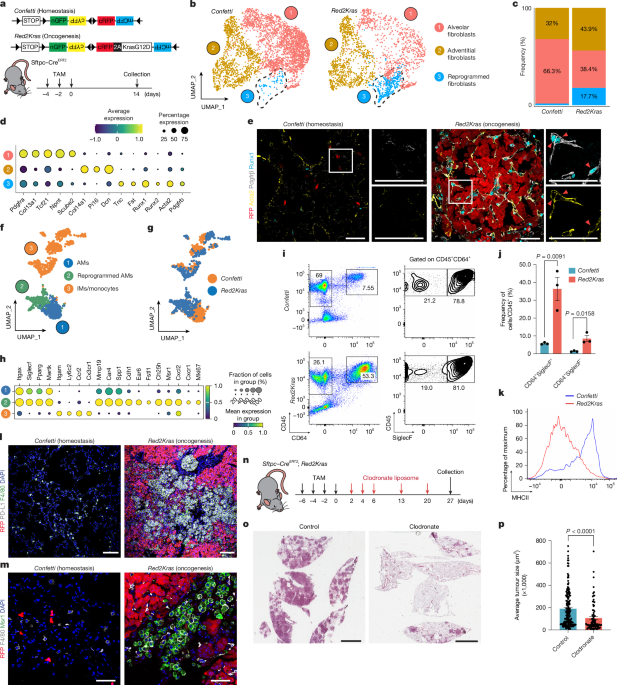
Mice
Sftpc–CreERT2 (028054), R26R–Confetti (013731), Pdgfrα–CreERT2 (032770), R26R–iDTR (007900), NOD/Scid Il2rg null Tg (NSG: 005557) and Ai6/RCL–ZsGreen (007906) animals were obtained from The Jackson Laboratory. AregloxP/loxP animals were kindly provided by M. Clatworthy from the University of Cambridge. Red2Kras mice were generated in-house as previously described22. CCR2–CreERT2 mice were kindly provided by B. Becher (University of Zurich)58. All transgenic mouse strains were maintained on a C57BL or C57BL/6Brd-Tyr 597 c-Brd mixed background. Mouse studies in the UK were approved under UK Home Office Project Licences PC7F8AE82 and PP3176550, and experiments in the US and Korea were approved by the Memorial Sloan Kettering Cancer Center (MSKCC) Institutional Animal Care and Use Committee (protocol no. 24-04-003) and Gwangju Institute of Science and Technology (GIST) Institutional Animal Care and Use Committee (protocol no. GIST-2022-043). All procedures complied with institutional and national guidelines. The mice were housed under specific pathogen-free conditions at the Gurdon Institute (University of Cambridge), MSKCC and GIST on a 12-h light/dark cycle with food and water provided ad libitum.
Both male and female mice aged 6–15 weeks were used. Experiments were randomized where feasible. Blinding was not performed, as treatment effects on tumour volume were readily distinguishable between groups. Humane end points were defined as a single tumour exceeding 2 cm in diameter, a tumour burden exceeding 10% of body mass or multiple tumours with a cumulative volume greater than 3,000 mm3. As this study focused on early tumour development, these limits were not approached or exceeded in any experiment.
Mouse procedures
Tamoxifen administration
Tamoxifen (Sigma; T5648) was dissolved in corn oil (Sigma; C8267) at 20 mg ml−1. Aliquots were heated to 50 °C and vortexed before administration. Animals were weighed and received tamoxifen by oral gavage. They received either a single dose (0.1 mg g−1 body weight) for clonal analysis or two to four doses (0.2 mg g−1 body weight) administered every other day. Tissue collection time points are specified in the relevant figures and detailed in Methods.
MRTX1133 administration
Red2Kras animals received two doses of tamoxifen through oral gavage (0.2 mg g−1 body weight) every other day to induce KrasG12D expression. At 4 weeks post-induction, the mice received freshly prepared MRTX1133 (MedChemExpress; HY-134813) through intraperitoneal injection at 15 mg kg−1 twice daily for 10 days. The stock solution was prepared in DMSO and diluted in 40% polyethylene glycol 300 (PEG300; MedChemExpress; HY-Y0873), 5% Tween-80 (MedChemExpress; HY-Y1891) and 45% phosphate-buffered saline (PBS) for injection, as recommended by the manufacturer.
