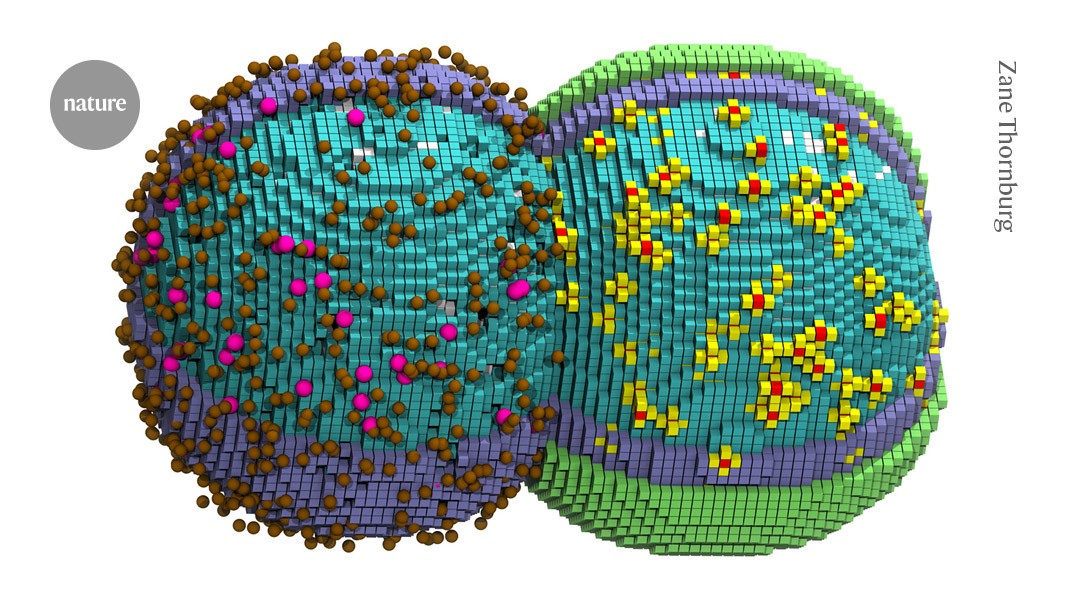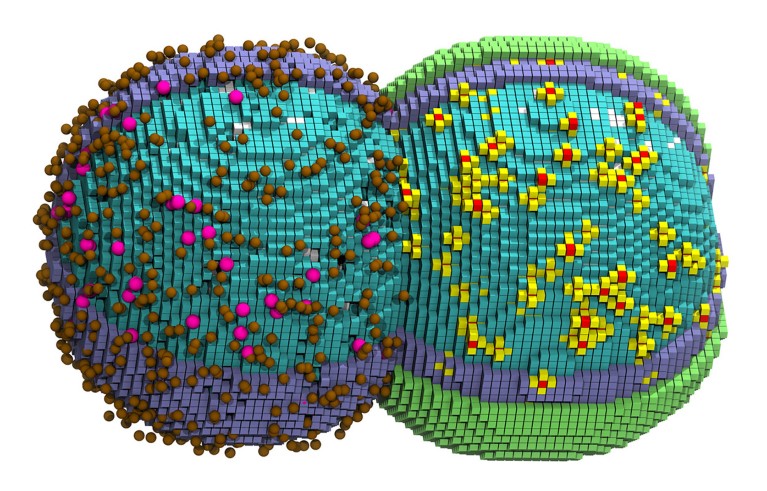
A computer generated illustration of a simulated cell in the early stages of division. Credit: Zane Thornburg
For the first time, researchers have simulated nearly every chemical reaction in a living bacterial cell. The simulation models the virtual cell copying its DNA and dividing in two.
The simulation could help researchers understand how interacting mix of proteins, nucleic acids, fats and other molecules within the wall of a cell gives rise to actual life, says Zane Thornburg, a computational biophysicist at the University of Illinois in Urbana-Champaign who co-led the 9 March study in Cell1.
Virtual life
To model bacterial life, Thornburg turned to one of its simplest examples: a bacterial cell with a ‘minimal’ genome. The organism, named JCVI-Syn3a, was created by whittling the genome of the parasite Mycoplasma mycoides down to just 493 genes, dispensing with more than 400 non-essential genes2.
Thornburg created a three-dimensional simulation that attempted to account for the cell’s DNA, proteins, ribosomes and other molecules of life, as they change over time. Specific molecules, such as a DNA-copying enzyme, obeyed rules based on real-world measurements, and reactions occurred when interacting partners became close in physical space.
Some details were fudged. For example, the functions of several dozen JCVI-Syn3a genes still aren’t known, so Thornburg’s team modelled them as inert spheres. Also, in real cells, multiple ribosomes can make protein using the same mRNA transcript, but Thornburg’s model allowed just one ribosome per transcript.
The team’s goal was to simulate JCVI-Syn3a over the time it takes to copy its DNA and divide into two, known as the cell cycle. Some early attempts failed, Thornburg says, because the genome kept falling apart faster than it could be made, or spewing out of the cell membrane.
After making tweaks to address such issues, they left the model to run over US Thanksgiving holiday in November. “We came back, and suddenly a whole cell cycle had run,” Thornburg says. “All of a sudden, it was just this huge leap.”
The computer simulation recapitulated many details of actual life, including how the shape of the cell swells and elongates as it divides into two. The 105 minutes the virtual cell took to divide was “scarily close” to the time the cell takes to reproduce in real life, Thornburg says. Simulating those 105 minutes, however, took 6 days on a supercomputer, underscoring how computationally intensive such cell models are to run.
Bernhard Palsson, a bioengineer at the University of California, San Diego, says the simulation is an important achievement because of the shear breadth of cellular activity it captures. “Getting all these processes to behave coherently during the cell cycle is a major challenge.”