Mice
Animal procedures were performed in accordance with National Institutes of Health guidelines with the approval of NYU Grossman School of Medicine’s Institutional Animal Care and Use Committee (IACUC). All animals were housed at 22–25 °C with 50–60% humidity. Animals had access to food and water ad libitum and were housed under a 12 h–12 h light–dark cycle. Experiments in Figs. 2 and 3 were on male 12-week-old C57BL/6J mice. For experiments in Fig. 4, the Saab laboratory crossbred mice1 expressing the tamoxifen-sensitive Cre recombinase cre-ERT2 under the control of the mouse Slc1a3 (encoding GLAST) promoter54 with mice carrying floxed Gjb6 (Gjb6fl/fl)55 and floxed Gja1 (Gja1fl/fl)56. Mice hemizygous for Slc1a3:cre-ERT2 were bred to noncarriers to generate Slc1a3:cre-ERT2+/ × Gja1fl/fl × Gjb6fl/fl experimental mice and Gja1fl/fl × Gjb6fl/fl littermate controls for in vivo experiments; in vivo experiments on this genotype were balanced for sex. When primary mouse astrocytes were isolated, Slc1a3:cre-ERT2 was kept homozygous to obtain a culture in which all astrocytes could be induced through 4-hydroxytamoxifen. All mice were on a C57BL/6 background. The primer sequences used for genotyping were as follows: for Gjb6 flox: 5′-TTCCCTATGCTGGTAGAGTGCTTGT-3′ and 5′-GCAGTAACTTATTGAAACCCTTCACCT-3′; for Gja1 flox: 5′-GGGATACAGACCCTTGGACTCC-3′ and 5′-TCACCCCAAGCTGACTCAACCG-3′; and for Slc1a3-creERT2: 5′-GAGGCACTTGGCTAGGCTCTGAGGA-3′, 5′-GAGGAGATCCTGACCGATCAGTTGG-3′ and 5′-GGTGTACGGTCAGTAAATTGGACAT-3′. The Saab laboratory previously analysed Cre reporter function1 by crossing with ROSA26-floxed-STOP-GCaMP6s (Ai96, JAX, 024106). After shipment, mice were rederived by NYU Grossman School of Medicine’s Rodent Genetic Engineering Laboratory.
Tamoxifen treatment
Tamoxifen treatment was performed as previously described11. For 3 consecutive days, both Slc1a3:cre-ERT2+/ × Gja1fl/fl × Gjb6fl/fl experimental and littermate control mice received daily (between 15:00 and 17:00) gavage of a tamoxifen solution (20 mg ml−1 dissolved in corn oil). The solution was prepared 2 h before administration, during which it was shaken at 37 °C in the dark. Any further experimental manipulations were performed at least 7 days after the last dose of tamoxifen.
Primary rat astrocyte cultures
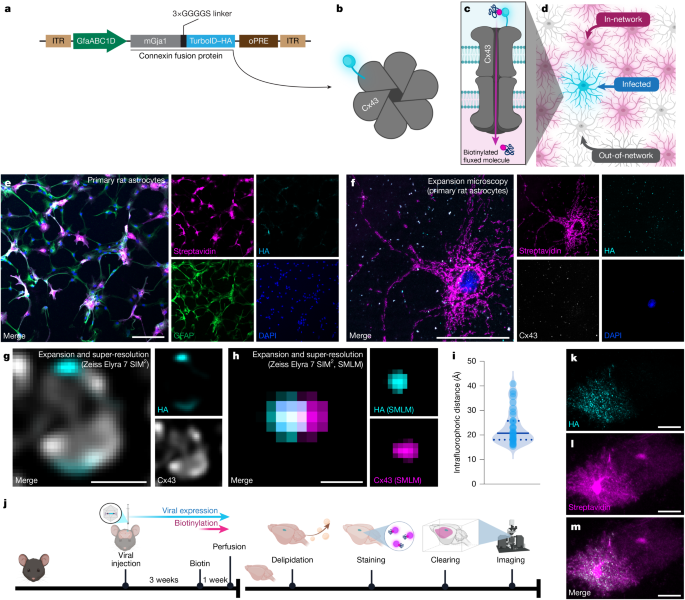
Isolation of primary rat astrocytes by immunopanning was performed as previously described21. In brief, cortices of postnatal day 6/7 Sprague–Dawley rats (Charles River) were blunt dissected, the meninges were removed and the cortices were enzymatically dissociated at 37 °C and 10% CO2 using papain (Worthington Biochemical, LS003126). Tissue was then triturated using a 5 ml serological pipette to generate a single cell suspension, resuspended in Dulbecco’s PBS (VWR, SH30264.FS) with BSA (BSA, Sigma-Aldrich, A4161) and DNase (Worthington Biochemical, LS002007), and filtered using a 20 µm nitex filter. The suspension was negatively panned for non-specific secondary antibody binding, endothelial cells (BSL-1, Vector Labs L-1100), microglia (CD45, BD Pharmingen, 553076) and oligodendrocyte lineage cells (O4 hybridoma, generated in house), and positively panned for astrocytes (ITGB5, Thermo Fisher Scientific, 