Gefitinib administration
Red2Kras animals received two doses of tamoxifen through oral gavage (0.2 mg g−1 body weight) every other day to induce KrasG12D expression. Four days after the final tamoxifen dose, the mice received freshly prepared gefitinib (80 mg kg−1 in 50 μl of DMSO) or DMSO (vehicle control) through intraperitoneal injection every 4 days for 20 days.
Clodronate administration
Red2Kras animals received four doses of tamoxifen through oral gavage (0.2 mg g−1 body weight) every other day to induce KrasG12D expression. For selective depletion of macrophages in the lungs, five doses of PBS-loaded or clodronate-loaded liposomes (5 mg ml−1; LIPOSOMA) were administered through intratracheal injection (25 µl) at the time points depicted in the experiment scheme, beginning 2 days after the final tamoxifen dose. Lungs were collected and analysed 7 days after the final clodronate liposome administration.
Diphtheria toxin administration
To deplete fibroblasts, 8–10-week-old Pdgfrα–CreERT2;ZsGreen;iDTR mice were used. The animals received four doses of tamoxifen through oral gavage (0.2 mg g−1 body weight) every other day to mark Pdgfrα+ cells. Following organoid engraftment (see section “Orthotopic engraftment of KrasG12D organoids”), diphtheria toxin (Sigma) was dissolved in PBS and administered intratracheally at 50 ng per mouse every other day for seven doses, starting 21 days after the final tamoxifen dose. Lungs were collected 7 days after the final diphtheria toxin injection for analysis.
Orthotopic engraftment of Kras
G12D organoids
RFP+ KrasG12D-mutant organoids co-cultured with mesenchymal cells were orthotopically engrafted into CCR2–CreERT2;ZsGreen or Pdgfrα–CreERT2;ZsGreen;iDTR mice. A total of 50,000 RFP+ mutant epithelial cells from one to two passages of organoids were isolated by fluorescence-activated cell sorting (FACS) and mixed with 20,000 freshly isolated lung mesenchymal cells (CD31–CD45–EpCAM–) from wild-type lungs to enhance epithelial cell recovery during engraftment. The epithelial–stromal cell mixture was resuspended in 20 µl of PBS and transplanted intratracheally into recipient mice 1 day after a single dose of bleomycin treatment (1.0 U kg−1). Lungs were collected and analysed 21 days post-engraftment to assess differentiation of the engrafted cells.
Generation of EGFR
L858R
–transduced organoids for orthotopic engraftment
AT2 organoids expressing tdTomato (passages 1 and 2) derived from Sftpc–CreERT2;tdTomato lungs were dissociated into single cells and transduced with lentivirus encoding pHAGE–EGFRL858R–EGFP (Addgene plasmid no. 116276) by spin infection (2,000 rpm; 32 °C; 60 min) in the presence of polybrene (8 μg ml−1; Sigma). Transduced cells were subsequently co-cultured with mesenchymal cells. FACS was used to isolate tdTomato+GFP+ cells on day 10 post-infection, which were then expanded in co-culture with mesenchymal cells for further passages. Organoids at passages 1 and 2 were used for orthotopic engraftments. A total of 50,000 FACS-isolated epithelial cells were combined with 20,000 freshly isolated lung mesenchymal cells (CD31–CD45–EpCAM–) from wild-type lungs to support epithelial cell survival. The cell mixture was resuspended in 20 µl of PBS and transplanted intratracheally into NSG mice 1 day after a single dose of bleomycin treatment (1.0 U kg−1). Lungs were collected and analysed 21 days post-engraftment to assess differentiation of the transplanted cells.
