Our results show that thymic decay in adults is highly individualized and that loss of thymic health increases mortality and disease incidence, including cancer and cardiovascular diseases. These findings were investigated in two independent prospectively collected clinical studies of asymptomatic adults, the NLST and the FHS. Among more than 25,000 participants in the NLST, higher thymic health was consistently associated with a significantly lower risk of mortality by any cause, lung cancer and cardiovascular diseases, independent of sex, age, smoking, prior diseases or cancer history. Similar results were found in the independent FHS, where participants with high thymic health had a significantly reduced risk of death from cardiovascular disease, a primary endpoint of the study, independent of sex, age and smoking. Considering the prevailing perception of the limited role of the thymus in adults, our findings may change our understanding of human health, emphasizing the complex yet critical role of the immune system in long-term well-being and longevity.
We found that thymic health was associated with critical health consequences. Presumed healthy participants in the NLST with high thymic health had an approximately 50% reduction in the risk of death, were 36% less likely to develop lung cancer and nearly 50% less likely to die from lung cancer, as compared to participants with low thymic health. Further, the significant impact of high thymic health on cardiovascular disease (CVD) was consistent across participants in the NLST and FHS, with risk reductions in CVD mortality ranging from 63% to 92%. Given the critical role of the thymus in generating a diverse T cell repertoire24, these results support the broad impact of a sustained adaptive immune system to combat disease and promote longevity.
Our results challenge the established notion that the cessation of thymic output in ageing adults is inconsequential as it is naturally replaced by peripheral expansion of T cells7. Rather, our results suggest that loss of thymic tissue in adults may forecast higher risks of disease and death. The main function of the thymus is to generate a diverse T cell repertoire, which provides adaptive immunity throughout life7. While the relevance and abundance of the T cell repertoire at a young age are well documented7, our results indicate that the thymus retains a continued role in T cell production throughout adulthood and that the pattern of decline of thymic function in adults is associated with poorer health outcomes.
A recent landmark study demonstrated an association between thymectomy and reduced lifespan and increased risk of cancer, among other effects8. While thymectomy is rare, we demonstrate that the thymic decay is highly individualized even in presumed healthy adults, indicating that thymic function can also be substantially reduced in individuals who did not have their thymus surgically removed. Our work therefore impacts the wider population and aligns with the well-established age-related decline in immune system function6,25,26. This is also consistent with previous work modelling declining T cell output as a major risk factor for age-related increase of cancer development13.
Lifestyle and metabolic health measures, such as smoking, physical activity or HDL levels, showed strong associations with thymic health. Likewise, in longitudinal blood-based evaluations, we found individuals with chronic inflammation, a hallmark of immunosenescence and commonly associated with chronic stress, carbohydrate-rich diet and obesity, had lower thymic health.
Among presumed healthy individuals from the FHS, lower thymic health was indeed associated with pro-inflammatory modifications of blood plasma protein levels, consistent with the presence of chronic inflammation. The pro-inflammatory pattern included increased levels of cytokines IL-6, IL-18 and OSM, as well as several CXCL chemokines, all of known relevance in systemic inflammatory diseases such as atherosclerosis, age-associated diseases such as arthritis, and cancer27.
Our findings are further supported by studies in which thymic involution was associated with immunosenescence and inflammation, contributing to illnesses such as metabolic or cardiovascular disease12. Metabolic syndrome affects more than a third of all adults in the USA, with continuously increasing prevalence28. Our results demonstrate significant associations between metabolic and thymic health. These findings are consistent with those of recent studies in which fatty degeneration of the thymus was associated with obesity18,19,20 and smoked pack-years19,20. Together, these findings suggest a profound impact of actionable lifestyle choices on thymic health and may further clarify why healthy behaviour improves well-being and lifespan.
In addition, the global increase in early-onset cancers might be linked to an accelerated rate of thymic decay, potentially driven by factors such as smoking, minimal physical activity and overall unhealthy lifestyles, leading to more inflammation in the body. Indeed, thymic health could refine disease and cancer screening strategies, especially for high-risk individuals.
Here we examined the impact of thymic health on lung cancer incidence separately in current and former smokers. Associations were primarily driven by current smokers, likely reflecting both higher statistical power due to higher event rates and the greater biological relevance of thymic function under ongoing tobacco exposure and thereby continuously increasing neoantigen load, making thymic health particularly relevant for immune surveillance in this setting29.
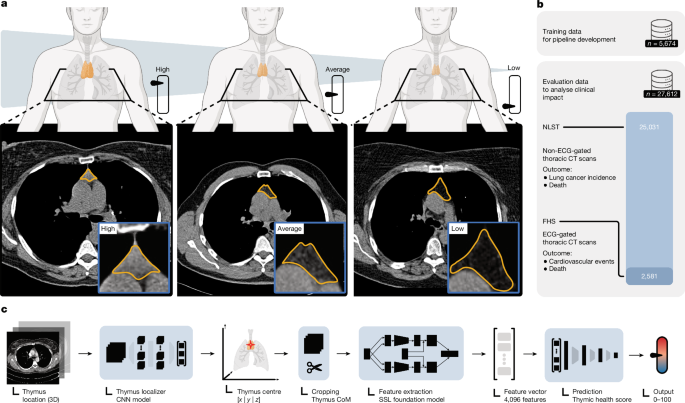
Previous studies have attempted to quantify thymic characteristics using imaging techniques, using conventional approaches such as estimating the proportion of fatty degeneration or measuring attenuation density18,19,20. However, these studies were limited in scope, focusing on the exploration of associations between thymic features and basic clinical and epidemiological measures. Previous studies did not find any associations between thymic imaging characteristics and outcomes such as survival or disease incidence. These previous investigations reported presumed residual thymic tissue in only a minor fraction of the examined population ranging from 41%18 to 26%19,20, that is, the studies estimated a fully fatty degenerated thymus in approximately 60–75% of individuals18. We find these results contradictory to biological associations of sustained T cell output in visually fully fatty degenerated thymic glands18, and we could corroborate that our measure of thymic health had preserved impact on outcomes across individuals who had no appreciable visual thymic tissue according to independent visual scoring, indicating insufficient thymic quantification using a visual scoring system. Our deep learning model found favourable thymic functionality among individuals with average and high thymic health, representing 75% of participants, consistent across both independent study populations and paralleled by substantial health benefits in these participants. Indeed, our findings are supported by Kooshesh et al.8, who found substantial negative health consequences after thymectomy in adults, indicating relevant thymic activity throughout life across the population.
