Nowell, P. C. The clonal evolution of tumor cell populations. Science 194, 23–28 (1976).
Hanahan, D. & Weinberg, R. A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011).
Bissell, M. J. & Hines, W. C. Why don’t we get more cancer? A proposed role of the microenvironment in restraining cancer progression. Nat. Med. 17, 320–329 (2011).
Greaves, M. & Maley, C. C. Clonal evolution in cancer. Nature 481, 306–313 (2012). This Article noted the significant parallels between clonal selection processes that lead to malignant tumours and the Darwinian processes that shape clonal diversity in response to genetic or environmental influences.
Boveri, T. Zur Frage Der Entstehung Maligner Tumoren (Gustav Fischer, 1914).
Balmain, A. Peto’s paradox revisited: black box vs mechanistic approaches to understanding the roles of mutations and promoting factors in cancer. Eur. J. Epidemiol. 38, 1251–1258 (2023).
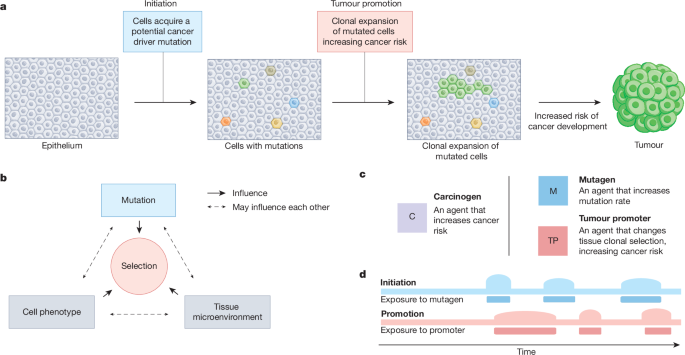
Berenblum, I. & Shubik, P. The role of croton oil applications, associated with a single painting of a carcinogen, in tumour induction of the mouse’s skin. Br. J. Cancer 1, 379–382 (1947). This study demonstrated that a single treatment with an established mutagenic carcinogen was sufficient for cancer initiation, when followed by repeated treatment with the tumour-promoting inflammatory agent croton oil, thus establishing the initiation–promotion model of carcinogenesis.
Yuspa, S. H. The pathogenesis of squamous cell cancer: lessons learned from studies of skin carcinogenesis. Cancer Res. 54, 1178–1189 (1994).
Slaga, T. J. Overview of tumor promotion in animals. Environ. Health Perspect. 50, 3–14 (1983).
Boutwell, R. K., Takigawa, M., Verma, A. K. & Ashendel, C. L. Observations on the mechanism of skin tumor promotion by phorbol esters. Princess Takamatsu Symp. 14, 177–193 (1983).
Parada, L. F., Tabin, C. J., Shih, C. & Weinberg, R. A. Human EJ bladder carcinoma oncogene is homologue of Harvey sarcoma virus ras gene. Nature 297, 474–478 (1982).
Reddy, E. P., Reynolds, R. K., Santos, E. & Barbacid, M. A point mutation is responsible for the acquisition of transforming properties by the T24 human bladder carcinoma oncogene. Nature 300, 149–152 (1982).
ICGC International network of cancer genome projects. Nature 464, 993–998 (2010).
Martínez-Jiménez, F. et al. Pan-cancer whole-genome comparison of primary and metastatic solid tumours. Nature 618, 333–341 (2023).
Campbell, P. J. et al. Pan-cancer analysis of whole genomes. Nature 578, 82–93 (2020).
Stratton, M. R., Campbell, P. J. & Futreal, P. A. The cancer genome. Nature 458, 719–724 (2009).
Sabarinathan, R. et al. The whole-genome panorama of cancer drivers. Preprint at bioRxiv https://doi.org/10.1101/190330 (2017).
Martincorena, I. et al. High burden and pervasive positive selection of somatic mutations in normal human skin. Science 348, 880–886 (2015). This study highlighted the huge mutation burden induced by UV exposure in histologically normal skin, raising the question of why cancers are so rare.
Lawson, A. R. J. et al. Extensive heterogeneity in somatic mutation and selection in the human bladder. Science 370, 75–82 (2020).
Mitchell, E. et al. Clonal dynamics of haematopoiesis across the human lifespan. Nature 606, 343–350 (2022).
Lee-Six, H. et al. The landscape of somatic mutation in normal colorectal epithelial cells. Nature 574, 532–537 (2019).
Yoshida, K. et al. Tobacco smoking and somatic mutations in human bronchial epithelium. Nature 578, 266–272 (2020).
Abby, E. et al. Notch1 mutations drive clonal expansion in normal esophageal epithelium but impair tumor growth. Nat. Genet. 55, 232–245 (2023).
Lawson, A. R. J. et al. Somatic mutation and selection at population scale. Nature 647, 411–420 (2025).