Human adult lung tissue
The Royal Papworth Hospital NHS Foundation Trust (Research Tissue Bank Generic Research Ethics Committee approval, Tissue Bank Project no. T02233) provided de-identified LUAD and matched normal background lung tissues obtained from lobectomies. Fresh tissues were either dissociated to establish human alveolar organoids, followed by KRASG12D introduction for single-cell profiling, or fixed overnight in 4% paraformaldehyde (PFA; Thermo Fisher Scientific; 10131580) at 4 °C and processed into paraffin-embedded sections (7 μm) for immunofluorescence analysis. Paraffin-embedded sections of human LUAD tissues harbouring confirmed KRASG12D mutations were obtained from MSKCC following surgical lobectomy (Institutional Review Board no. 12-245). For human alveolar organoid co-cultures, de-identified non-tumour lung parenchymal tissues were obtained from lobectomies at Severance Hospital (IRB nos. 4-2019-0447, 4-2012-0685 and 4-2013-0770). Written informed consent was obtained from all donors before tissue collection under approved IRB protocols. No human participants were recruited specifically for this study.
Tissue collection, fixation and sectioning
All animals were euthanized by cervical dislocation, and lungs were perfused with 10 ml of PBS (Sigma; D8537) to remove blood. Lungs were slowly inflated through intratracheal injection of 2–3 ml of 4% PFA (Thermo Fisher Scientific; 10131580) in PBS, dissected and fixed in 4% PFA for 2–4 h at 4 °C. Tissues were washed three times in PBS at room temperature for 15–20 min each and then stored in PBS overnight at 4 °C. The lungs were dehydrated sequentially in 15% then 20% sucrose (Sigma; S5016) in PBS at room temperature for 1 h each, followed by immersion in 30% sucrose in PBS overnight at 4 °C. Individual lobes were separated, trimmed into smaller pieces and embedded in cryomolds filled with optimal cutting temperature compound (VWR; 361603E). Moulds were frozen on dry ice and stored at −80 °C. Frozen tissues were sectioned at 15–20 µm thickness using a cryostat, mounted onto glass slides and stored at −80 °C until staining.
Lung tissue dissociation
For lung cell isolation, mice were euthanized by cervical dislocation, and lungs were perfused with 10 ml of PBS to remove blood. The lungs were inflated through intratracheal instillation with 2–3 ml of dispase solution (Thermo Fisher Scientific; 11553550) through intratracheal injection. For mesenchymal cell isolation, collagenase I (Gibco; 17100017) was added to the dispase solution at 350 U ml−1 before inflation. The lungs were carefully dissected from the thoracic cavity and then placed on ice. Individual lobes were separated, transferred to 50-ml tubes and minced into small fragments. Tissue fragments were washed down with 3 ml of PBS. For epithelial cell isolation, 60 µl of 100 mg ml−1 of collagenase–dispase solution was added per tube. Samples were incubated in a shaking incubator at 37 °C, 190 rpm, for 45 min. DNase I (7.5 µl of 1% solution; Sigma; D4527) was added during the final 10 min of incubation. Cell suspensions were sequentially filtered through 100-µm and 40-µm strainers and washed with 2 ml of PF10 (10% fetal bovine serum (FBS) in PBS). Samples were centrifuged at 800 rpm for 5 min at 4 °C. Supernatants were removed, and pellets were resuspended in 1 ml of red blood cell lysis buffer (prepared in-house: 150 mM NH4Cl and 10 mM KHCO3 in distilled H2O) for 60 s at room temperature. Lysis was neutralized with 6 ml of Dulbecco’s modified Eagle’s medium (DMEM)/F12 (Invitrogen; 11330057). To enrich for viable cells, 500 µl of filtered FBS was carefully layered at the bottom of each tube, followed by centrifugation at 800 rpm for 5 min at 4 °C. Final cell pellets were resuspended in PF10 and transferred to 1.5-ml tubes for antibody staining.
