Experimental procedures
We did not use statistical methods to predetermine sample size. We also did not randomize experiments. During experiments and outcome assessments, investigators were not blinded to allocation.
Mice
We purchased wild-type 129SV mice from Taconic Biosciences. We have previously described the generation and characterization of 129SV mice harbouring the Cer/Sis deletion22. Mouse work was performed in compliance with the relevant ethical regulations established by the Institutional Animal Care and Use Committee of Boston Children’s Hospital. The mouse work was performed under protocols approved by the Boston Children’s Hospital Institutional Animal Care and Use Committee. Mice were maintained on a 14-h light/10-h dark schedule in a temperature (22 ± 3 °C) and humidity (35% to roughly 70% ± 5%) controlled environment, with food and water provided ad libitum. Both male and female 4–8-week-old mice were used for experiments. Mouse experiments are not randomized and not blinded to investigators.
Generation of chimeras from splenic B cell-derived iPS cells
Our laboratory previously generated multiple iPS cell lines reprogramed from splenic B cells, which could contribute to high-grade chimerism in mice30. By screening these iPS cell lines using HTGTS-V(D)J-seq assays with baits for each of the functional mouse Jκ coding ends, we determined their immunoglobulin light chain rearrangements. We selected an iPS cell line harbouring, respectively, a productive Vκ6-23/Jκ5 rearrangement on one allele and a non-productive Vκ4-57/Jκ2 rearrangement that left Jκ4 and Jκ5 intact for potential Igk secondary recombination on the other allele. We denoted this line as ‘parental iPS cell’. The sequences of the Vκ6-23/Jκ5 and Vκ4-57/Jκ2 rearrangements were presented (Supplementary Table 2). We further used HTGTS-V(D)J-seq assay with a Jκ1 RSS-end bait to determine the retained primary Vκ19-93/Jκ1 RSS junction in the Vκ4-57/Jκ2 allele of the parental iPS cell (Supplementary Table 2). To enhance Igk secondary rearrangements, we further mutated the productive Vκ6-23/Jκ5 rearrangements by CRISPR–Cas9 editing in the parental iPS cell line using single-guide RNA 1 (sgRNA1) targeting the Vκ6-23/Jκ5 segment (edited iPS cell, Supplementary Table 3). The parental and edited iPS cell lines, which tested negative for mycoplasma, were injected, respectively, into RAG-deficient blastocysts to generate chimeric mice by means of RAG2-deficient blastocyst complementation as described in ref. 31.
Purification of BM precursor B cells and immature B cells
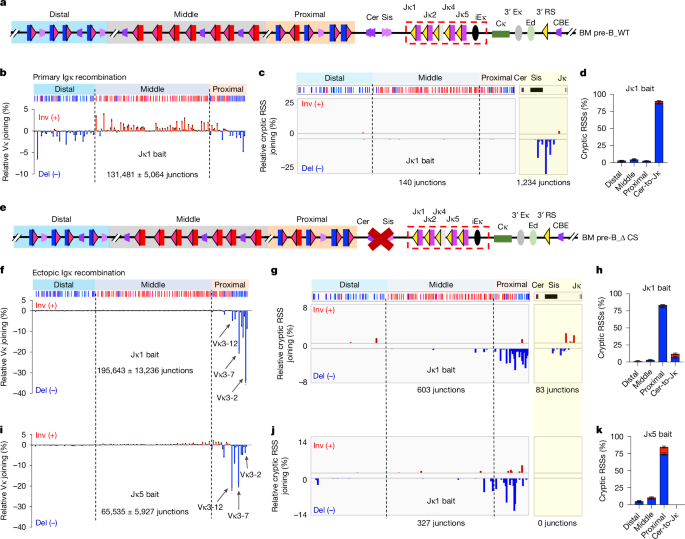