Calvet, F. et al. Sex and smoking bias in the selection of somatic mutations in human bladder. Nature 647, 436–444 (2025). This was one of the first studies showing the statistical power of somatic mutations detected through ultra-deep sequencing to identify associations between the clonal landscape of normal urothelium and the two most salient bladder cancer risk factors.
Riva, L. et al. The mutational signature profile of known and suspected human carcinogens in mice. Nat. Genet. 52, 1189–1197 (2020). In this study, whole genome sequencing of tumours induced in mice by chronic treatment with environmental carcinogens led to the conclusion that most chemical carcinogens have no mutational signature and therefore act as tumour promoters.
Lopez-Bigas, N. & Gonzalez-Perez, A. Are carcinogens direct mutagens? Nat. Genet. 52, 1137–1138 (2020).
Moody, S. et al. Mutational signatures in esophageal squamous cell carcinoma from eight countries with varying incidence. Nat. Genet. 53, 1553–1563 (2021). A search for new mutational signatures indicative of mutagenic exposures as a cause of oesophageal cancers in high-risk countries was proven to be negative, emphasizing the potential role of non-mutagenic carcinogens in human cancer risk.
Senkin, S. et al. Geographic variation of mutagenic exposures in kidney cancer genomes. Nature 629, 910–918 (2024).
Díaz-Gay, M. et al. Geographic and age variations in mutational processes in colorectal cancer. Nature 643, 230–240 (2025).
Dobzhansky, T. Nothing in Biology Makes Sense except in the Light of Evolution. Am. Biol. Teach. 35, 125–129 (1973).
Alexandrov, L. B. et al. The repertoire of mutational signatures in human cancer. Nature 578, 94–101 (2020). This landmark study showed the diversity of mutational signatures in human cancers, induced either by environmental mutagens or endogenous processes that control DNA damage and repair.
Gonzalez-Perez, A., Sabarinathan, R. & Lopez-Bigas, N. Local determinants of the mutational landscape of the human genome. Cell 177, 101–114 (2019).
Supek, F. & Lehner, B. Scales and mechanisms of somatic mutation rate variation across the human genome. DNA Repair 81, 102647 (2019).
Stamatoyannopoulos, J. A. et al. Human mutation rate associated with DNA replication timing. Nat. Genet. 41, 393–395 (2009).
Polak, P. et al. Cell-of-origin chromatin organization shapes the mutational landscape of cancer. Nature 518, 360–364 (2015).
Lawrence, M. S. et al. Mutational heterogeneity in cancer and the search for new cancer-associated genes. Nature 499, 214–218 (2013).
Hill, W. et al. Lung adenocarcinoma promotion by air pollutants. Nature 616, 159–167 (2023). This study presents integrated data from a worldwide, multidisciplinary study, encompassing epidemiology, human genetics and mouse lung tumour models, concluding that air pollution contributes to human lung cancer mainly through its activity as a lung tumour promoter.
Hill, W., Weeden, C. E. & Swanton, C. Tumor promoters and opportunities for molecular cancer prevention. Cancer Discov. 14, 1154–1160 (2024).
Wynder, E. L. & Hoffmann, D. Some laboratory and epidemiological aspects of air pollution carcinogenesis. J. Air Pollut. Control Assoc. 15, 155–159 (1965).
Clemo, G. R. & Miller, E. W. Tumour promotion by the neutral fraction of cigarette smoke. Br. J. Cancer 14, 651–656 (1960).
Van Duuren, B. L., Sivak, A., Segal, A., Orris, L. & Langseth, L. The tumor-promoting agents of tobacco leaf and tobacco smoke condensate. J. Natl. Cancer Inst. 37, 519–526 (1966). References 40–42 drew attention to tumour-promoting effects of tobacco smoking and air pollution, suggesting that some carcinogens can act as both mutagens and tumour promoters.
Alexandrov, L. B. et al. Mutational signatures associated with tobacco smoking in human cancer. Science 354, 618–622 (2016).
Klein, A. M., Brash, D. E., Jones, P. H. & Simons, B. D. Stochastic fate of p53-mutant epidermal progenitor cells is tilted toward proliferation by UV B during preneoplasia. Proc. Natl Acad. Sci. USA 107, 270–275 (2010). This study showed that UV light, a known mutagenic factor, causes clonal expansion by a non-mutagenic process, altering cell fate decisions to promote the outgrowth of p53 mutant cells.
Iversen, O. H. A course of very small doses of DMBA, each of them allegedly with no promoting potency, acts with clear synergistic effect as a strong promoter of DMBA-initiated mouse skin carcinogenesis. A comparison of the tumorigenic and carcinogenic effects of DMBA (7,12-dimethylbenz-alpha-anthracene) and TPA (12-O-tetradecanoyl-phorbol-13-acetate) used as initiators and promoters in classical two-stage experimental protocols. APMIS. Suppl. 41, 1–38 (1994).