Flow cytometry analysis
Fluorophore-conjugated antibodies were added to each sample according to the cell population being sorted. Antibodies (Supplementary Table 1) were used at a 1:200 dilution in PF10 and incubated for 20–40 min at 4 °C. DAPI was added during the final 10 min of incubation to label dead cells. A small aliquot of each sample was reserved for unstained and single-stained controls. Following incubation, cells were centrifuged, and pellets were resuspended in PF10. Cell suspensions were filtered through a 35-µm cell strainer (VWR; 352235) into polypropylene FACS tubes (Corning; 352063). Samples were sorted using a BD Influx cell sorter equipped with a 100-µm nozzle, and individual cell populations were collected into chilled 1.5-ml tubes containing 500 µl of FBS.
Primary 3D mouse lung organoid cultures
All established organoids were validated by genotyping and routinely tested for Mycoplasma contamination.
Feeder-free organoid cultures
At 7–10 days after three doses of tamoxifen (0.2 mg g−1 body weight) injection every other day, RFP+ cells were obtained from Red2Kras lungs. Organoids from RFP+ labelled cells were established as previously described7. Briefly, sorted CD31–CD45–EpCAM+RFP+ lineage-labelled cells were centrifuged at 300g for 10 min at 4 °C and resuspended in Wnt basal medium (Cambridge Stem Cell Institute’s Tissue Culture Core Facility) containing advanced DMEM (Thermo Fisher Scientific; 12491023) supplemented with 10 mM HEPES buffer (Invitrogen; 15630080), 1% penicillin–streptomycin and 1% l-glutamine (Cambridge Stem Cell Institute). Cells were counted, centrifuged and resuspended in growth-factor-reduced Matrigel (Matrigel Growth Factor Reduced (GFR); Corning; 356231). A total of 5,000–10,000 cells in 20 µl of Matrigel GFR were plated per well in eight-well LabTek Chamber Slides (Thermo Fisher Scientific; 154534 K). Matrigel was allowed to solidify at 37 °C for 30 min before adding 300 µl complete Wnt medium per well. Complete Wnt medium comprised Wnt basal medium supplemented with 1× B-27 (Thermo Fisher Scientific; 17504044), 100 ng ml−1 of recombinant FGF7 (PeproTech; 100-19-100), FGF10 (PeproTech; 100-26-100), Noggin (PeproTech; 250-38), 50 ng ml−1 of recombinant EGF (Life Technologies; PMG8043), 1 mM N-acetylcysteine, 10 mM nicotinamide and 2 µM CHIR99021 (Tocris Bioscience; 4423). Organoids were cultured at 37 °C with 5% CO2, and the medium was changed every other day. The rho kinase (ROCK) inhibitor Y-27632 (10 µM; Cambridge Bioscience; SM02-1) was added during the first 48 h of culture. For EGFR inhibition experiments, gefitinib (Selleckchem; S1025) was added from day 2 post-plating at a final concentration of 5 µM and maintained throughout the experiment. DMSO-treated cultures served as controls.
Organoid co-cultures with lung fibroblasts
At 7–10 days after three doses of tamoxifen (0.2 mg g−1 body weight) injection every other day, RFP+ mutant (CD31–CD45–EpCAM+RFP+) and tdTomato+ AT2 (CD31–CD45–EpCAM+tdTomato+) cells were obtained from Red2Kras and Sftpc–CreERT2;tdTomato lungs, respectively. To isolate lineage-labelled fibroblasts (CD31–CD45–EpCAM–ZsGreen+), Pdgfrα–CreERT2;ZsGreen mice received five daily doses of tamoxifen (0.2 mg g−1 body weight), and lungs were collected 7 days post-induction. Lung organoid co-cultures were established as previously described21. Briefly, freshly sorted epithelial cells and fibroblasts were centrifuged at 300g for 10 min at 4 °C and resuspended in 3D basic medium comprising DMEM/F12 (Gibco; 11330-032) supplemented with 10% FBS and insulin–transferrin–selenium (Corning; 25-800-CR). Cells were counted and combined at a ratio of 7,000–9,000 epithelial cells to 45,000–50,000 fibroblasts per well. Following centrifugation, cell pellets were resuspended in Matrigel GFR. A 30-µl Matrigel–cell mixture was plated per well in eight-well LabTek Chamber Slides for whole-mount staining. Domes were allowed to solidify at 37 °C for 30 min before adding 300 µl of 3D basal medium per well. Cultures were maintained at 37 °C with 5% CO2. The medium was changed every other day. The ROCK inhibitor Y-27632 (10 µM) was included for the first 48 h of culture. For EGFR inhibition experiments, gefitinib (5 µM) was added from day 2 post-plating and maintained until the end of the culture period. DMSO-treated cultures served as controls.
