Mice
All procedures were approved by the New York University Langone Medical Center Institutional Animal Care and Use Committee in compliance with the US National Institutes of Health Guidelines for the Care and Use of Laboratory Animals. Adult mice 12–38 weeks old were used for all of the studies. Mice were housed at 18–23 °C with 40–60% humidity under a 12 h–12 h light–dark cycle (dark cycle, 22:00 to 10:00), with food and water available ad libitum. For C57 and SW mixed-background mice, we bred the F1 mixed-background mice by crossing a male C57BL/6N (Charles River, 027), C57 Esr1-2A-cre (Jackson, 017911), Esr1-2A-Flpo (Jackson, 037009), Npy2r-ires-cre (Jackson, 029285), or Oxt-ires-cre (Jackson, 024234) with a female SW (Taconic, SW-F) mouse. For mixed-background Npy2r-ires-cre/Esr1-2A-Flpo double transgenic female mice, we bred the F3 mixed-background mice by crossing F2 mixed-background homozygous Npy2r-ires-cre female mice with C57 homozygous Esr1-2A-Flpo male mice. For hybrid Oxtrflox/flox female mice (Jackson, 008471), we bred the F2 mixed-background mice by crossing F1 SW and C57 mixed background Oxtrflox/+ mice. These mixed-background breeding strategies enabled cell-type-specific manipulations in aggressive lactating female mice. C57 Esr1-ZsGreen mice were generated and provided by the Y. Xu lab originally and then bred in-house. Stimulus animals were adult (>8 weeks old) and juvenile (15 days old in one case, and 19–28 days old in all other cases) C57BL/6N female and male mice, originally purchased from Charles River and then bred in-house. Stimulus animals were group-housed. After surgery, all test virgin animals were singly housed, and all lactating animals were co-housed with their pups except during the pup-separation procedure. All experiments were performed during the dark cycle (10:00 to 16:00) of the animals.
Viruses
AAV2-CAG-FLEX-GFP (4.00 × 1012 viral genomes (vg) per ml), AAV2-hSyn-GFP (7.30 × 1012 vg per ml), AAV2-Ef1α-DIO-ChR2-eYFP (4.00 × 1012 vg per ml), AAV5-hSyn-Chronos-GFP (2.10 × 1013 vg per ml) and AAV2-Syn-Flex-ChrimsonR-tdTomato (6.00 × 1012 vg per ml) viruses were purchased from the University of North Carolina vector core. AAV1-CMV-Hl-eGFP-cre (2.63 × 1013 vg per ml), AAV2-Ef1α-DIO-hM4Di-mCherry (5.00 × 1012 vg per ml), AAV2-Ef1α-fDIO-mCherry (8.60 × 1012 vg per ml), AAV8-Ef1α-fDIO-GCaMP6s (1.80 × 1013 vg per ml), AAVrg-Ef1α-Flpo (7 × 1012 vg per ml) and AAV8-Ef1α-Coff/Fon-mCherry48 (2.00 × 1013 vg per ml) were purchased from Addgene. AAV2-CAG-FLEX-GCaMP6f-WPRE-SV40 (2.27 × 1013 vg per ml) was custom-made by the University of Pennsylvania vector core facility. AAVDJ-Ef1α-fDIO-hM4Di-mCherry (2.65 × 1013 vg per ml) was custom-made by Vigene Biosciences, and the plasmid was provided by B. Lim at USCD. HSV-hEf1α-LS1L-Flpo (1.00 × 109 vg per ml) was purchased from the Massachusetts General Hospital Neuroscience Center vector core.
Drugs
For chemogenetic inhibition, 5 mg per kg CNO (Sigma-Aldrich, C0832) dissolved in saline was administered i.p. To block OXTRs in the PA and VMHvl, 250 nl per side of 100 µM L-371,257 hydrochloride (Tocris, 2410) in saline was bilaterally injected through the implanted cannulae.
Stereotaxic surgery
Mice (12–20 weeks old) were anaesthetized with isoflurane (1–1.5%) and secured in a stereotaxic apparatus (Kopf Instruments, Model 1900). Viruses and tracers were delivered into the targeted brain regions as previously described16,49. In brief, injections were performed using glass capillaries and a nanoinjector (World Precision Instruments, Nanoliter) at rates of 10 nl min−1 for viruses and 5 nl min−1 for tracers. Only animals with correctly verified injection sites were included in the final analysis (n = 176 out of 194).