Ashman, L. K., Murray, A. W., Cook, M. G. & Kotlarski, I. Two-stage skin carcinogenesis in sensitive and resistant mouse strains. Carcinogenesis 3, 99–102 (1982).
Taylor, M. A. et al. Stem-cell states converge in multistage cutaneous squamous cell carcinoma development. Science 384, eadi7453 (2024).
Reeves, M. Q. & Balmain, A. Mutations, bottlenecks, and clonal sweeps: how environmental carcinogens and genomic changes shape clonal evolution during tumor progression. Cold Spring Harb. Perspect. Med. 14, a041388 (2024).
Huang, S. et al. Lgr6 marks epidermal stem cells with a nerve-dependent role in wound re-epithelialization. Cell Stem Cell 28, 1582–1596.e6 (2021).
Choi, E. et al. Lrig1+ gastric isthmal progenitor cells restore normal gastric lineage cells during damage recovery in adult mouse stomach. Gut 67, 1595–1605 (2018).
Chen, X. et al. IL-17R–EGFR axis links wound healing to tumorigenesis in Lrig1+ stem cells. J. Exp. Med. 216, 195–214 (2019).
Morata, G. & Calleja, M. Cell competition and tumorigenesis in the imaginal discs of Drosophila. Semin. Cancer Biol. 63, 19–26 (2020).
Nicholson, A. M. et al. Fixation and spread of somatic mutations in adult human colonic epithelium. Cell Stem Cell 22, 909–918.e8 (2018).
Yokoyama, A. et al. Age-related remodelling of oesophageal epithelia by mutated cancer drivers. Nature 565, 312–317 (2019).
Fabre, M. A. et al. The longitudinal dynamics and natural history of clonal haematopoiesis. Nature 606, 335–342 (2022).
Mottram, J. C. A developing factor in experimental blastogenesis. J. Pathol. Bacteriol. 56, 181–187 (1944). A landmark study providing evidence that a single treatment with a mutagenic carcinogen was sufficient for cancer initiation, that the initiated cells could lie dormant until exposed to inflammation, and that tissue wounding acts as a strong tumour promoter.
Berenblum, I. The cocarcinogenic action of croton resin. Cancer Res. 1, 44–48 (1941).
Berenblum, I. & Shubik, P. The persistence of latent tumour cells induced in the mouse’s skin by a single application of 9:10-dimethyl-1:2-benzanthracene. Br. J. Cancer 3, 384–386 (1949). This work established and extended the concept of initiated cell dormancy originally suggested by Mottram by showing that initiated cells could survive for most of the mouse lifespan without giving rise to tumours, but could give rise to them after short-term exposure to tumour promoters.
Boutwell, R. K., Verma, A. K., Ashendel, C. L. & Astrup, E. Mouse skin: a useful model system for studying the mechanism of chemical carcinogenesis. Carcinog. Compr. Surv. 7, 1–12 (1982).
Marks, F., Fürstenberger, G. & Müller-Decker, K. in Cancer Prevention. Recent Results in Cancer Research, Vol. 174 (eds Senn, H. J. & Kapp, U.) 37–47 (Springer, 2007).
Hecker, E. Three stage carcinogenesis in mouse skin—recent results and present status of an advanced model system of chemical carcinogenesis. Toxicol. Pathol. 15, 245–258 (1987).
Slaga, T. J. & Butler, A. P. Cellular and biochemical changes during multistage skin tumor promotion. Princess Takamatsu Symp. 14, 291–301 (1983).
Reeves, M. Q., Kandyba, E., Harris, S., Del Rosario, R. & Balmain, A. Multicolour lineage tracing reveals clonal dynamics of squamous carcinoma evolution from initiation to metastasis. Nat. Cell Biol. 20, 699–709 (2018).
McCreery, M. Q. et al. Evolution of metastasis revealed by mutational landscapes of chemically induced skin cancers. Nat. Med. 21, 1514–1520 (2015).
Buqué, A. et al. MPA/DMBA-driven mammary carcinomas. Methods Cell. Biol. 163, 1–19 (2021).
Quintanilla, M., Brown, K., Ramsden, M. & Balmain, A. Carcinogen-specific mutation and amplification of Ha-ras during mouse skin carcinogenesis. Nature 322, 78–80 (1986). This work identified a carcinogen-specific signature mutation in over 90% of mouse skin tumours induced by the mutagen DMBA and promoted with the classical tumour promoter TPA, thus identifying the initiating event in the multistage mouse skin model of carcinogenesis.
Goerttler, K., Loehrke, H., Hesse, B., Milz, A. & Schweizer, J. Diaplacental initiation of NMRI mice with 7, 12-dimethylbenz[a]anthracene during gestation days 6–20 and postnatal treatment of the F1-generation with the phorbol ester 12-O-tetradecanoylphorbol-13-acetate: tumor incidence in organs other than the skin. Carcinogenesis 2, 1087–1094 (1981).