Participants included in this study were prospectively enrolled in the FHS and NLST, covering a wide age range for both sexes. They are, however, predominantly white, and further validation in more ethnically diverse populations is warranted. While we observed an association between thymic health and overall survival in both cohorts, this did not remain significant following thorough multivariate adjustment in the smaller FHS. This further indicates that long-term outcomes may be influenced by dynamic lifestyle choices in this population that benefits from regular medical examinations and follow-up, and supports the notion that active engagement in health-promoting behaviours may potentially attenuate the long-term negative impact of unhealthy characteristics associated with low thymic health30. Taken together, our results provide evidence of multifaceted associations between thymic health and clinically relevant health consequences across clinical, epidemiological and biological characteristics and strongly suggest the critical relevance of the thymus for adult health.
Before the thymic health deep learning model developed here can be applied in a clinical setting, it is essential to prove generalizability. Development and application of the thymic health model were performed in fully independent datasets, with high robustness as demonstrated by test–retest stability. Given substantial differences in acquisition protocols, scanners and population characteristics between the FHS and the NLST, the thymic health analyses were conducted using population-specific thresholding, and no universal cutoffs can be assumed. Future studies with international and external validation are needed to explicitly address batch and scanner variability and enable direct cross-cohort comparisons and generalizable thresholds.
Our results connect thymic health with longevity and lower disease incidence. Our results suggest that inflammation potentially drives an accelerated rate of thymic decay. This finding may provide new opportunities for preventive strategies that aim to reduce thymic decay and potentially even reverse it. These can include the use of anti-inflammatory and anti-obesity drugs and open new avenues of drug development that aim to improve long-term immune health. Furthermore, even practical approaches, such as lifestyle changes including exercise and sleep, as well as healthy food choices and supplement intake, are likely to notably impact thymic health.
While this study investigated the role of thymic health in healthy individuals, the state of the immune system could also have an important role in individuals with disease. This is particularly relevant for treatments that rely on triggering an immune response, such as immunotherapies used to treat patients with cancer. However, it may also be relevant for other diseases: for example, during the recent COVID-19 pandemic, the response to the virus varied significantly between sex and age groups, with older men particularly affected31.
Our analysis reveals previously unrecognized, possible negative consequences of reduced thymic health. While thymic health declines with age, we also find considerable variation in thymic health within age groups, indicating that the rate of decay varies considerably between individuals. When we analysed blood inflammatory proteins, we found an association between increased levels of inflammatory proteins and thymic decay. Although this analysis was limited by a relatively long interval of 10.4 years between the CT scans used for thymic health assessment and blood samples used to assess protein content, these results were supported by an orthogonal assessment of CRP levels, supporting that chronic inflammation may be a likely driver of poor thymic health. It is also likely that a genetic component exists, predisposing certain individuals to increased or decreased rates of thymic decay. A recent large-scale study highlighted substantial individual variability in immune resilience and its link to long-term health outcomes32. Gene expression profiles associated with immune competence and low inflammation were linked to longevity, while pro-inflammatory signatures correlated with poorer outcomes. These findings align with our observations that thymic health varies widely across individuals and is negatively associated with chronic inflammation. Awareness of a genetic predisposition may lead to preventive measures and increased surveillance, and this should be elucidated in future studies that match single nucleotide polymorphisms with rates of thymic decay. Other limitations of this study include the older age and heavy smoking status of individuals in the NLST cohort relative to the FHS cohort, both likely to affect thymic health.
An important implication of our study is that the retrospective observational design does not allow conclusions about causality. It is possible that lower thymic health contributes to adverse outcomes by weakening immune resilience, but it is also possible that pathological processes leading to reduced health status, mortality and disease drive thymic decline. Clarifying the direction and nature of these associations will require future prospective and mechanistic studies and will be essential to determine whether thymic health can serve as a target for prevention or intervention strategies. Recent large-scale initiatives, such as the Advanced Research Projects Agency for Health-funded Thymus Rejuvenation programme, underscore growing interest in developing regenerative approaches to restore thymic function, further highlighting the high clinical relevance of this work33.
In summary, this study underscores the highly personalized nature of thymic health and emphasizes the previously unrecognized possible critical role of maintaining thymic health to preserve an agile, adaptive immune response that will accommodate long-term well-being and longevity. Today, thymic assessments do not have an established clinical standard, and the thymus is not examined in routine clinical care. The extent to which the adult appearance of the thymus is associated with health and whether actionable lifestyle or risk factors may be harnessed to improve thymic health was unknown. By analysing 27,612 individuals, our results provide evidence that thymic health is directly associated with critical outcomes and diseases and may be directly targetable by various approaches, such as smoking reduction and weight loss in overweight and obese individuals.
Our results underline the relevance of the thymus throughout life. Clinical investigations of preventive or regenerative strategies will be vital to help us understand how to harness the thymic potential to improve population health34,35,36,37,38.