For retrograde tracing experiments, 50 nl of Alexa-Fluor-555-conjugated CTB (Thermo Fisher Scientific, C22843) was unilaterally injected into the VMHvl (bregma coordinates: anteroposterior (AP) −1.45 mm; mediolateral (ML) ±0.680 mm; dorsoventral (DV) −5.680 mm). Brains were collected 10 days later. Only mice with confirmed targeting of the VMHvl were included in the final analysis (n = 5 out of 8).
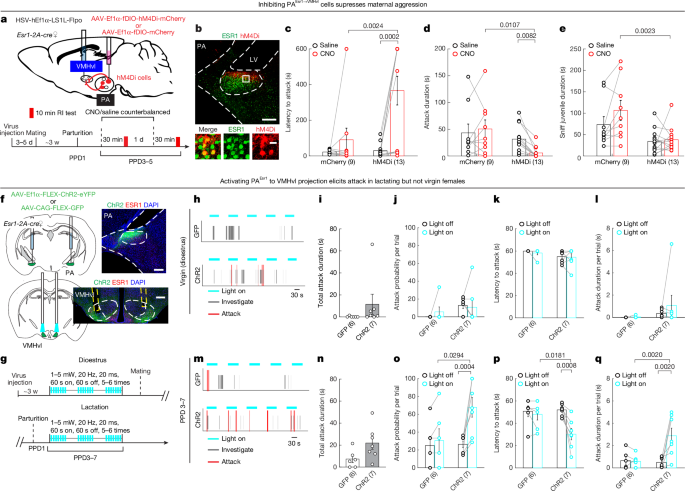
For pharmacogenetic inhibition of PAEsr1 cells, 120 nl of AAV2-hSyn-DIO-hM4Di-mCherry was bilaterally injected into the PA of Esr1-2A-cre mice (bregma coordinates: AP, −2.30 mm; ML, ±2.40 mm; DV, −4.80 mm). Only animals with confirmed bilateral targeting of the PA were included in the final analysis (n = 7 out of 8).
For pharmacogenetic inhibition of PAEsr1→VMHvl cells, 130 nl of HSV-hEf1α-LS1L-Flpo was injected into the VMHvl while 120 nl of AAVDJ-Ef1α-fDIO-hM4Di-mCherry was injected into the PA bilaterally of Esr1-2A-cre mice. Control Esr1-2A-cre animals were bilaterally injected with 130 nl of HSV-hEf1α-LS1L-Flpo into the VMHvl and 100 nl of AAV2-Ef1α-fDIO-mCherry into the PA. Test animals that showed correct bilateral targeting in the VMHvl and virus expression in the PA were included in the final analysis (n = 7 out of 9).
For optogenetic stimulation of the PA-VMHvl projection, 120 nl of AAV2-Ef1α-DIO-ChR2-eYFP was bilaterally injected into the PA of Esr1-2A-cre mice. Control Esr1-2A-cre mice received 100 nl of AAV2-CAG-FLEX-GFP bilaterally into the PA. During the same surgery, a 200-µm optic fibre assembly (Thorlabs, FT200EMT, CFLC230) was implanted 200 µm above each side of the VMHvl. Only animals with confirmed bilateral expression of ChR2–eYFP or GFP in the PA and correct bilateral fibre placements in the VMHvl were included in the final analysis (ChR2–eYFP: n = 14 out of 17; GFP: n = 11 out of 11).
For optogenetic stimulation of VMHvlNpy2r cells, 120 nl of AAV2-Ef1α-DIO-ChR2-eYFP was bilaterally injected into the VMHvl of Npy2r-ires-cre mice. During the same surgery, a 200-µm optic fibre assembly (Thorlabs, FT200EMT, CFLC230) was implanted 200 µm above each side of the VMHvl. Only animals with confirmed bilateral expression of ChR2–eYFP and correct fibre placements were included in the final analysis (ChR2–eYFP: n = 3 out of 3).
For optogenetic stimulation of PVNOxt cells, 120 nl of AAV2-Ef1α-DIO-ChR2-eYFP was bilaterally injected into the PVN of Oxt-ires-cre mice (bregma coordinates: AP, −0.80 mm; ML, ±0.250 mm; DV, −4.50 mm). Control Oxt-ires-cre mice received 100 nl of AAV2-CAG-FLEX-GFP bilaterally into the PVN. During the same surgery, a 200-µm optic fibre assembly (Thorlabs, FT200EMT, CFLC230) was implanted 200 µm above each side of the PVN. Only mice with confirmed bilateral expression of ChR2–eYFP or GFP and correct fibre placements were included in the final analysis (ChR2–eYFP: n = 5 out of 6; GFP: n = 6 out of 6).