14-0497-82). Astrocytes were removed from the positive panning plate using TrypLE (Thermo Fisher Scientific, 12-605-010) and plated at 10,000 cells per well in an eight-well chamber slide precoated with poly-l-lysine (Ibidi 80804). Astrocytes were cultured in serum-free medium containing 50% neurobasal, 50% DMEM, 100 U ml−1 penicillin, 100 μg ml−1 streptomycin, 1 mM sodium pyruvate, 292 μg ml−1 l-glutamine, 1× SATO, 5 μg ml−1 N-acetylcysteine and 5 ng ml−1 HBEGF (Sigma-Aldrich, E4643-50UG). After 7 days, AAV was added to the medium change, resulting in an effective titre of 105. Cells were incubated for a further 7 days, with biotin (effective concentration, 250 µM) added to the medium 2 days before fixation.
Expansion microscopy
Expansion microscopy was performed based on published protocols20. Primary astrocytes were quickly washed in PBS, then fixed in 4% PFA in PBS at room temperature for 15 min. After three 5-min washes in PBS, the cells were permeabilized for 15 min in PBS with 0.5% Triton X-100, then blocked for 1 h in 5% BSA and 0.2% Tween-20 in PBS with shaking at room temperature. They were then incubated in primary antibody solution overnight at 4 °C (3% BSA, 0.2% Tween-20 in PBS containing mouse monoclonal anti HA-tag (Cell Signaling, 2367) directly conjugated to ATTO488-NHS (ATTO-TEC AD 488-31), rabbit polyclonal anti-Cx43 (Cell Signaling, 3512) directly conjugated to ATTO565-NHS (ATTO-TEC AD 565-31) and ATTO643-streptavidin (ATTO-TEC, AD 643-61)). Dye conjugation was performed according to the manufacturer’s protocol. Antibodies were added for an effective concentration of 2 µM for anti-Cx43 and 10 µM for anti-HA, and streptavidin (stock concentration 2 μg ml−1) was added at 1:1,000. Cells were then washed three times for 5 min in PBS and incubated for 10 min in 300 nM DAPI in PBS, then imaged on a spinning-disk confocal microscope (CrestOptics X-LIGHT V3 Confocal on Nikon Ti2) with a ×60/1.4 NA oil lens to later establish an expansion coefficient. Then, 250 µl Acryloyl-X, SE (Invitrogen, A20770) was added for overnight incubation at room temperature.
Cells were washed twice for 15 min in PBS, then incubated in 300 µl Gelation Solution per well (for 2 ml: 542 µl 4 M Na acrylate (VWR, S03880), 1 ml PROTOGEL (Thermo Fisher Scientific, 50-899-90119), 200 µl 10× PBS, 198 µl H2O (MQ), 30 µl 10% ammonium persulfate (Thermo Fisher Scientific, 17874), 30 µl tetramethylethylenediamine (TEMED, Thermo Fisher Scientific, 17919)) for 1 h at 37 °C. Then, 300 μl digestion buffer was added to each well and the gels were carefully outlined with a needle to facilitate detachment; once the gels detached, they were each transferred to one well of a 12-well plate for the remainder of the 4 h incubation at 37 °C (volume per gel/well of 12-well plate: 1,550 µl Tris-acetate-EDTA buffer, 100 µl 10% Triton X-100 in PBS, 320 µl 5 M NaCl with 26 µl proteinase K (Thermo Fisher Scientific, EO0491) added immediately before use). The gels were then each transferred to 15 cm Petri dishes filled with room-temperature H2O (MQ) and incubated for 30 min at room temperature. H2O (MQ) was then replaced and the gels were left overnight at room temperature to expand. The gels were then stored at 4 °C until imaging.