Organoid tri-cultures with lung mesenchymal cells and alveolar macrophages
To investigate the effect of gefitinib in organoid co-cultures, RFP+ mutant and tdTomato+ AT2 cells were isolated as described above. AMs (CD45+CD64+SiglecF+) and lung mesenchymal cells (CD31–CD45–EpCAM–) were isolated from wild-type lungs. Freshly sorted cells were centrifuged at 300g for 10 min at 4 °C and resuspended in 3D basic medium. Cells were counted and combined at a ratio of 1:5:10 (5,000 AT2 cells, 30,000 AMs and 50,000 mesenchymal cells per well). The cell mixture was centrifuged, resuspended in 100-μl Matrigel GFR containing 50% 3D basic medium and plated into 24-well Transwell inserts with 0.4-μm pore size. A total of 500 μl of 3D basic medium was added to the lower chamber, and cultures were maintained at 37 °C with 5% CO2. For inhibition of EGFR signalling, gefitinib was added at a final concentration of 5 μM. DMSO-treated cultures served as controls.
To investigate the effect of Areg and Ereg in initiating niche remodelling, tdTomato+ AT2 cells were co-cultured with wild-type mesenchymal cells and AMs as described above. A total of 500 μl 3D basic medium supplemented with granulocyte–macrophage colony-stimulating factor (20 ng ml−1; PeproTech) was added to the lower chamber, and cultures were maintained at 37 °C with 5% CO2. Recombinant Areg (20 ng ml−1; PeproTech; 315-36) and/or Ereg (20 ng ml−1; PeproTech; 100-04-5) was added to the medium and maintained for 5 days.
Co-cultures of lung mesenchymal cells and alveolar macrophages
To evaluate whether tumour-derived mesenchymal cells modulate AMs, co-cultures were established. AMs (CD45+CD64+SiglecF+) were isolated from wild-type lungs, and mesenchymal cells (CD31–CD45–EpCAM–) were isolated from either wild-type or Red2Kras lungs at 4 weeks after three doses of tamoxifen (0.2 mg g−1 body weight) injection on alternate days. Freshly sorted AMs and mesenchymal cells were centrifuged at 300g for 10 min at 4 °C and resuspended in 3D basic medium. Cells were counted and combined to create mixtures of 30,000 AMs with either 50,000 wild-type or 25,000 Red2Kras mesenchymal cells per well. Cells were centrifuged, resuspended in 100-μl Matrigel GFR containing 50% 3D basic medium and plated into 24-well Transwell inserts with 0.4-μm pore size. A total of 500-μl 3D basic medium was added to the lower chamber and replaced every other day. Cultures were maintained for 14 days, after which AMs were analysed.
To assess the effect of TLR4 inhibition on fibroblast–AM interactions, wild-type AMs and mesenchymal cells from either wild-type or Red2Kras lungs were isolated as described above. Cells were counted, combined at a ratio of 30,000 AMs to 40,000 mesenchymal cells and then plated in eight-well LabTek Chamber Slides. Co-cultures were maintained for 4 days. For TLR4 inhibition, selected wells were treated with the TLR4 inhibitor TAK-242 (3 µM; Sigma; 614316) beginning at plating (day 0) and continuing throughout the experiment. To assess the effect of inflammatory cues in fibroblasts, wild-type mesenchymal cells were treated with IL-1β (20 ng ml–1; PeproTech 211-11B-10UG) or co-cultured with AMs isolated from Red2Kras lungs for 48 h.