For combined pharmacological inhibition of PA OXTR and optogenetic stimulation of PVNOxt cells, 120 nl of AAV2-Ef1α-DIO-ChR2-eYFP was bilaterally injected into the PVN. During the same surgery, a 200-µm optic fibre assembly (Thorlabs, FT200EMT, CFLC230) was implanted 200 µm above the PVN, and cannulae (RWD Life Science, 62102) were implanted 500 µm above the PA bilaterally. Only animals with confirmed bilateral expression of ChR2–eYFP in the PVN and correct placements of both fibre and cannulae were included in the final analysis (n = 7 out of 7).
For fibre photometric recording of PAEsr1→VMHvl cells, 130 nl of HSV-hEf1α-LS1L-Flpo was injected into the VMHvl, and 120 nl of AAV8-Ef1α-fDIO-GCaMP6s was injected into the ipsilateral PA of Esr1-2A-cre mice. A custom-made optic-fibre assembly (Thorlabs, FR400URT, CF440) was then implanted around 150 µm above the dorsal boundary of the PA and secured with dental cement (C&B Metabond, S380). Recordings commenced 3 weeks after virus injection, during both virgin and lactating states. Only animals with confirmed unilateral VMHvl targeting, correct GCaMP6s expression and proper fibre placement in the PA were included in the final analysis (n = 16 out of 19).
For fibre photometric recording of VMHvlNpy2r cells, 120 nl of AAV2-CAG-FLEX-GCaMP6f-WPRE-SV40 was injected into the VMHvl of Npy2r-ires-cre mice. A custom-made optic fibre assembly (Thorlabs, FR400URT, CF440) was then implanted ~150 µm above the dorsal boundary of the VMHvl and secured with dental cement (C&B Metabond, S380). Recordings began 3 weeks after virus injection. Only animals with confirmed GCaMP6f expression in the VMHvl were included in the final analysis (n = 6 out of 7).
For combined fibre photometry recording of VMHvl-projecting PA cells and optogenetic stimulation of PVNOxt cells, 130 nl of AAVrg-Ef1α-Flpo was injected into the VMHvl, 120 nl of AAV8-Ef1α-fDIO-GCaMP6s into the ipsilateral PA and 120 nl of AAV2-Syn-FLEX-ChrimsonR-tdTomato was bilaterally injected into the PVN of Oxt-ires-cre mice. During the same surgery, a 400-µm optic fibre assembly (Thorlabs, FR400URT, CF440) was implanted unilaterally, around 200 µm above both the PVN and PA. Only animals with confirmed bilateral ChrimsonR-tdTomato expression in the PVN, GCaMP6 expression in the PA and correct fibre placements were included in the final analysis (n = 6 out of 8).
For pharmacological inhibition of OXTR, cannulae (RWD Life Science, 62102, 62129) were bilaterally implanted 0.5 mm above the PA or VMHvl of WT mice. Animals included in the final analysis were as follows: PA, 10 out of 11 (C57 × SW F1 hybrid); PA, 3 out of 3 (SW); VMHvl, 7 out of 7 (C57 × SW F1 hybrid).
For conditional Oxtr knockout, 200 nl of AAV1-CMV-HI-eGFP-cre was bilaterally injected into the PA or VMHvl. Control animals received 100 nl of AAV2-hSyn-GFP bilaterally in the same regions. Only animals with confirmed GFP expression in the PA or VMHvl were included in the final analysis (PA with eGFP-cre: n = 8 out of 9; PA with GFP: n = 7 out of 7; VMHvl with eGFP-cre: n = 6 out of 6; VMHvl with GFP: n = 5 out of 5).
For slice recordings of VMHvl-projecting PAEsr1 cells, 30 red retrobeads (Lumafluor, R170) were bilaterally injected into the VMHvl of Esr1-zsGreen mice. Brains were collected for recording 7–10 days later, with females in either the virgin or lactating state. Only animals with confirmed VMHvl targeting were included in the final analysis (n = 4 out of 4).
For slice recordings of VMHvlNpy2r cells, 100 nl of AAV2-CAG-FLEX-GFP was bilaterally injected into the VMHvl of Npy2r-ires-cre mice. Brains were collected for recording 3–4 weeks later, with females in either the virgin or lactating state. Only animals with confirmed VMHvl targeting were included in the final analysis (n = 15 out of 15).