Single-molecule imaging and localization
Gels were mounted into 35 mm dishes with #1.5 coverslip bottoms (Ibidi, 81158) with SlowFade Glass soft-set antifade mountant (RI 1.52; Invitrogen, S36917) immediately before imaging. For lattice-SIM imaging, we used the ZEISS Elyra 7 system with an Alpha Plan Apochromat ×63/1.46 NA oil total internal reflection fluorescence microscopy objective. Dual PCO.Edge 4.2 sCMOS cameras collected data (1,280 × 1,280 pixels) with a Dual Camera Beam Splitter (SBS LP 560) used for channel separation on Zen Black 3.0 SR software corresponding to 15 phases of a 27.5 µm grating period for both 488 and 561 channels. SIM2 (structured illumination microscopy) 3D Leap processing was run using the following parameters: input SNR, low; Iterations, 25; regularization weight, 0.01; processing sampling and output sampling, 2. At least 10,000 frames with a 30 ms acquisition time were collected from each sample for each wavelength channel and processed using the Localization Microscopy processing function in 3D. All single- and multi-emitter events were fitted as single emitter events.
Primary mouse astrocyte cultures
Mouse astrocytes were obtained from Slc1a3:cre-ERT2+/+ × Gja1fl/fl × Gjb6fl/fl (note the homozygous Cre, opposed to hemizygous for in vivo experiments where mice could be genotyped). Cortices of postnatal day 3 mice were blunt dissected, meninges were removed, and the cortices were enzymatically dissociated at 37 °C and 10% CO2 by papain (Worthington Biochemical, LS003126). Tissue was then triturated using a 5 ml serological pipette to generate a single-cell suspension, resuspended in Dulbecco’s PBS (VWR, SH30264.FS) with BSA (Sigma-Aldrich, A4161) and DNase (Worthington Biochemical, LS002007), and filtered using a 20 µm nitex filter. The suspension was negatively panned for non-specific secondary antibody binding, microglia (CD45, BD Pharmingen, 553076), oligodendrocyte lineage cells (O4 hybridoma57, generated in house) and L1 (Millipore, rat anti-L1, MAB5272), and positively panned for astrocytes (HepaCAM58; human HepaCAM antibody, MAB4108, R&D Systems). Astrocytes were removed from the positive panning plate using TrypLE (Thermo Fisher Scientific, 12-605-010) and plated at 10,000 cells per well in an eight-well chamber slide pre-coated with poly-l-lysine (Ibidi 80804). Astrocytes were cultured in serum-free medium containing 50% neurobasal, 50% DMEM, 100 U ml−1 penicillin, 100 μg ml−1 streptomycin, 1 mM sodium pyruvate, 292 μg ml−1 l-glutamine, 1× SATO, 5 μg ml−1 N-acetylcysteine and 5 ng ml−1 HBEGF (Sigma-Aldrich, E4643-50UG). On day 6, 4-hydroxytamoxifen (Sigma-Aldrich, SML1666) was added at 1:5,000 (or concentration appropriate for gradient experiments); ethanol:isopropanol (95:5) was added to control wells at the same concentration. On day 7, AAV was added to the medium change, resulting in an effective titre of 1.5 × 104. Cells were incubated for a further 7 days, with biotin (effective concentration 250 µM) added to the medium 2 days before fixation. Cells were washed twice in PBS, then fixed in 4% PFA in PBS for 15 min before immunocytochemistry.
Immunocytochemistry
Fixed cells were washed twice in PBS/azide, then blocked for 2 h in 5% normal donkey serum (NDS) with 0.1% Tween-20 in PBS/azide. They were then incubated overnight at 4 °C with shaking in 3% NDS, 0.1% Triton X-100 in PBS/azide with corresponding primary antibodies (rabbit polyclonal Anti-HA: Cell Signaling, 3724, 1:500; goat polyclonal anti-GFAP, Abcam, ab53554, 1:500). Cells were washed three times for 5 min in PBS/azide, then incubated for 2 h at room temperature with shaking in 1% NDS, 0.1% Triton X-100 in PBS/azide with the corresponding secondary antibodies (donkey anti-goat conjugated to Alexa Fluor 488, Invitrogen, A-110055, 1:500; donkey anti-rabbit conjugated to Alexa Fluor 555, Invitrogen, A-31572, 1:500) and streptavidin conjugated to Alexa Fluor 647 (Thermo Fisher Scientific, S32357, 1:500). Cells were washed three times for 5 min in PBS/azide, then imaged (2,048 × 2,048) on the Zeiss 880 laser-scanning confocal microscope using a ×20/0.8 NA plan apochromat air objective.