Brown, K. et al. v-ras genes from Harvey and BALB murine sarcoma viruses can act as initiators of two-stage mouse skin carcinogenesis. Cell 46, 447–456 (1986).
Li, Y. R. et al. Long-term latency of highly mutated cells in normal mouse skin is reversed by exposure to tumor promoters and chronic tissue damage. Cancer Discov. 15, 1115–1128 (2025).
Bailleul, B. et al. Skin hyperkeratosis and papilloma formation in transgenic mice expressing a ras oncogene from a suprabasal keratin promoter. Cell 62, 697–708 (1990). References 68–70 provided genetic and functional validation of the model of multistage carcinogenesis first proposed in the 1940s, demonstrating that cells with strong cancer driver mutations can lie dormant in mouse skin without causing tumour formation, but respond rapidly to tumour promoters or wounding to produce cancers within a few weeks.
Doll, R. Strategy for detection of cancer hazards to man. Nature 265, 589–596 (1977).
Doll, R. & Peto, R. The causes of cancer: quantitative estimates of avoidable risks of cancer in the United States today. J. Natl. Cancer Inst. 66, 1191–1308 (1981).
Brennan, P. & Davey-Smith, G. Identifying novel causes of cancers to enhance cancer prevention: new strategies are needed. J. Natl. Cancer Inst. 114, 353–360 (2022).
Nordling, C. O. A new theory on cancer-inducing mechanism. Br. J. Cancer 7, 68–72 (1953).
Armitage, P. & Doll, R. The age distribution of cancer and a multi-stage theory of carcinogenesis. Br. J. Cancer 8, 1–12 (1954). This landmark publication showed the relationship between cancer incidence and age in human populations, identifying a series of 4 to 6 ‘events’ that could explain age-incidence curves.
Armitage, P. & Doll, R. A two-stage theory of carcinogenesis in relation to the age distribution of human cancer. Br. J. Cancer 11, 161–169 (1957).
Vineis, P. & Berwick, M. The population dynamics of cancer: a Darwinian perspective. Int. J. Epidemiol. 35, 1151–1159 (2006).
Peto, R. The need for Ignorance in cancer research. Eur. J. Epidemiol. 38, 1239–1243 (2023).
Moolgavkar, S. H., Dewanji, A. & Venzon, D. J. A stochastic two-stage model for cancer risk assessment. I. The hazard function and the probability of tumor. Risk Anal. 8, 383–392 (1988).
Luebeck, E. G. & Moolgavkar, S. H. Multistage carcinogenesis and the incidence of colorectal cancer. Proc. Natl Acad. Sci. USA 99, 15095–15100 (2002).
Armitage, P. & Doll, R. in Proc. 4th Berkeley Symposium on Mathematical Statistics and Probability 19–38 (Univ. California Press, 1961).
Sporn, M. B., Dunlop, N. M., Newton, D. L. & Smith, J. M. Prevention of chemical carcinogenesis by vitamin A and its synthetic analogs (retinoids). Fed. Proc. 35, 1332–1338 (1976).
Sporn, M. B. & Roberts, A. B. Role of retinoids in differentiation and carcinogenesis. Cancer Res. 43, 3034–3040 (1983).
Ziegler, R. G. et al. Dietary carotene and vitamin A and risk of lung cancer among white men in New Jersey. J. Natl. Cancer Inst. 73, 1429–1435 (1984).
Abar, L. et al. Blood concentrations of carotenoids and retinol and lung cancer risk: an update of the WCRF-AICR systematic review of published prospective studies. Cancer Med. 5, 2069–2083 (2016).
Omenn, G. S. Chemoprevention of lung cancers: lessons from CARET, the beta-carotene and retinol efficacy trial, and prospects for the future. Eur. J. Cancer Prev. 16, 184 (2007). This study presented an important but sobering lesson from prior attempts at cancer prevention in human populations based on inadequate knowledge of mechanisms of cancer promotion.
Omenn, G. S. et al. Effects of a combination of beta carotene and vitamin A on lung cancer and cardiovascular disease. N. Engl. J. Med. 334, 1150–1155 (1996).
Alpha-Tocopherol, Beta Carotene Cancer Prevention Study Group. The effect of vitamin E and beta carotene on the incidence of lung cancer and other cancers in male smokers. N. Engl. J. Med. 330, 1029–1035 (1994).
Lippman, S. M. et al. Effect of selenium and vitamin E on risk of prostate cancer and other cancers: the Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA 301, 39–51 (2009).
Hennings, H., Wenk, M. L. & Donahoe, R. Retinoic acid promotion of papilloma formation in mouse skin. Cancer Lett. 16, 1–5 (1982).
Tennenbaum, T. et al. Topical retinoic acid reduces skin papilloma formation but resistant papillomas are at high risk for malignant conversion. Cancer Res. 58, 1435–1443 (1998).