Mesenchymal cultures
To test whether EGFR activation induces fibrotic phenotypes, mesenchymal cells (CD31–CD45–EpCAM–) were isolated from wild-type lungs, centrifuged at 300g for 10 min at 4 °C and resuspended in 30-μl Matrigel GFR. A total of 50,000 mesenchymal cells per well were seeded in eight-well LabTek Chamber Slides for whole-mount staining. The Matrigel GFR domes were left to set for 30 min at 37 °C, before 300-µl 3D basic medium was added to each well. Recombinant Areg (100 ng ml−1; PeproTech; 315-36) was added to the medium for 5 days.
Alveolar macrophage cultures
AMs sorted from wild-type lungs were cultured under three conditions: (1) AMs alone; (2) AMs co-cultured with tumour cells; and (3) AMs co-cultured with tumour cells and fibroblasts. For each condition, cell numbers were as follows: 50,000 AMs for condition 1, 50,000 AMs with 5,000 RFP+ AT2 tumour cells for condition 2 and 50,000 AMs with 5,000 RFP+ tumour AT2 cells and 25,000 fibroblasts for condition 3. Cells were embedded in 20-µl Matrigel GFR domes and cultured in 3D basic medium for 7 days. For Areg treatment, recombinant Areg (20 ng ml−1) was added to the culture medium.
To assess responses to Tnc, AMs were isolated from wild-type lungs, plated in eight-well LabTek Chamber Slides and left to adhere overnight. Cells were then treated with Tnc (2 µg ml−1; MedChemExpress; HY-P700834) alone or in combination with the TLR4 inhibitor TAK-242 (3 µM; Sigma; 614316) for 48 h.
Inducible human LUAD organoid development
Primary human lung alveolar organoid cultures
Human alveolar organoids were established following a previous study48. AT2 cells (CD45–EpCAM+HTII-280+) were isolated from non-tumour lung parenchyma tissues using FACS, resuspended in 20-μl Matrigel GFR and plated in 48-well plates. Domes were incubated at 37 °C for 15 min to allow solidification before adding 250-μl alveolar medium. Alveolar medium consisted of DMEM/F12 supplemented with 1× B27 (Thermo Fisher Scientific; 17504044), 50 ng ml−1 of murine EGF (PeproTech; 100-15), 100 ng ml−1 of human FGF7/KGF (PeproTech; 100-19), 100 ng ml−1 of human FGF10 (PeproTech; 100-26), 100 ng ml−1 of human NOGGIN (PeproTech; 120-10 C), 3 μM CHIR99021 (Tocris; 4423), 500 nM A83-01 (Tocris; 2939), 10 μM SB431542 (Tocris; 616461), 1× penicillin–streptomycin, 500 μg ml−1 of Primocin (InvivoGen; ant-pm-1) and 1.25 mM N-acetylcysteine (Merck; A9165). The ROCK inhibitor Y-27632 (10 μM) was added to the medium for the first 2 days of culture, and the medium was replaced every 2–3 days.
Inducible vector construction, viral production and organoid infection
The plasmid pHAGE–KRASG12D was a gift from G. Mills and K. Scott (Addgene plasmid no. 116423; PIRD: Addgene_116423). For the inducible system, the KRASG12D sequence was cloned into the EF1a–TagRFP–2A–tet3G vector using In-Fusion cloning (vector kindly provided by the Emma Rawlins laboratory, University of Cambridge). Lentivirus was produced by transfecting HEK293T cells (American Type Culture Collection; CRL-11268) using a calcium phosphate protocol, and viral supernatants were collected 48 h post-transfection. Human AT2-cell-derived alveolar organoids (passages 0–2) were recovered from Matrigel GFR using dispase (1 mg ml−1; 40 min; 37 °C) and dissociated to single cells with TrypLE (5 min; 37 °C). Cells were subjected to spin infection (2,000 rpm; 32 °C; 60 min) with viral supernatant in the presence of polybrene (8 μg ml−1; Sigma), followed by feeder-free culture in Matrigel GFR supplemented with alveolar medium as described above. RFP+ cells were then isolated by FACS on day 14 or 21 post-infection, and approximately 50,000 cells were embedded in Matrigel GFR for co-culture experiments or for single-cell profiling under feeder-free conditions, respectively. For induction of the KRASG12D gene, doxycycline (2 μg ml−1; Merck) was added every 2 days, starting on day 7.