To probe responses of VMHvlNpy2r and VMHvlNpy2r(−)Esr1(+) cells to PA inputs, 200 nl of AAV5-hSyn-Chronos-GFP was bilaterally injected into the PA, and 200 nl of a 1:1 mixture of AAV2-CAG-FLEX-GFP and AAV8-Ef1α-Coff/Fon-mCherry was bilaterally injected into the VMHvl of Npy2r-ires-cre × Esr1-2A-Flpo mice. Brains were collected for recording 3–4 weeks later, with females in either the virgin or lactating state. Only animals with confirmed VMHvl targeting were included in the final analysis (n = 14 out of 14).
To examine the effect of oxytocin on PAEsr1 cells, 120 nl of AAV2-Ef1α-fDIO-mCherry was bilaterally injected into the PA of Esr1-2A-Flpo mice. Brains were collected for recording 3–4 weeks later, with females in either the virgin or lactating state. Only animals with confirmed PA targeting were included in the final analysis (n = 4 out of 4).
Behavioural analysis
Animal behaviours in all experiments were video recorded from both the side and top of the cage using two synchronized cameras (Basler, acA640-100gm) and commercial acquisition software (StreamPix 5, Norpix) in a semi-dark room with infrared illumination at 25 fps. Behavioural annotation and tracking were performed frame by frame using custom MATLAB software (https://pdollar.github.io/toolbox/). For female sexual behaviours, definitions were as follows: lordosis—the female was on the ground, motionless or arched her back while the male was mounting or intromitting. The lordosis quotient was calculated as the ratio of lordosis events to male mounting events. The deep-thrust success rate was calculated as the ratio of mounts followed by intromission to total mount events. For male sexual behaviours: shallow thrust—the male placed his forelegs over the female’s back with hindlimbs on the ground, accompanied by shallow pelvic thrusts; deep thrust—a deep, rhythmic thrust following the shallow thrust. For aggression: attack was defined as a sequence of actions initiated by the resident toward the juvenile intruder, including lunges, bites, tumbling and rapid locomotion between such behaviours. Investigation was defined as nose contact with any part of the stimulus animal. Pup retrieval was defined as lifting a pup with the jaw until it was placed in or near the nest. All mouse behaviours were manually annotated frame by frame by experimenters who were either blinded or not blinded to group assignment. We compared annotations of 15 videos performed both blinded and non-blinded and found a high level of consistency.
Female aggression test
To assess female aggression, we performed the resident–intruder (RI) test during the dark cycle (22:00 to 04:00). A male or female juvenile intruder (19–28 days old), an adult C57BL/6N male or an adult C57BL/6N female was introduced into the female’s home cage (Sealsafe Plus, GM500, 39.1 × 19.9 × 16 cm) for 10 min. No differences were observed in female aggression toward male versus female juvenile intruders.
To evaluate aggression across reproductive states, RI tests were conducted at approximately the same time the day before mating and on postpartum days 1, 3, 5, 7, 14 and 21. When multiple intruders were tested on the same day, the juvenile intruder was always tested first, followed by adult female and adult male intruders, with at least a 2 min interval between tests. During the postpartum period, pups were removed 15 min before the RI test and returned immediately afterward.
To examine the effect of pup separation on maternal aggression, all pups were removed from the home cage of lactating females on postpartum day 3 and cared for by a foster female. Then, 24 h after pup removal, a juvenile mouse was introduced into the test female’s cage for 10 min. After behavioural testing, the pups were returned to the mother’s cage, and maternal aggression towards a juvenile intruder was reassessed 24 h later.
Fibre photometry
Due to loss of Ca2+ signals in the PA after prolonged virus incubation in some animals, GCaMP6 activity of PAEsr1→VMHvl cells was recorded during virgin and lactating states in separate groups of females. For virgin females, recordings began 3–4 weeks after virus injection. Oestrous and dioestrous states were determined through vaginal cytology, and females were recorded during dioestrus and oestrus on separate days. During each recording session, a male or female juvenile (19–28 days old) was introduced into the cage for 10 min. For lactating females, animals were co-housed with males 3–4 days after surgery until pregnancy was visually confirmed. Cages were checked daily, and the day that pups were first observed was designated PPD1. Recordings in the lactating state were conducted on PPD3–PPD5, typically 4–5 weeks after virus injection. During these sessions, pups were temporarily removed, and a male or female juvenile (19–28 days old) was introduced for 10 min.
For head-fixed recording, mice were habituated to head fixation for at least 3 days, 30 min per day. On the recording day, mice were head-fixed immediately after the RI test. An anaesthetized male, female or juvenile C57BL/6N mouse, or a 15 ml conical tube, was placed approximately 2 mm in front of the recording mouse’s nostrils by an experimenter. Each stimulus was presented four times for 5 s, with 20 s intervals between presentations and 1 min between different stimuli.