Cui, W. et al. TGFβ1 inhibits the formation of benign skin tumors, but enhances progression to invasive spindle carcinomas in transgenic mice. Cell 86, 531–542 (1996).
Perillo, B. et al. ROS in cancer therapy: the bright side of the moon. Exp. Mol. Med. 52, 192–203 (2020).
Sayin, V. I. et al. Antioxidants accelerate lung cancer progression in mice. Sci. Transl. Med. 6, 221ra15 (2014).
Swamynathan, M. M. et al. Dietary pro-oxidant therapy by a vitamin K precursor targets PI 3-kinase VPS34 function. Science 386, eadk9167 (2024).
Hecker, E. Cocarcinogenic principles from the seed oil of Croton tiglium and from other Euphorbiaceae. Cancer Res. 28, 2338–2349 (1968).
Van Duuren, B. L., Langseth, L., Sivak, A. & Orris, L. The tumor-enhancing principles of Croton tiglium L. II. A comparative study. Cancer Res. 26, 1729–1733 (1966). References 96 and 97 described the chemical constituents of croton oil, the original tumour promoter used by Mottram, Berenblum and others, leading to the structural identification and ultimate synthesis of the now classical tumour promoter TPA.
Blumberg, P. M. et al. Phorbol ester receptors-insights into the initial events in the mechanism of action of the phorbol esters. Princess Takamatsu Symp. 14, 75–87 (1983).
Castagna, M. et al. Direct activation of calcium-activated, phospholipid-dependent protein kinase by tumor-promoting phorbol esters. J. Biol. Chem. 257, 7847–7851 (1982). Identification of protein kinase C as a direct target of the tumour promoting phorbol ester TPA, enabling many subsequent analyses of the biology of tumour promotion.
Schönwasser, D. C., Marais, R. M., Marshall, C. J. & Parker, P. J. Activation of the mitogen-activated protein kinase/extracellular signal-regulated kinase pathway by conventional, novel, and atypical protein kinase C isotypes. Mol. Cell. Biol. 18, 790–798 (1998).
Nagao, M. et al. Role of protein phosphatases in malignant transformation. Princess Takamatsu Symp. 20, 177–184 (1989).
Suganuma, M. et al. Okadaic acid: an additional non-phorbol-12-tetradecanoate-13-acetate-type tumor promoter. Proc. Natl Acad. Sci. USA 85, 1768–1771 (1988). The laboratory of Sugimura identified okadaic acid as a new class of chemical tumour promoter, ultimately shown to act by inhibition of PP2A, thus showing the diversity of tumour-promoting chemicals.
Thastrup, O., Linnebjerg, H., Bjerrum, P. J., Knudsen, J. B. & Christensen, S. B. The inflammatory and tumor-promoting sesquiterpene lactone, thapsigargin, activates platelets by selective mobilization of calcium as shown by protein phosphorylations. Biochim. Biophys. Acta 927, 65–73 (1987).
Marks, F., Fürstenberger, G. & Kownatzki, E. Prostaglandin E-mediated mitogenic stimulation of mouse epidermis in vivo by divalent cation ionophore A 23187 and by tumor promoter 12-O-tetradecanoylphorbol-13-acetate. Cancer Res. 41, 696–702 (1981).
Dwyer-Nield, L. D. et al. Epistatic interactions govern chemically-induced lung tumor susceptibility and Kras mutation site in murine C57BL/6J-ChrA/J chromosome substitution strains. Int. J. Cancer 126, 125–132 (2010).
Westcott, P. M. K. et al. The mutational landscapes of genetic and chemical models of Kras-driven lung cancer. Nature 517, 489–492 (2015).
Levayer, R. & Moreno, E. Mechanisms of cell competition: themes and variations. J. Cell Biol. 200, 689–698 (2013).
Aydinlik, H., Nguyen, T. D., Moennikes, O., Buchmann, A. & Schwarz, M. Selective pressure during tumor promotion by phenobarbital leads to clonal outgrowth of beta-catenin-mutated mouse liver tumors. Oncogene 20, 7812–7816 (2001).
Witherspoon, J. G. et al. Mutant Nrf2E79Q enhances the promotion and progression of a subset of oncogenic Ras keratinocytes and skin tumors. Redox Biol. 75, 103261 (2024).
Baggiolini, A. et al. Developmental chromatin programs determine oncogenic competence in melanoma. Science 373, eabc1048 (2021).
Lapouge, G. et al. Identifying the cellular origin of squamous skin tumors. Proc. Natl Acad. Sci. USA 108, 7431–7436 (2011).
Brown, K., Strathdee, D., Bryson, S., Lambie, W. & Balmain, A. The malignant capacity of skin tumours induced by expression of a mutant H-ras transgene depends on the cell type targeted. Curr. Biol. 8, 516–524 (1998).
