islands and non-island regions, RefSeq genes, ENCODE open chromatin, ENCODE transcription factor-binding sites and FANTOM5 enhancers. To perform the assay, 4 µl of converted DNA was denatured with 4 µl 0.1 N sodium hydroxide. DNA was then amplified, hybridized to the EPIC bead chip, and an extension reaction was performed using fluorophore-labelled nucleotides per the manufacturer’s protocol. Array beadchips were scanned on the Illumina iScan platform and probe-specific calls were made using Illumina Genome Studio software. ThermoCas9 target sites with contrasting methylation scores between the MCF-7 and the MCF-10 cells were identified from the processed EPIC array data using an in-house script.
Data processing for Infinium Methylation EPIC array
The R package SeSAMe71 was used to process Illumina microarray platform files in IDAT format generated from the EPIC v2.0 array, followed by downstream differential methylation locus (DML) and region analyses. The ‘openSesame’ function from SeSAMe was used to convert the files into DNA methylation level (β value) matrices in R. For DML detection, SeSAMe applies linear models to identify DMLs between two groups in a contrast. For differential methylation region analysis, neighbouring CpGs that show consistent methylation variation were merged into differentially methylated regions, and adjusted P values were calculated using the Benjamini–Hochberg procedure. Methylation sites were annotated using SesameData71, and additional annotation regarding genomic context and proximity to nearby genes was obtained from Noguera-Castells et al.72.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.