For fibre photometry recording of VMHvlNpy2r cells, Ca2+ activity was monitored in the same females during both virgin and lactating states. Then, 3 weeks after virus injection, recordings were performed during dioestrus and oestrus. During each session, a male or female juvenile (19–28 days old) was introduced for 10 min. Subsequently, each female was paired with an adult male, and the male was removed once the female became visibly pregnant. Cell responses to a juvenile intruder were then recorded for 10 min on PPD3–PPD5.
The fibre photometry setup was as previously described11,16. In brief, a 390 Hz sinusoidal blue LED light (30 µW; LED, M470F1; LED driver, LEDD1B; Thorlabs) was band-pass filtered (passing band: 472 ± 15 nm; FF02-472/30-25, Semrock) and delivered to the brain to excite GCaMP6. Emission light travelled back through the same optic fibre, was band-pass filtered (passing band: 535 ± 25 nm; FF01-535/505, Semrock), passed through an adjustable zoom lens (SM1NR01, Thorlabs; Edmund Optics, 62-561), detected by a Femtowatt Silicon Photoreceiver (Newport, 2151) and recorded using a real-time processor (RZ5, TDT). The envelope of the 390 Hz signals, reflecting GCaMP6 intensity, was extracted in real time using a custom TDT OpenEx program and low-pass filtered at 5 Hz. For data analysis, the MATLAB function msbackadj with a moving window of 25% of the total recording duration was first applied to obtain the instantaneous baseline signal. The instantaneous ΔF/F was calculated as: ΔF/F = (Fraw − Fbaseline)/Fbaseline. Peri-event time histograms were constructed by aligning ΔF/F signals to behaviour onset. The peak ΔF/F for each behavioural episode was defined as the maximum ΔF/F from 0 to 3 s after behaviour onset minus the average ΔF/F from −10 to −1 s before onset. The introduction peak ΔF/F for each stimulus was calculated as the maximum ΔF/F from 0 to 10 s after stimulus introduction minus the average ΔF/F from −15 to −1 s before onset.
Pharmacogenetic inactivation
To examine the effect of PAEsr1 or PAEsr1→ VMHvl inhibition on female sexual behaviours, the oestrous state of each female was first determined through vaginal cytology on the test day. If a female was in oestrus, a highly sexually experienced male was introduced into her home cage for around 5 min to assess sexual receptivity. The male typically attempted to mount immediately, and, if intromission occurred during this pretest, the female was considered sexually receptive and proceeded to the test.
Females first received an i.p. injection of saline. Then, 30 min later, a different sexually experienced male was introduced into the female’s home cage for 10 min or until the male attempted 10 mounts. Next, 1 h later, the same female was i.p. injected with 5 mg per kg CNO (Sigma-Aldrich, C0832) and, 30 min later, a different sexually experienced male was introduced again to reassess sexual receptivity. Different males were used for each test to prevent the confounding effect of heightened sexual eagerness after previous encounters.
After the mating tests, all females were paired with males until pregnancy was visually confirmed. Maternal care and aggression were then tested during PPD3–PPD5. For the maternal care test, all pups except for one were removed immediately after CNO or saline injection. Then, 30 min later, five pups were introduced far from the nest, and interactions were observed for 10 min. Next, all of the pups were removed, and 2 min later, a male or female juvenile intruder (19–28 days old) was introduced for a 10-minute RI test. Maternal care and aggression tests were repeated the next day using the same female mice 30 min after CNO or saline injection. CNO and saline administration was counterbalanced across animals.
Optogenetic activation
After 3 weeks of viral incubation, on test days, two 200-µm or 400-µm multimode optical fibres (Thorlabs, FT200EMT, FR400URT) were connected to the bilaterally or unilaterally implanted fibre assemblies using matching sleeves (Thorlabs, ADAL1). To activate the PAEsr1→VMHvl projection in virgin females, vaginal smears were examined 30 min before testing to identify dioestrous females. During the test, each female was introduced into the home cage of a sexually experienced male. After introduction, the male typically attempted to mount immediately. Once mounting occurred frequently and regularly, light pulses (5 mW, 20 Hz, 20 ms, 60 s on/60 s off) were delivered through the bilaterally implanted fibres using a laser (Shanghai Dream Laser) controlled by OpenEx (TDT). Light stimulation was repeated 5–6 times per session.