Western blotting
Cells were lysed in ice-cold 1× RIPA buffer (diluted in double-distilled H2O from 10×, Cell Signaling, 9806) with 1 mM PMSF (Cell Signaling, 8553) and Halt Protease & Phosphatase Single-Use Inhibitor Cocktail (Thermo Fisher Scientific, 78440). The samples were sonicated and the lysates were separated from insoluble material by centrifugation at 20,000g. Then, 1.5 mm 10% acrylamide gels (Running gel: 4 ml PROTOGEL (Thermo Fisher Scientific, 50-899-90119), 3 ml 1.5 M Tris pH 8.8 + SDS, 5 ml double-distilled H2O, 120 µl 10% ammonium persulfate (Thermo Fisher Scientific, 17874) and 12 µl TEMED (Thermo Fisher Scientific, 17919); stacking gel: 780 µl PROTOGEL (Thermo Fisher Scientific, 50-899-90119), 1.5 ml 0.5 M Tris pH 6.8 + SDS, 3.75 ml double-distilled H2O, 60 µl 10% ammonium persulfate (Thermo Fisher Scientific, 17874) and 18 µl TEMED (Thermo Fisher Scientific, 17919)). The gels were loaded with 10 µg protein per well and run at 120 V; transfers onto PVDF membranes (Immoblion-FL, IPFL00010; pore size 0.45 µm) occurred overnight at 4 °C at 20 V with stirring. After a 1 h block (LI-COR Intercept PBS blocking buffer (LI-COR 927-70001)), the blots were incubated with rabbit anti-Cx43 (1:1,000; Cell Signaling, 3512) and mouse anti-actin (1:1,000; Abcam, ab8226) antibodies in LI-COR Intercept PBS blocking buffer (LI-COR 927-70001) with 0.1% Tween-20 (Sigma-Aldrich, P9416) for 4 h, then washed four times for 5 min with PBST. They were then incubated for 1 h in LI-COR Intercept PBS blocking buffer (LI-COR 927-70001) containing 0.1% Tween-20 and 0.1% SDS with donkey anti-mouse IRDye 680LT (1:20,000, LI-COR, 926-68072) and donkey anti-rabbit IRDye 800CW (1:20,000, LI-COR, 926-32213). After three 5 min PBST washes and one 5 min PBS wash, the blots were imaged on the ChemiDoc MP (Bio-Rad) system using Image Lab v.2.4 (Bio-Rad). The band peaks were quantified in FIJI (ImageJ) using the Gels function.
qPCR
RNA was obtained from cultures using the Qiagen RNeasy Micro Kit (74004) and converted to cDNA using the Qiagen QuantiTect Reverse Transcription Kit (205311) according to the manufacturer’s instructions. qPCR was performed using SYBR Green Universal Master Mix (Thermo Fisher Scientific, 4309155) on the StepOne Plus Real-Time PCR System (Applied Biosystems) using the following primers: Gja1 (F: TCATTGGGGGAAAGGCGTGA, R: CATGTCTGGGCACCTCTCTTTCA), Aldh1l1 (F: TCCCGTCTTTGACCTTGGGT, R: CGCCACCGAGGGAACTTAAA), Slc1a3 (F: CCCCTTACAAAATCAGAAAAGTTGT, R: CCCATCTTGGGCTCTTCTCC), Sox10: (F: GAAGAAGGCTCCCCCATGTC, R: TTGGGTGGCAGGTATTGGTC), Mog (F: GCAGGTCTCTGTAGGCCTTG, R: CCCTCAGGAAGTGAGGATCAAA), Aif1 (F: CTGGGCAAGAGATCTGCCAT, R: ACCAGTTGGCCTCTTGTGTT), Cd14 (F: ACTGAAGCCTTTCTCGGAGC, R: TGAAAGCGCTGGACCAATCT), Tubb3 (F: ACCATGGACAGTGTTCGGTC, R: ACACTCTTTCCGCACGACAT) and Syp (F: CGGCTACCAGCCTGACTATG, R: CTGGGCTTCACTGACCAGAC).