Su, F. et al. RAS mutations in cutaneous squamous-cell carcinomas in patients treated with BRAF inhibitors. N. Engl. J. Med. 366, 207–215 (2012).
Yang, Y. et al. The pioneer factor SOX9 competes for epigenetic factors to switch stem cell fates. Nat. Cell Biol. 25, 1185–1195 (2023).
Sorg, B., Fürstenberger, G., Berry, D. L., Hecker, E. & Marks, F. Preparation of retinoic acid esters of phorbol derivatives. J. Lipid Res. 23, 443–447 (1982).
Akhurst, R. J., Fee, F. & Balmain, A. Localized production of TGF-β mRNA in tumour promoter-stimulated mouse epidermis. Nature 331, 363–365 (1988). This work demonstrated that the tumour promoter TPA induces activation of the TGFβ signalling pathway, subsequently shown to be important in growth control, invasion, immunosuppression, and tumour progression.
Glick, A. B. & Yuspa, S. H. Tissue homeostasis and the control of the neoplastic phenotype in epithelial cancers. Semin. Cancer Biol. 15, 75–83 (2005).
Trosko, J. E. & Ruch, R. J. Gap junctions as targets for cancer chemoprevention and chemotherapy. Curr. Drug Targets 3, 465–482 (2002).
MacCarthy-Morrogh, L. & Martin, P. The hallmarks of cancer are also the hallmarks of wound healing. Sci. Signal. 13, eaay8690 (2020).
Brown, L. M., Malkinson, A. M., Rannels, D. E. & Rannels, S. R. Compensatory lung growth after partial pneumonectomy enhances lung tumorigenesis induced by 3-methylcholanthrene. Cancer Res. 59, 5089–5092 (1999).
Argyris, T. S. Regeneration and the mechanism of epidermal tumor promotion. Crit. Rev. Toxicol. 14, 211–258 (1985).
Pitot, H. C. The natural history of neoplasia. Newer insights into an old problem. Am. J. Pathol. 89, 402–411 (1977).
Mangiante, L. et al. Multiomic analysis of malignant pleural mesothelioma identifies molecular axes and specialized tumor profiles driving intertumor heterogeneity. Nat. Genet. 55, 607–618 (2023).
Rubin, H. Selective clonal expansion and microenvironmental permissiveness in tobacco carcinogenesis. Oncogene 21, 7392–7411 (2002).
Chen, Y. et al. Consumption of hot beverages and foods and the risk of esophageal cancer: a meta-analysis of observational studies. BMC Cancer 15, 449 (2015).
Rapozo, D. C. M. et al. Recurrent acute thermal lesion induces esophageal hyperproliferative premalignant lesions in mice esophagus. Exp. Mol. Pathol. 100, 325–331 (2016).
Berrington de González, A. et al. Advisory Group recommendations on priorities for the IARC Monographs. Lancet Oncol. 25, 546–548 (2024).
Bolton, K. L. et al. Cancer therapy shapes the fitness landscape of clonal hematopoiesis. Nat. Genet. 52, 1219–1226 (2020).
Pich, O., Reyes-Salazar, I., Gonzalez-Perez, A. & Lopez-Bigas, N. Discovering the drivers of clonal hematopoiesis. Nat. Commun. 13, 4267 (2022).
Hagiwara, K. et al. Dynamics of age- versus therapy-related clonal hematopoiesis in long-term survivors of pediatric cancer. Cancer Discov. 13, 844–857 (2023).
Moore, T., Beltran, L., Carbajal, S., Hursting, S. D. & DiGiovanni, J. Energy balance modulates mouse skin tumor promotion through altered IGF-1R and EGFR crosstalk. Cancer Prev. Res. 5, 1236–1246 (2012).
Halliwill, K. D. et al. Panx3 links body mass index and tumorigenesis in a genetically heterogeneous mouse model of carcinogen-induced cancer. Genome Med. 8, 83 (2016).
Kurz, E. et al. Exercise-induced engagement of the IL-15/IL-15Rα axis promotes anti-tumor immunity in pancreatic cancer. Cancer Cell 40, 720–737.e5 (2022).
Ding, R. et al. Size-dependent toxicity of polystyrene microplastics on the gastrointestinal tract: oxidative stress related-DNA damage and potential carcinogenicity. Sci. Total Environ. 912, 169514 (2024).
Zahm, S. et al. Carcinogenicity of perfluorooctanoic acid and perfluorooctanesulfonic acid. Lancet Oncol. 25, 16–17 (2024).
Tommasi, S. et al. Deregulation of biologically significant genes and associated molecular pathways in the oral epithelium of electronic cigarette users. Int. J. Mol. Sci. 20, 738 (2019).
Tang, M.-S. et al. Electronic-cigarette smoke induces lung adenocarcinoma and bladder urothelial hyperplasia in mice. Proc. Natl Acad. Sci. USA 116, 21727–21731 (2019).