BM pre-B cells from wild-type or Cer/Sis-deleted 129SV mice were isolated as previously described in ref. 20. Briefly, single cell suspensions were derived from BM of 4–8-week-old wild-type or Cer/Sis-deleted 129SV mice. Then, the suspended cells were staining with anti-B220-APC (eBioscience, no. 17-0452-83, 1:1,000 dilution), anti-CD43-PE (BD Biosciences, 553271, 1:400 dilution) and anti-IgM-FITC (eBioscience, no. 11-5790-81, 1:500 dilution). Finally, B220+CD43lowIgM+ population was used to determine BM precursor B cells by means of fluorescence-activated cell sorting. As our iPS cell lines contain mixed genetic background of C57BL/6 and 129SV, we used a modified method for our iPS cell-derived chimeras that distinguishes BM progenitor B cells and precursor B cells in C57BL/6 mice19. Briefly, single cell suspensions were derived from BM of 4–8-week-old parental and edited chimeras. Then, the suspended cells were staining with anti-B220-BV711 (BioLegend, 103255, 1:300 dilution), anti-CD25-PE (BD Pharmingen, 561065, 1:300 dilution), anti-IgG1-FITC (BD Biosciences, 553443, 1:500 dilution), anti-IgM-APC (Invitrogen, 17-5790-82, 1:500 dilution), anti-CD19-BV421 (BD Biosciences, 562701, 1:300 dilution) and anti-c-Kit-PE/Cy7 (eBioscience, 25-1171-81, 1:300 dilution). Finally, B220+CD19+IgM−IgG1−CD25+ KIT− and B220+ CD19+ IgM− IgG1+ populations were used to determine BM precursor B cells and immature B cells, respectively, through fluorescence-activated cell sorting. For DNA extraction and HTGTS-V(D)J-seq assays, we could not get sufficient numbers of BM precursor B cells from the parental chimeras, due to the pre-existing functional Vκ6-23/Jκ5 rearrangement pushing cells through this developmental stage. Therefore, we used BM immature B cells from both the parental and edited chimeras for DNA extraction and HTGTS-V(D)J-seq assays using a Jκ4 coding bait.
Generation of Jκ1 pre-rearranged v-Abl cell lines and derivatives
To generate various Jκ1-pre-rearranged v-Abl cell lines, the previously established Rag2−/−; Eμ-Bcl2+ v-Abl cell line with single Igk allele was used as the initial v-Abl cell line3. We further mutated the downstream 3′ RS by CRISPR–Cas9 editing to minimize potential extra-chromosomal rearrangement events. RAG2 open reading frame-containing plasmids were transiently transfected into v-Abl cell lines by nucleofection. Subsequently STI-571 (Selleck, S2475) was added to the culture medium for 1 day to induce G1-arrest and transient RAG2 expression in the v-Abl cell line. STI-571 was removed by changing culture medium and the v-Abl cells were cultured for 2 more days before plating single clones in the 96-well plates at a density of roughly 0.3 multiplicity of infection. After culturing for 7 more days, half of cells from each single clone were used for PCR screening that used specific primers for, respectively, germline Jκ1, Jκ2, Jκ4 and Jκ5. Finally, pre-rearranged Vκ1-117/Jκ1, Vκ5-37/Jκ1 and Vκ3-12/Jκ1 v-Abl cell lines in which, respectively, Jκ1 was joined to the distal deletional Vκ1-117, the middle inversional Vκ5-37 and the proximal deletional Vκ3-12, were selected and used for subsequent experiments. The sequences of retained Vκ1-117/Jκ1, Vκ5-37/Jκ1 and Vκ3-12/Jκ1 coding junctions in these lines were presented (Supplementary Table 2).