Perfusion and tissue preparation
Mice were always perfused between 14:00 and 17:00 to control for circadian effects. Mice were heavily anaesthetized with an overdose of pentobarbital (Euthasol: 390 mg pentobarbital, 50 mg phenytoin per ml at 2 μl g−1) and transcardially perfused with PBS containing 10 mg l−1 heparin (Sigma-Aldrich, heparin sodium salt from porcine intestinal mucosa, H3393) until the liver and right ventricle were clear of blood. Perfusion solution was then switched to 4% PFA in PBS; all solutions were administered at room temperature at 70% cardiac perfusion pressure. Brains were immediately dissected, taking care to leave the surface fully intact, and then post-fixed in 4% PFA in PBS overnight to stabilize protein tertiary structures. The fixed samples were washed in PBS with 0.01% sodium azide (NaN3) three times for 1 h, then stored in PBS/azide at 4 °C.
When L. esculentum (tomato) lectin was used to image vasculature, 100 µg of TL conjugated to DyLight 649 diluted in 100 µl PBS was injected directly into the left ventricle of the heart before perfusion. After circulating through the beating heart for 1 min, transcardial perfusion resumed as above.
Slice immunohistochemistry
Hemibrains were delipidated (see the ‘Clearing’ section), rehydrated through the same methanol gradient to B1n, then washed three times in PBS/azide. The samples were then sectioned sagittally at 200 µm on a vibrating microtome (Leica, VT1000 S). Floating sections were blocked for 2 h in 5% NDS with 0.1% Tween-20 in PBS/azide, then incubated overnight at 4 °C with shaking in 3% NDS, 0.1% Triton X-100 in PBS/azide with corresponding primary antibodies (rabbit polyclonal anti-Cx43, Cell Signaling, 3512, 1:500; or rabbit polyclonal anti-Cx30, Invitrogen, 71-2200, 1:250; goat polyclonal anti-GFAP, Abcam, ab53554, 1:500). The sections were washed five times for 10 min in PBS/azide, then incubated overnight at 4 °C with shaking in 1% NDS, 0.1% Triton X-100 in PBS/azide with the corresponding secondary antibodies (donkey anti-goat conjugated to Alexa Fluor 488, Invitrogen, A-110055, 1:500; donkey anti-rabbit conjugated to Alexa Fluor 555, Invitrogen, A-31572, 1:500). The sections were washed five times for 10 min in PBS/azide and mounted in ProLong Diamond Antifade Mountant with DAPI (Invitrogen, P36962) to obtain a close refractive index match to the delipidated sections.
Confocal imaging
Sections were imaged on the Zeiss 800 confocal microscope; montages were obtained using a ×20 plan apochromat NA 0.75 air objective at 512 × 512 per image and high-resolution inset images were obtained using a ×63 plan apochromat NA 1.4 oil objective at 2,048 × 2,048 resolution. z stacks were obtained at a step size of 8.2 µm (×20) or 4.4 µm (×63) using Zen Blue 3.2 (Zeiss). All directly compared sections were obtained at the same magnification and were imaged using the same settings. Maximum z projections were rendered using FIJI (ImageJ v.1.54f).
Connexin and vasculature relative localization analysis
Confocal images were analysed in FIJI/ImageJ (v.1.54m). Three confocal micrographs for each of three mice per condition were analysed using ImageJ’s Plot Profile function for three equally spaced line segments perpendicular to imaged vasculature. Line segments were placed so that they began on the edge of a vessel, completely transected it and then passed through the remainder of the micrograph. Profile values for each line were averaged to represent one micrograph. The graph is presented as the distance from the far end of each blood vessel with the average in-vessel portion of all images demarcated.
