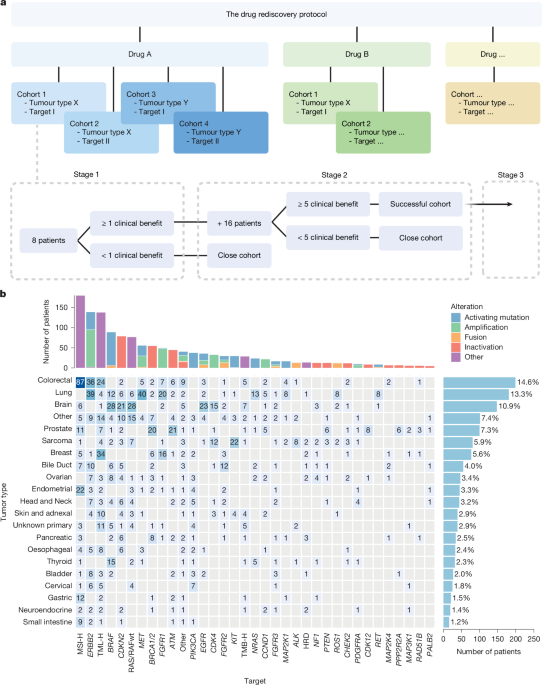
Over the past nine years, DRUP provided opportunities for off-label treatment to more than 1,600 patients with advanced cancers with no remaining standard therapeutic options. This included a substantial proportion (39.1%) of patients with rare cancers—patients who typically face limited histology-based options and who benefited equally from the precision strategy24. Across patients treated in stage 1/2 cohorts, 34.9% experienced clinical benefit, 15.7% achieved an objective response and 7.0% were exceptional responders. The CBR and ORR of DRUP have remained stable for the duration of the study1,24 and are in line with those reported in other prospective precision oncology basket trials, such as NCI-MATCH (Molecular Analysis for Therapy Choice; ORR 10.3%), MyPathway (ORR 23%) and in meta-analysis (ORR 14%)25,26,27.
Off-label drugs are frequently prescribed outside trial settings and without equivalent oversight, which can result in significant harm. Patients, who are often in fragile conditions after exhausting standard treatments, risk serious toxicity—as seen in DRUP, where 28.4% experienced grade 3 or higher TRAEs—and face uneven access15. For healthcare systems, high drug prices, inconsistent reimbursement and the cost of managing toxicities contribute to financial burden15,16,28,29. These concerns argue for stricter regulation of off-label use. Structured initiatives such as DRUP, TAPUR30 (Targeting Agent and Profiling Utilization Registry), CAPTUR31 (Canadian Profiling and Targeted Agent Utilization Trial) and European DRUP-like clinical trials united under the PCM4EU (Personalised Cancer Medicine for all EU Citizens) and PRIME-ROSE (Precision Cancer Medicine Repurposing System Using Pragmatic Clinical Trials) consortia21 help reduce these risks by granting controlled access to treatment while generating evidence. They offer options for patients with rare cancers, monitor outcomes and identify early signals of activity or, importantly, lack thereof. To illustrate, high submission and inclusion rates for DRUP cohorts targeting alterations in the CDK4/6 pathway through single-agent inhibitors reflected strong interest from clinicians, but efficacy proved limited19. Capturing and publishing such negative results is essential to protect future patients from ineffective strategies. Including negative cohorts in clinical guidelines can further help prevent unnecessary costs, false expectations and avoidable toxicity.
Despite the overall modest activity across many cohorts, DRUP also demonstrated that substantial benefit is achievable. Several exceptional responders emerged across different molecular subgroups, illustrating the potential of precision oncology to deliver profound and durable benefit, even in rare, heavily pretreated or difficult-to-treat cancers (for example, eight exceptional responders with BRAF p.V600E-mutated brain tumours). Moreover, one DRUP expansion cohort generated sufficient evidence to achieve full national reimbursement for its off-label indication20, underscoring the idea that structured evaluation can meaningfully expand patient access when efficacy is clear. These positive signals motivated a deeper examination of the variables in our study design that influence patient benefit, including relevance of molecular targets, number of prior treatments, tissue context and cohort expansion.
First, the highest-performing targets in DRUP shared a common denominator: a strong biological rationale and prior clinical evidence of activity. This is reflected in classification systems such as the ESMO Scale for Clinical Actionability of molecular Targets (ESCAT)32 or OncoKB33, which currently rank these alterations among the most actionable. For example, BRAF p.V600E, MSI-H and TMB-H are now marked as ESCAT Tier I-C and OncoKB level 1 tumour-agnostically34. Similarly, MET mutations in NSCLC and BRCA1/2 inactivation in prostate, breast and pancreatic cancers are classified as tier I-B/C and level 1 within their respective tumour types34. These findings, supported by other studies6,35, emphasize the value of evidence-based frameworks for target prioritization to optimize benefit from genome-guided therapy.
Second, improved outcomes were consistently observed in patients with fewer prior treatment lines, a finding echoed in other precision oncology trials22,36. This highlights the potential benefit of administering precision therapies earlier in the disease course, but should be considered in comparison with the benefit of standard treatment strategies available.