For activating PAEsr1 to VMHvl terminals, a male or female juvenile intruder (15–28 days old) was introduced into the female’s home cage, and light pulses (5 mW or 0 mW, 20 Hz, 20 ms, 60 s on/60 s off, 5–6 times each) were delivered 2 min after intruder introduction. For activating VMHvlNpy2r cells in virgin females, 5 mW or 0 mW, 473 nm, 20 ms, 20 Hz light was delivered for 30 s, each for 4–5 times, 2–3 min after juvenile introduction. The order of sham (0 mW) and real (5 mW) stimulation was counterbalanced across animals. The light intensity at the fibre tip was measured during pulsing using an optical power meter (Thorlabs, PM100D).
To examine the effect of PVNOxt cell activation on the serum oxytocin levels, PA cell activity and maternal aggression, baseline aggression was first assessed 24 h after pup separation. A male or female juvenile intruder (19–28 days old) was introduced into the test female’s home cage for 10 min. Subsequently, blue or yellow light (5 mW, 20 Hz, 20 ms pulses, 20 s on/40 s off) was delivered through the implanted optic fibres for 1 h. Immediately after light stimulation, a different juvenile intruder was introduced into the female mouse’s cage for 10 min, during which maternal behaviours and, in some cases, PA cell responses, were monitored.
Oxtr region-specific knockout
To knock out Oxtr in the PA and VMHvl, we bilaterally injected 120 nl of AAV1 expressing GFP-cre (control: GFP) into the PA or VMHvl of Oxtrflox/flox female mice. We have previously shown that this approach effectively knocks out Oxtr in the VMHvl (ref. 46). Then, 3–5 days after surgery, each female was co-housed with a male until pregnancy was visually confirmed. Between PPD3 and PPD5, maternal aggression was assessed by introducing a juvenile intruder into the female’s home cage for 10 min.
In vivo OXTR antagonist application
To block OXTR in the PA and VMHvl, cannulae (RWD Life Science, 62102, 62129) were bilaterally implanted 0.5 mm above the PA or VMHvl of female mice. Then, 3–5 days after surgery, each female was co-housed with a male until pregnancy was visually confirmed. Between PPD3 and PPD5, 100 nl of 100 µM L-371,257 hydrochloride (Tocris, 2641) was injected into each side of the PA or VMHvl through the cannula using a Hamilton syringe (65457-02) while the test animal was head-fixed on a running wheel. After injection, females were returned to their home cage. Then, 30 min later, a juvenile intruder was introduced, and the female’s behaviour was observed for 10 min. Control animals received saline injections and underwent the same behavioural testing.
Measure oxytocin
Oxytocin levels in mouse tail blood were measured using a previously described method50. Mice were anaesthetized with 1.5% isoflurane and placed in a stereotaxic apparatus. Tail blood was collected and centrifuged at 4 °C, 1,500 rcf for 10 min to isolate serum, which was stored at −80 °C until all samples were collected. Serum oxytocin levels were then measured using an ELISA kit (Arbor Assays, K048-H5) according to the manufacturer’s instructions. Blood samples were collected only once per animal to avoid repeated sampling-induced alterations in maternal behaviour or oxytocin levels.
In vitro electrophysiological recording
To prepare brain slices for patch-clamp recording, mice were anaesthetized with isoflurane, and brains were quickly removed and then immersed in ice-cold cutting solution for 1–2 min (110 mM choline chloride, 25 mM NaHCO3, 2.5 mM KCl, 7 mM MgCl2, 0.5 mM CaCl2, 1.25 mM NaH2PO4, 25 mM glucose, 11.6 mM ascorbic acid and 3.1 mM pyruvic acid). PA or VMHvl coronal sections (275 μm) were cut using the Leica VT1200s vibratome, collected in oxygenated (95% O2 and 5% CO2) and preheated (32–34 °C) ACSF solution (125 mM NaCl, 2.5 mM KCl, 1.25 mM NaH2PO4, 25 mM NaHCO3, 1 mM MgCl2, 2 mM CaCl2 and 11 mM glucose) and incubated for 30 min. The sections were then transferred to room temperature and continuously oxygenated until use.