Intracerebral viral injections
Surgeries were performed under aseptic conditions in accordance with NYU Grossman School of Medicine’s institutional biosafety guidelines. A glass micropipette was pulled to a tip diameter of approximately 20 μm, filled with mineral oil and attached to a Nanoject III microinjector (Drummond Scientific, 3-000-207). The micropipette was then backfilled with 1 μl of adeno-associated Virus (AAV) at a titre of 1011 genomic units per μl. Mice were anaesthetized with 0.7–2.5% isoflurane (adjusted on the basis of scored reflexes and breathing rate during surgery) and placed into a stereotaxic apparatus (Kopf), and a single craniotomy was performed over the experimental brain region (coordinates are provided below); the micropipette was inserted and 200 nl of virus was injected (40 cycles, 5 s delay between cycles, 5 nl injected per cycle and 5 nl s−1). Then, 10 min after injection, the micropipette was withdrawn and the wound was sutured closed. Buprenorphine (0.1 mg per kg) was subcutaneously administered immediately after surgery and twice daily for the next 72 h. We used the following stereotaxic coordinates: motor cortex (anteroposterior (AP), −2.6 mm; mediolateral (ML), 1.3 mm; dorsoventral (DV), −0.8 mm), hippocampus (AP, −2.0 mm; ML, 1.5 mm; DV, −1.5 mm), paraventricular nucleus (AP, 0.12 mm; ML, −0.71 mm; DV, −4.8 mm), prefrontal cortex (AP, 0.35 mm; ML, 1.9 mm; DV, −2.5 mm), barrel cortex (AP, −1.5 mm; ML, 3.5 mm; DV, −0.7 mm). The astrocyte network tracer (AAV5-GfaABC1D-Cx43:TID:HA, Addgene, 252501) was constructed and packaged by VectorBuilder. The AAV9-CaMKIIa-mCherry was obtained from Addgene as a gift from K. Deisseroth (Addgene, 114469-AAV9). All vectors were injected at a titre of 1 × 211.
Biotin administration
Biotin (Sigma-Aldrich,B4639) stocks were diluted to 100 mM in DMSO, aliquoted for single-use and stored frozen at −20 °C. Then, 21 days after intracerebral injections, biotin stock was diluted in distilled water to 160 µM (400 µl of 100 mM stock per 250 ml of distilled water) and provided ad libitum for 7 days, after which the mice were perfused (always between 14:00 and 17:00 to control for circadian effects).
Whisker trimming
Every other day from P28 to P84, mice were lightly anaesthetized with 0.7–1.5% isoflurane (adjusted on the basis of scored reflexes and breathing rate). All whiskers one side of the face were trimmed with scissors. On P56, whisker trimming coincided with intracerebral viral injections (see above). For P77–P84, mice received biotin-supplemented drinking water (see above); perfusion occurred on P84 (see above).
Clearing
The clearing protocol uses similar principles to the iDISCO10 or AdipoClear9 protocol with several adjustments specific to brain tissue and astrocyte morphological maintenance. To delipidate, fixed samples were washed in B1N buffer (H2O, 0.1% Triton X-100, 0.3 M glycine, 0.01% sodium azide, pH 7) for 1 h, followed by 1 h washes each in 20%, 40%, 60% and 80% methanol in B1N. The samples were fully dehydrated in 100% methanol for three 1 h washes, then delipidated in 2:1 dichloromethane (DCM; Sigma-Aldrich, SHBR8133) overnight. Delipidation resumed the next day in three 1 h washes in 100% DCM. The samples were then washed twice in 100% methanol for 1 h. Residual fluorescence was quenched through an overnight incubation in 80% methanol, 5% H2O2 and 15% H2O at 4 °C. The brains were then rehydrated by 1 h incubations in each 60%, 40% and 20% methanol in B1N. The samples were then washed in B1N overnight.
The samples were permeabilized in PTxwH buffer (PBS, 0.1% Triton X-100, 0.05% Tween-20, 2 µg ml−1 heparin, 0.02% sodium azide) with added 45 g l−1 glycine and 50 ml l−1 dimethyl sulfoxide (DMSO), two times for 1 h each. The samples were then washed three times in PTxwH for 1 h.
The samples were incubated in primary antibody (HA-tag, Cell Signaling, 3724, 1:2,000) and conjugated streptavidin (Alexa Fluor 647 streptavidin, Thermo Fisher Scientific, S32357, 1:800) for 1 week at 37 °C with rotation. After primary incubation, the samples were washed in PTxwH with eight solution changes over a period of 3 days. The samples were then incubated in secondary antibody (donkey anti-rabbit conjugated to Alexa Fluor Plus 555, Thermo Fisher Scientific, A32794) and conjugated streptavidin (1:800) for 1 week at 37 °C with rotation. As after secondary incubation, the samples were washed in PTxwH with 8 solution changes over a period of 3 days.