Third, we addressed the relevance of tissue context. Following the contrasting outcomes of BRAF inhibition in colorectal cancer and melanoma12,13, DRUP was designed on the premise that tissue context affects treatment response. However, this resulted in many small, tumour-specific, often slow-accruing cohorts, complicating outcome evaluation. As tumour-agnostic inclusion would have simplified the study, we analysed whether tumour type actually influenced outcomes. This was true for 4 out of 17 tested drug–target subgroups (23.5%), which aligns with findings from NCI-MATCH, where tumour-agnostic responses were for instance seen in the BRAF cohort treated with dabrafenib and trametinib, but not for AKT-mutated cancers treated with capivasertib or ipatasertib25. Similarly, real-world data show considerable variation in outcomes for tumour-agnostically approved drugs across tumour types37. These observations suggest that ignoring histology altogether may be premature.
Finally, to facilitate seamless translation of positive findings to clinical practice, DRUP includes a third-stage expansion option with a personalized reimbursement model17. After more than 8 years, 14 cohorts formally met the predefined success criteria, but only one expansion cohort has been completed. A critical appraisal of this limited progression from stage 2 to stage 3 identified two main factors. First, some cohorts met the predefined success criteria, but lacked sufficient clinical impact to justify expansion. In hindsight, the stage 2 success threshold in DRUP did not align with the current regulatory standards. The criterion, clinical benefit in 5 out of 24 patients (21.8%), including stable disease for at least 16 weeks, may overestimate efficacy, particularly in indolent tumours. Our post hoc analysis revealed that only half of the patients with stable disease for at least 16 weeks sustained stable disease beyond 24 weeks. Regulatory frameworks such as the PASKWIL-criteria (used by Dutch Health Technology Assessment bodies)38 and the ESMO-MCBS (European Society for Medical Oncology–Magnitude of Clinical Benefit Scale)39 do not consider short-term stable disease a strong efficacy signal. Instead, end-points such as PFS of at least 6 months and durable, high ORR better reflect clinical relevance and reimbursement criteria. To avoid extending cohorts that are unlikely to impact clinical practice and to ensure promising findings receive adequate follow-up, future precision oncology trials may align primary end-points more closely with regulatory standards.
A second barrier to expanding stage 2 cohorts is patent life. Often, by the time a cohort concluded, the investigated drug faced patent expiry or market competition, reducing pharmaceutical interest in further trials, even under personalized reimbursement models. As a result, patients who could benefit from these effective off-label therapies are left without access. Besides the aforementioned tumour-specific design, other factors that slowed trial progress included limited adoption of molecular diagnostics in clinical practice and the rarity of certain indications. Unlike NCI-MATCH25, DRUP did not provide molecular testing. As access to comprehensive molecular diagnostics remains uneven, many eligible patients were most likely missed40. Nonetheless, even with full identification, some indications were simply too rare to complete cohorts in a country the size of the Netherlands. Faster progress in such settings requires stronger international collaboration. Since its launch in 2016, DRUP has served as a blueprint for national precision oncology trials across Europe, including in Denmark (ProTarget), Sweden (MEGALiT (Molecularly Guided Anti-Cancer Drug Off-Label Trial)), Norway (IMPRESS-Norway) and Finland (FINPROVE)21. The PCM4EU and PRIME-ROSE projects, supported by European grants, now promote shared protocols, aligned end-points and data sharing, helping to accelerate enrollment and validate promising findings within the commercial lifespan of drugs21.
Several limitations of our study should be considered when interpreting these findings. First, many of the drugs evaluated in DRUP were developed before the recent wave of next-generation targeted therapies and immunotherapies, and therefore may not fully reflect the potential of contemporary precision oncology. Similarly, several molecular alterations included in the study are now recognized as weak or non-actionable biomarkers, which helps explain the modest activity observed in some cohorts. Second, although all patients were required to have exhausted standard-of-care treatment options, the level of prior treatment remained heterogeneous. Many patients had received multiple additional lines of therapy, including experimental agents from other trials. This real-world variability reflects the context in which off-label use is typically considered, but may have reduced our ability to detect clinical activity in some settings. As the field evolves, improved biological understanding, more selective and potent drugs, and more sophisticated clinical decision-support tools may largely enhance patient selection and treatment benefit. Our results should therefore not be interpreted as evidence that off-label precision medicines are inherently ineffective, particularly given the presence of multiple exceptional responders across molecularly defined subgroups. Rather, our experiences from 2016 to 2024 strongly argue for confining off-label use to structured, data-generating frameworks until new advancements prove otherwise to allow for a better informed and safer choice of treatment.
In conclusion, oncologists often treat patients with off-label anticancer treatment on the basis of a scientific rationale and hope for a positive outcome. However, in the absence of a framework to systematically capture outcomes, this practice carries significant risks of toxicity, costs and inequitable access. Our analysis of more than 1,600 patients treated in DRUP highlights both the challenges and the opportunities for off-label use. Although overall activity across all indications was limited, we observed meaningful clinical benefit in defined subgroups and several exceptional responders. These positive signals underscore that effective off-label use is possible, but that it requires robust evidence, careful biomarker selection and prospective evaluation to ensure stepwise drug evaluation towards potential label expansion. Although better drugs and improved decision-support systems may widen the therapeutic window, our experience indicates that for now, off-label precision medicines should only be used within clinical trial settings that ensure robust data collection and systematic outcome monitoring.