Current and voltage whole-cell patch-clamp recordings were performed using micropipettes filled with intracellular solution containing 145 mM potassium gluconate, 2 mM MgCl2, 2 mM Na2ATP, 10 mM HEPES, 0.2 mM EGTA (286 mOsm, pH 7.2) or 135 mM CsMeSO3, 10 mM HEPES, 1 mM EGTA, 3.3 mM QX-314 (chloride salt), 4 mM Mg-ATP, 0.3 mM Na-GTP and 8 mM sodium phosphocreatine (pH 7.3 adjusted with CsOH). The signals were recorded using the MultiClamp 700B amplifier (Molecular Devices) and Clampex 11.0 software (Axon Instruments), and digitized at 20 kHz with Digidata 1550B (Axon Instruments). After recording, data were analysed using Clampfit (Molecular Devices) or MATLAB (MathWorks).
To characterize the physiological and synaptic properties of PAEsr1, VMHvlNpy2r and VMHvlNpy2r(−)Esr1(+) cells, we identified ZsGreen-, GFP- and mCherry-positive cells in the PA or VMHvl on slices from Esr1-ZsGreen mice and Npy2r-ires-cre × Esr1-2A-Flpo mice injected with floxed GFP and Coff/Fon mCherry viruses using an Olympus ×40 water-immersion objective coupled with GFP and TXRED filters. To investigate intrinsic excitability, cells were recorded in the current-clamp mode, and the number of action potentials was counted over 500-ms current steps. The current steps consisted of 18 sweeps from −20 pA to 150 pA at 10 pA per step. sEPSCs and sIPSCs were recorded in the voltage-clamp mode. The membrane voltage was held at −70 mV for sEPSC recordings and 0 mV for sIPSC recordings.
To investigate the synaptic connectivity between the PA and VMHvl, Npy2r-ires-cre × Esr1-2A-Flpo female mice were injected with 200 nl of AAV5-hSyn-Chronos-GFP into the bilateral PA and 200 nl of a 1:1 mixture of AAV2-CAG-FLEX-GFP and AAV8-Ef1α-Coff/Fon-mCherry into the bilateral VMHvl. Then, 3 weeks after virus injection and during recording, we voltage-clamp recorded Npy2r-expressing (GFP+) or Npy2r−Esr1+ (mCherry+) VMHvl cells and then delivered 1-ms pulses of full-field illumination using a blue LED light (pE-300white; CoolLED) at 10-s intervals to activate Chronos-expressing PA terminals. oEPSCs and oIPSCs were recorded when holding the membrane potential of recorded neurons at −70 and 0 mV, respectively (corrected for a ~6 mV liquid junction potential). oEPSPs and APs were recorded under the current-clamp mode.
To examine the effect of oxytocin on the cell properties of PAEsr1 cells, we injected 120 nl of AAV2-Ef1α-fDIO-mCherry into the PA of Esr1-2A-Flpo female mice. After 3 weeks of virus incubation, we obtained brain slices and performed current-clamp recordings of PAEsr1 (mCherry+) cells. Cell excitability was measured by injecting 500-ms current steps before and after 250 nM TGOT perfusion. The physiological properties of the cells were characterized −10–0 min before and 10–20 min after TGOT perfusion.
Immunohistochemistry
Immunofluorescence staining was performed as previously described16. In brief, mice were perfused transcardially with 1× PBS followed by 4% paraformaldehyde (PFA) in 0.1 M PBS. Brains were extracted, post-fixed in 4% PFA for 3-4 h at 4 °C followed by 48 h in 20% sucrose, embedded in OCT mounting medium, frozen on dry ice, cut to 50-μm-thick sections using a cryostat (Leica Biosystems) and collected in PBS using a 12-well plate. For antibody staining, brain sections were washed with PBS three times and blocked in PBS-T (0.3% Triton X-100 in 1× PBS) with 10% normal donkey serum for 1 h at room temperature. The sections were then incubated in primary antibody diluted in blocking solution at 4 °C for 24–72 h. We stained for ESR1 (rabbit anti-ESR1; 1:1,000; Invitrogen, PA1-309, YG378288) or FOS (guinea pig anti-FOS; 1:1,000, Synaptic Systems, 226 308). The sections were then washed with PBS-T three times and incubated in secondary antibodies diluted in blocking solution (donkey anti-rabbit or guinea pig, Alexa Fluor 488 or Cy3, 1:1,000; Jackson ImmunoResearch, 711-545-152, 711-165-152 and 706-165-148) with DAPI (1:20,000, Thermo Fisher Scientific, D1306) or NeuroTrace 435/455 Blue Fluorescent Nissl Stain (1:200, Thermo Fisher Scientific, N21479) for 2–3 h at room temperature. The sections were then washed three times with PBS, mounted onto Superfrost slides (Thermo Fisher Scientific, 12-550-15), and coverslipped for imaging on a confocal microscope (Zeiss LSM 700 microscope) and/or a virtual slide scanner (Olympus, VS120).