For tissue clearing, the samples were dehydrated in a 20%, 40%, 60%, 80%, 100%, 100%, 100% methanol/H2O series for 45 min each. After dehydration, samples were washed in 2:1 DCM:methanol overnight followed by three 1 h washes in 100% DCM. The samples were cleared overnight in dibenzyl ether (Sigma-Aldrich) and stored at room temperature in the dark in amber glass vials until imaging.
Light-sheet imaging
Whole, cleared brains were imaged on a light sheet microscope (Zeiss Z1) equipped with a ×5 EC plan neofluar NA 0.16 objective lens and ×20 Clr plan neofluar NA 1.0 objective lens. The images were collected with two 1,920 × 1,920 pixel sCMOS cameras and acquired using Zen Blue 3.1 LS (Zeiss). The samples were staged in a custom imaging chamber (Translucence Biosystems) filled with dibenzyl ether and illuminated from both sides by the laser light sheet (5× light sheet thickness, 13.08 µm; step size, 4.5 µm; ×20 light sheet thickness, 4.35 µm; step size, 0.61 µm).
Light-sheet image analysis
For cKO experiments, Imaris (v.9.9) was used to generate surfaces around the positive signal (calculated by thresholding) in the HA and streptavidin channels. The total volume of streptavidin signal divided by the total volume of HA signal was used to generate a relative network volume representing each brain. For whisker-trimming experiments, individual astrocytes were counted in FIJI (ImageJ v.1.54b) after background thresholding. The number of streptavidin-positive astrocytes was divided by the number of HA positive astrocytes to generate a network size value representing each brain.
For the analyses in Fig. 3, image tiles were stitched into full-volume 3D TIFF stacks using Stitchy v.1.15.0 (Translucence Biosystems)—proprietary software that performs automated tile registration by aligning overlapping regions to generate seamless volumetric reconstructions. Stitching was performed on Windows workstations with a sigmoidal blending algorithm applied across overlapping tiles to minimize boundary artifacts. Each reconstructed brain volume averaged approximately 280 GB in size. In some instances, the software reoriented spatial axes as needed to maintain a consistent anatomical orientation across samples.
Segmentation and quantification were performed using Voxels v.1.10.0 (client) and v.1.9.1 (server) (Translucence Biosystems), a cloud-ready image-analysis platform for large-scale processing. Raw fluorescence volumes were converted into 3D voxel-wise probability maps using a random-forest pixel classifier trained on manually annotated crops from seven representative brain volumes to classify voxels as signal of interest or background. Probability maps were refined with threshold- and morphology-based filters to generate binary total-signal masks at full imaging resolution, which were then used to exclude background voxels during downstream regional quantification and atlas registration.
Each sample was registered to the Allen Reference Atlas 2, based on the Allen Mouse Common Coordinate Framework (CCFv3), using the 647 nm fluorescence channel as the reference. The resulting full-resolution total-signal masks were resampled to the atlas voxel resolution for non-linear registration, preserving segmentation fidelity and quantitative accuracy. After alignment, masked signal volumes were partitioned by the midline into left and right hemispheres to assess potential lateralization.
A hierarchical atlas annotation encompassing >500 regions enabled multiscale quantification, allowing integrated intensity values to be aggregated from fine substructures (for example, dentate gyrus) to parent structures (for example, hippocampal formation) and to whole-hemisphere levels. All samples met quality-control criteria for registration accuracy, confirmed by visual inspection of anatomical landmarks relative to the reference atlas and annotation boundaries.
Statistical analysis and reproducibility
Data are presented as mean ± s.d. Mice were assigned at random to groups. Experiments were not performed in a blinded manner. Sample sizes were determined through reference to previous literature. Statistical significance for each experiment was determined as described in the respective figure legend. In the figure legends, n values represent the number of biologically independent animals per group. For data in all figures other than Fig. 3 and Extended Data Fig. 8, statistical analyses were performed in Prism 10 software (GraphPad). For Fig. 3 and Extended Data Fig. 8, regions smaller than 10,000 voxels were excluded owing to potential variance in atlas registration. Student’s t-tests assess regional differences and the Benjamini–Hochberg procedure was used to control the false discovery rate. No datapoints were excluded.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.