Uemura, N. et al. Helicobacter pylori infection and the development of gastric cancer. N. Engl. J. Med. 345, 784–789 (2001).
Ushijima, T. & Hattori, N. Molecular pathways: involvement of Helicobacter pylori-triggered inflammation in the formation of an epigenetic field defect, and its usefulness as cancer risk and exposure markers. Clin. Cancer Res. 18, 923–929 (2012).
Pleguezuelos-Manzano, C. et al. Mutational signature in colorectal cancer caused by genotoxic pks+ E. coli. Nature 580, 269–273 (2020).
O’Dowd, A. GLP-1 receptor agonists: MHRA to study possible side effects after acute pancreatitis cases. Brit. Med. J. 389, r1344 (2025).
Braxton, A. M. et al. 3D genomic mapping reveals multifocality of human pancreatic precancers. Nature 629, 679–687 (2024). This study provides a graphic demonstration of the presence of PANINs harbouring strong oncogenic KRAS mutations in normal human pancreas samples across a wide range of patient ages, supporting the view that promoter-induced activation of inflammation and pancreatitis may be a rate-limiting step in development of pancreatic cancers.
Ames, B. N., Durston, W. E., Yamasaki, E. & Lee, F. D. Carcinogens are mutagens: a simple test system combining liver homogenates for activation and bacteria for detection. Proc. Natl Acad. Sci. USA 70, 2281–2285 (1973).
Kennedy, S. R. et al. Detecting ultralow-frequency mutations by duplex sequencing. Nat. Protoc. 9, 2586–2606 (2014).
Abascal, F. et al. Somatic mutation landscapes at single-molecule resolution. Nature 593, 405–410 (2021).
Valentine, C. C. et al. Direct quantification of in vivo mutagenesis and carcinogenesis using duplex sequencing. Proc. Natl Acad. Sci. USA 117, 33414–33425 (2020).
Yokoyama, A. et al. Somatic mosaicism in the buccal mucosa reflects lifestyle and germline risk factors for esophageal squamous cell carcinoma. Sci. Transl. Med. 17, eadq6740 (2025).
Grundberg, I. et al. In situ mutation detection and visualization of intratumor heterogeneity for cancer research and diagnostics. Oncotarget 4, 2407–2418 (2013).
Torrens, L. et al. The complexity of tobacco smoke-induced mutagenesis in head and neck cancer. Nat. Genet. 57, 884–896 (2025).
Sudlow, C. et al. UK Biobank: an open access resource for identifying the causes of a wide range of complex diseases of middle and old age. PLoS Med. 12, e1001779 (2015).
Sun, B. B. et al. Plasma proteomic associations with genetics and health in the UK Biobank. Nature 622, 329–338 (2023).
Wang, X. et al. A pathology foundation model for cancer diagnosis and prognosis prediction. Nature 634, 970–978 (2024).
Flanagan, D. J. et al. NOTUM from Apc-mutant cells biases clonal competition to initiate cancer. Nature 594, 430–435 (2021).
van Neerven, S. M. et al. Apc-mutant cells act as supercompetitors in intestinal tumour initiation. Nature 594, 436–441 (2021).
Slaga, T. J. Sivak, A. & Boutwell, R. K. (eds) Mechanisms of Tumor Promotion and Cocarcinogenesis (Raven Press, 1978).
Slaga, T. J. (ed.) Tumor Promotion in Internal Organs (CRC Press, 1983).
George, J., Prasad, S., Mahmood, Z. & Shukla, Y. Studies on glyphosate-induced carcinogenicity in mouse skin: a proteomic approach. J. Proteomics 73, 951–964 (2010).
Bauer, A. K., Dwyer-Nield, L. D., Keil, K., Koski, K. & Malkinson, A. M. Butylated hydroxytoluene (BHT) induction of pulmonary inflammation: a role in tumor promotion. Exp. Lung Res. 27, 197–216 (2001).
Guerra, C. et al. Chronic pancreatitis is essential for induction of pancreatic ductal adenocarcinoma by K-Ras oncogenes in adult mice. Cancer Cell 11, 291–302 (2007).
Liang, C. et al. Orthotopic inflammation-related pancreatic carcinogenesis in a wild-type mouse induced by combined application of caerulein and dimethylbenzanthracene. Tumour Biol. 36, 7557–7568 (2015).
Tanaka, T. et al. A novel inflammation-related mouse colon carcinogenesis model induced by azoxymethane and dextran sodium sulfate. Cancer Sci. 94, 965–973 (2003).
Braeuning, A. et al. Tumor promotion and inhibition by phenobarbital in livers of conditional Apc-deficient mice. Arch. Toxicol. 90, 1481–1494 (2016).
Poland, A., Palen, D. & Glover, E. Tumour promotion by TCDD in skin of HRS/J hairless mice. Nature 300, 271–273 (1982).