To generate various derivatives of Jκ1-pre-rearranged v-Abl cell lines, plasmids that expressing Cas9 and sgRNAs were transfected into the v-Abl cells by nucleofection to introduce targeted genetic modifications as previously described3. Specifically, sgRNA2 and sgRNA3 that, respectively, target upstream and downstream of Vκ18-36 promoter were transfected into the pre-rearranged Vκ5-37/Jκ1 v-Abl cell line to generate its Vκ18-36 promoter-deleted derivative; sgRNA4 and sgRNA5 that, respectively, target upstream of Vκ9-129 and downstream of Vκ1-118 were transfected into the pre-rearranged Vκ1-117/Jκ1 v-Abl cell line to generate a Vκs-inverted derivative; sgRNA6 and sgRNA7 that, respectively, target upstream of Jκ3 and downstream of Jκ5 were transfected into the pre-rearranged Vκ3-12/Jκ1 v-Abl cell line to delete the DNA segment from upstream of Jκ3 through downstream of Jκ5; sgRNA8 and sgRNA9 that, respectively, target upstream and downstream of Jκ2 were transfected into the pre-rearranged Vκ3-12/Jκ1 v-Abl cell line to generate its Jκ2-inverted derivative. All candidate clones with desired genetic modifications were confirmed by Sanger sequencing and presented in Supplementary Table 2. Sequences of all sgRNAs mentioned in this section are listed in Supplementary Table 3. None of the v-Abl cell lines was tested for mycoplasma contamination.
Generation of Cer/Sis-deleted v-Abl cell lines
To generate Cer/Sis-deleted v-Abl cell lines, plasmids that expressing Cas9 and sgRNAs were transfected into the Rag2−/−; Eμ-Bcl2+ v-Abl cell line by nucleofection to introduce targeted genetic modification as previously described in refs. 3,18. Specifically, sgRNA12 and sgRNA13 that, respectively, target upstream of Cer element and downstream of Sis element were transfected into the Rag2−/−;Eμ-Bcl2+ v-Abl cell line to delete Cer/Sis elements. The junctional sequence confirming the Cer/Sis-deletion in this line is presented in Supplementary Table 2. Sequences of all sgRNAs mentioned in this section are listed in Supplementary Table 3.
RSS replacement experiments
RSS replacement experiments were performed using plasmids expressing Cas9 and sgRNA, and short single-stranded DNA oligonucleotides (ssODN) as donor templates for homology-directed DNA repair as previously described3. Briefly, plasmids expressing Cas9 and sgRNA, and ssODN were cotransfected into the Jκ1-pre-rearranged v-Abl cell lines. Specifically, sgRNA10 and ssODN1 were used to replace the Vκ18-36 12RSS with a Vκ11-125 12RSS in the pre-rearranged Vκ5-37/Jκ1 v-Abl cell line; sgRNA11 and ssODN2–6, respectively, were used to replace the Vκ6-13 12RSS with the 12RSSs from Vκ11-125, DFL16.1-upstream, DQ52-upstream, Vκ12-44 and Vκ19-93 in the pre-rearranged Vκ3-12/Jκ1 v-Abl cell line. All candidate clones with desired genetic modifications were confirmed by Sanger sequencing (Supplementary Table 2). Sequences of all sgRNAs and ssODNs mentioned in this section are listed in Supplementary Table 3.
ATAC-seq and data analyses
ATAC-seq libraries were prepared using the Zymo-Seq ATAC Library Kit (D5458) according to the manual protocol. Briefly, 50,000 living cells from G1-arrest wild-type and Cer/Sis-deleted RAG2-deficient v–Abl cells were used for cell lysis and nuclei isolation. The prepared nuclei pellet was incubated with transposition mix containing Tn5 transposase for DNA tagmentation and sequencing adaptor ligation. Finally, ATAC-seq libraries were prepared from adaptor-ligated DNA by PCR amplification and sequenced on Illumina NextSeq2000 using a paired-end 100-bp sequencing kit. ATAC-seq libraries were processed with a previously described pipeline42.
HTGTS-V(D)J-seq and data analyses
HTGTS-V(D)J-seq libraries were prepared as described3. Briefly, 2 μg of DNA from sorted BM pre-B cells from wild-type and Cer/Sis-deleted mice or BM immature B cells of iPS cell-derived chimeras were used for HTGTS-V(D)J-seq library construction using indicated bait primers listed in figures and figure legends. For RAG2-deficient Jκ1-pre-rearranged v-Abl cell lines, RAG2 was reconstituted by means of retroviral infection with the pMSCV-FLAG-RAG2-GFP vector. After adding signal transduction inhibitor to arrest v-Abl cells in the G1 stage for 4 days, v-Abl cells were collected for DNA purification. Then 10 μg of DNA from G1-arrested RAG-complemented v-Abl cells was used for preparing HTGTS-V(D)J-seq libraries using indicated bait primers. The final libraries were sequenced on Illumina NextSeq550 or NextSeq2000 using a paired-end 150-bp sequencing kit. HTGTS-V(D)J-seq libraries were processed with a previously described pipeline3. All HTGTS libraries were normalized to 500,000 total reads for comparison. Sequences of bait primers used are listed in Supplementary Table 3.