Fluorescence in situ hybridization
Extracted brains were frozen on dry ice, and 18-µm coronal sections were prepared using a cryostat (Leica Biosystems). RNAscope labelling was performed for all genes using Esr1 (478201-C1), Npy2r (315951-C2) and Oxtr (412171-C3) probes (Advanced Cell Diagnostics) according to the manufacturer’s instructions26.
Dual in situ hybridization and immunohistochemistry
Brain sections were treated with protease-free PretreatPro reagent according to the manufacturer’s protocol. After RNA in situ hybridization, the sections were blocked in PBS-T (0.3% Triton X-100 in 1× PBS) containing 10% normal donkey serum and 1% BSA for 30 min at room temperature. The sections were then incubated with mCherry primary antibody (rabbit anti-DsRed, 1:1,000, Takara, 632496) diluted in PBS-T with 1% BSA for 1 h at room temperature. After two washes in PBS-T, sections were incubated with secondary antibodies (donkey anti-rabbit, Alexa Fluor 594, 1:500; Thermo Fisher Scientific, A21207) diluted in PBS-T with 1% BSA.
Cell counting and axon-terminal quantification
To analyse labelled cells, ×20 confocal or epifluorescence images were acquired, and cells were counted manually using ImageJ.
For DAPI-, Nissl-, CTB- and ESR1-positive cells, ×20 fluorescence confocal images were acquired. For GCaMP6- and mCherry-positive cells in pharmacogenetic and fibre photometry experiments, ×20 fluorescence images were obtained using a virtual slide scanner (Olympus, VS120). For Esr1-, Npy2r– and Oxtr-positive cells, ×20 fluorescence confocal images were acquired. To quantify single-cell expression of Esr1, Npy2r and Oxtr, puncta in ten individual cells from a single section were manually counted by a blinded experimenter using the ImageJ Cell Counter plugin.
To quantify the PAEsr1 axon density in the VMHvl, a 200 × 200 μm2 boxed area containing fibres was selected in each subregion, and the average pixel intensity (Fraw) was measured using ImageJ and Photoshop. A same-size area in the dorsomedial VMH (VMHdm) lacking fibre terminals was used to determine background intensity (Fbackground). Fsignal was then calculated as Fraw − Fbackground. For each animal, Fsignal was normalized to the maximum Fsignal across all analysed regions. The normalized Fsignal values were then used to calculate the average terminal field intensity across animals.
Statistics
Sample sizes were not predetermined statistically but are comparable to previously reported studies16,49. All experiments were conducted using 2 to 11 cohorts of animals. For functional and in vivo recording experiments, histology images were collected from all animals. For slice recording experiments, histology images were only collected from a subset of animals, although the correct virus expression was always confirmed during the recording on the basis of the fluorescence protein expression. Histology images are representative and qualitatively similar to images from other animals. The results were reproducible across cohorts and combined for final analysis. All statistical analyses were performed using MATLAB 2020a (MathWorks) or Prism 10 (GraphPad). All datasets were tested for normality with the Shapiro–Wilk test, except for repeated-measures two-way ANOVA, mixed-effects analysis, Cochran’s Q test, McNemar test, χ2 test and Fisher’s exact test. For datasets that passed the normality test, parametric tests were applied, including unpaired t-tests, paired t-tests, and repeated-measures or ordinary one-way ANOVA with Geisser–Greenhouse correction followed by Tukey’s multiple-comparison post hoc test. For datasets that did not meet normality criteria, nonparametric tests were used, including the Mann–Whitney U-test, Wilcoxon matched-pairs signed-rank test, Friedman test with Dunn’s multiple-comparison post hoc test and Kruskal–Wallis test with Dunn’s multiple-comparison test. Repeated-measures and ordinary two-way ANOVA with Bonferroni’s multiple-comparison post hoc test were used for analyses involving two variables. Mixed-effects models were used to analyse repeated-measures data with missing values. Post hoc Bonferroni’s tests were performed after repeated-measures two-way ANOVA or mixed-effects analysis. Categorical data were analysed using Cochran’s Q test, χ2 test, McNemar test or Fisher’s exact test. When multiple pairwise comparisons were performed, P values were adjusted using the FDR (FDR = 0.05). Details of each statistical test, including exact P values, F values, t values, d.f. and cohort numbers, are provided in Supplementary Table 1. All error bars or error shades represent ±s.e.m. All P and q values equal to or smaller than 0.05 are indicated in the figures.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.