Luebeck, E. G., Buchmann, A., Stinchcombe, S., Moolgavkar, S. H. & Schwarz, M. Effects of 2,3,7,8-tetrachlorodibenzo-p-dioxin on initiation and promotion of GST-P-positive foci in rat liver: A quantitative analysis of experimental data using a stochastic model. Toxicol. Appl. Pharmacol. 167, 63–73 (2000).
D’Orazio, J., Jarrett, S., Amaro-Ortiz, A. & Scott, T. UV radiation and the skin. Int. J. Mol. Sci. 14, 12222–12248 (2013).
Cohen, A. J. et al. Estimates and 25-year trends of the global burden of disease attributable to ambient air pollution: an analysis of data from the Global Burden of Diseases Study 2015. Lancet 389, 1907–1918 (2017).
Wang, M. et al. Particulate matter air pollution as a cause of lung cancer: epidemiological and experimental evidence. Br. J. Cancer 132, 986–996 (2025).
Das, A. The emerging role of microplastics in systemic toxicity: Involvement of reactive oxygen species (ROS). Sci. Total Environ. 895, 165076 (2023).
Omoike, O. E. et al. Association between per and polyfluoroalkyl substances and markers of inflammation and oxidative stress. Environ. Res. 196, 110361 (2021).
Shane, H. L., Baur, R., Lukomska, E., Weatherly, L. & Anderson, S. E. Immunotoxicity and allergenic potential induced by topical application of perfluorooctanoic acid (PFOA) in a murine model. Food Chem. Toxicol. 136, 111114 (2020).
Seyyedsalehi, M. S. & Boffetta, P. Per- and poly-fluoroalkyl substances (PFAS) exposure and risk of kidney, liver, and testicular cancers. Med. Lav. 114, e2023040 (2023).
Alpert, N., van Gerwen, M. & Taioli, E. Epidemiology of mesothelioma in the 21st century in Europe and the United States, 40 years after restricted/banned asbestos use. Transl. Lung Cancer Res. 9, S28–S38 (2020).
Eriksson, M., Hardell, L., Carlberg, M. & Akerman, M. Pesticide exposure as risk factor for non-Hodgkin lymphoma including histopathological subgroup analysis. Int. J. Cancer 123, 1657–1663 (2008).
Chen, D. et al. Childhood and adolescent residential and farm pesticide exposures and inflammatory bowel disease incidence in a U.S. cohort of women. Sci. Total Environ. 946, 174475 (2024).
Schinasi, L. & Leon, M. E. Non-Hodgkin lymphoma and occupational exposure to agricultural pesticide chemical groups and active ingredients: a systematic review and meta-analysis. Int. J. Environ. Res. Public. Health 11, 4449–4527 (2014).
Kielhorn, J., Melber, C., Wahnschaffe, U., Aitio, A. & Mangelsdorf, I. Vinyl chloride: still a cause for concern. Environ. Health Perspect. 108, 579–588 (2000).
Yang, X., Du, J., Jia, C., Yang, T. & Shao, S. Groundwater pollution risk, health effects and sustainable management of halocarbons in typical industrial parks. Environ. Res. 250, 118422 (2024).
Leja, M. Where are we with gastric cancer screening in Europe in 2024? Gut 73, 2074–2082 (2024).
Cornish, A. J. et al. The genomic landscape of 2,023 colorectal cancers. Nature 633, 127–136 (2024).
Cohen, P. A., Jhingran, A., Oaknin, A. & Denny, L. Cervical cancer. Lancet 393, 169–182 (2019).
Capasso, M., Cossiga, V., Guarino, M., Ranieri, L. & Morisco, F. The role of hepatitis viruses as drivers of hepatocancerogenesis. Cancers 16, 1505 (2024).
Roe, F. J., Salaman, M. H. & Cohen, J. Incomplete carcinogens in cigarette smoke condensate: tumour-promotion by a phenolic fraction. Br. J. Cancer 13, 623–633 (1959).
Glantz, S. A., Nguyen, N. & Oliveira da Silva, A. L. Population-based disease odds for e-cigarettes and dual use versus cigarettes. NEJM Evid. 3, EVIDoa2300229 (2024).
Kinouchi, K. & Sassone-Corsi, P. Metabolic rivalry: circadian homeostasis and tumorigenesis. Nat. Rev. Cancer 20, 645–661 (2020).
Costa, G. [Shift work and breast cancer]. G. Ital. Med. Lav. Ergon. 32, 454–457 (2010).
Rumgay, H. et al. Global burden of cancer in 2020 attributable to alcohol consumption: a population-based study. Lancet Oncol. 22, 1071–1080 (2021).
Bagnardi, V. et al. Light alcohol drinking and cancer: a meta-analysis. Ann. Oncol. 24, 301–308 (2013).
Scoccianti, C. et al. European Code against Cancer 4th Edition: alcohol drinking and cancer. Cancer Epidemiol. 45, 181–188 (2016).