We developed a mouse Igk-specific cryptic RSS usage analysis pipeline. First, cryptic RSS junctions were extracted from HTGTS-V(D)J-seq data using a described pipeline that removes annotated and unannotated bona fide RSSs in the Igk locus3. Second, for each junction, we annotated deletion length from the RSS and insertion length excluding P-elements of bona fide non-productive Vκ to Jκ rearrangements (to avoid influences of selection) in mouse BM pre-B cells and v-Abl cell lines. This analysis revealed essentially no bona fide RSS-associated junctions with greater than 5 bp deletions in Jκ coding bait sequences or greater than 7 bp deletions in Vκ coding prey sequences, and only 1–2% with greater than 2 bp insertions between Vκ-to-Jκ junctions. Accordingly, we filtered out cryptic RSS junctions containing deletions greater than 5 bp in bait, greater than 7 bp deletions in prey, or greater than 2 bp junctional insertions. Third, to exclude misaligned reads, we filtered out cryptic RSS junctions containing more than 2 mutations within 20 bp of the cryptic RSS-associated surrogate coding prey sequences. The programs for this mouse Igk-specific cryptic RSS usage analysis along with annotated data on which it is based have been added to the GitHub repository (https://github.com/Yyx2626/HTGTS_related/tree/main/Igk_specific_anno_and_filter). We note that this pipeline should not be used for off-target analysis of cryptic RSS junctions in human pre-B cells because, unlike mouse pre-B cells, human pre-B cells express terminal deoxynucleotidyl transferase and add N-regions22,38.
3C-HTGTS and data analyses
3C-HTGTS libraries using an iEκ bait were prepared from the G1-arrested RAG2-deficient v-Abl cells as described in ref. 3. Final 3C-HTGTS libraries were sequenced on Illumina NextSeq550 or NextSeq2000 using a paired-end 150-bp sequencing kit, and were processed with a previously described pipeline3 (for details, see ‘Code availability’). All 3C-HTGTS libraries were normalized to 65,488 total junctions, which represents the smallest recovered number of junctions among the 3C-HTGTS libraries that were compared. Primer sequences used for generating 3C-HTGTS libraries are listed in Supplementary Table 3.
GRO-seq and data analyses
Global run-on sequencing (GRO-seq) libraries were prepared from the G1-arrested, RAG2-deficient v-Abl cell lines as described in ref. 20, sequenced using Illumina NextSeq550 or NextSeq2000 using paired-end 150-bp sequencing kits and processed with a previously described pipeline in ref. 20.
Quantification, statistics and reproducibility
All HTGTS-V(D)J-seq, 3C-HTGTS, ATAC-seq and GRO-seq experiments have been repeated three times independently with similar results. NextSeq550 control software (v.2.2.0) and NextSeq 1000/2000 control software (v.1.5.0.42699) were used for high-throughput sequencing data collection. Graphs were generated using GraphPad Prism 9, IGV (v.2.22.1) and R version 3.6.1. FlowJo (v.9.3.2) was used for analysing the fluorescence-activated cell sorting data. After normalization, HTGTS-V(D)J-seq, 3C-HTGTS, ATAC-seq and GRO-seq signals of three repeats were merged as average ± s.e.m. of the maximum value. An unpaired, two-sided Welch’s t-test was used to determine the statistical significance of difference between indicated samples, with P values presented in relevant figure legends.
Availability of materials
All plasmids, cell lines and mouse lines used in this study are available from the authors upon request.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.