Primary human lung alveolar organoid co-cultures with primary human mesenchymal cells
Fourteen days post-infection, RFP+ infected cells were sorted by FACS and co-cultured with freshly isolated primary human lung mesenchymal cells (EpCAM−CD31−CD45−) at a 1:5 ratio (approximately 1,000 RFP+ cells with 5,000 mesenchymal cells per well) in 20-μl Matrigel GFR. Cultures were established in eight-well chamber slides (μ-Slide 8 wells; ibidi) and maintained in co-culture medium consisting of a 1:1 mixture of alveolar medium and PneumaCult (STEMCELL Technologies). KRASG12D expression was induced with doxycycline (2 μg ml−1; Merck), added every 2 days starting on day 7. Immunofluorescence analysis was performed 7 days post-induction. For EGFR inhibition experiments, gefitinib (Selleckchem; S1025; 5 µM) was added from day 7 following doxycycline induction and maintained for extra 7 days. DMSO-treated cultures served as controls.
Immunofluorescence staining
Mouse lung tissue sections
Individual cryosections were circled using a Hydrophobic PAP Pen (Sigma; Z377821) and placed in a humidified chamber. Sections were permeabilized with 0.3% Triton X-100 (Sigma; X100) in PBS for 15 min, followed by blocking with 0.3% Triton X-100 in PBS containing 5% normal donkey serum (Jackson ImmunoResearch Labs; 017-000-121) for 1 h at room temperature. Primary antibodies (Supplementary Table 1) were diluted in blocking buffer and incubated overnight at 4 °C. Sections were washed three times in 0.2% Tween-20 (Sigma; P9416) in PBS for 5 min each at room temperature, followed by incubation with secondary antibodies (Supplementary Table 1) diluted in PBS for 1 h at room temperature. The nuclear staining DAPI (Sigma; D9542) was added to the secondary antibody mix at 0.5 µg ml−1. Following staining, sections were washed three times in PBS, mounted in RapiClear 1.52 (SUNJin Lab; RC152002), enclosed with glass coverslips (VWR; 631-1574) and sealed with nail polish.
Human lung tissue sections
Human paraffin-embedded tissue sections were deparaffinized, and antigen retrieval was performed by incubation at 95 °C for 15 min in sodium citrate buffer (pH 6) (Sigma; S4641) containing 0.05% Tween-20. For immunofluorescence staining, the protocol described above for cryosections was followed.
Organoid whole-mount staining
Organoid cultures grown in LabTek Chamber Slides were fixed with 200 µl of 4% PFA for 20 min at room temperature and washed three times with PBS. Cells were permeabilized with 0.5% Triton X-100 in PBS for 15 min at room temperature, followed by blocking in 0.3% Triton X-100 in PBS containing 5% normal donkey serum for 1 h. Primary antibodies (Supplementary Table 1) diluted in blocking buffer were added to each well and incubated overnight at 4 °C. Samples were washed three times with 0.2% Tween-20 in PBS and incubated with secondary antibodies (Supplementary Table 1) diluted in 0.2% Tween-20 in PBS for 2 h at room temperature. DAPI (0.5 µg ml−1) was included in the secondary antibody solution for nuclear staining. Wells were washed three times with PBS. Chambers were then removed according to the manufacturer’s instructions. Samples were mounted in RapiClear 1.52, enclosed with glass coverslips and sealed with nail polish. The slides were allowed to dry at room temperature before imaging.
Confocal imaging, processing and quantification
Immunofluorescence images of stained sections and organoids were acquired using a Leica STELLARIS 8 white light laser inverted confocal microscope or an Olympus FV3000RS. Standard configurations were used for all experiments. All representative images were acquired using ×20 or ×40 oil objectives, except for whole-lobe tile scans, which were acquired using a ×10 objective. Confocal images were processed and analysed using Fiji (ImageJ). Signal thresholds were manually adjusted during image processing, and identical settings were applied to all representative images within the same experiment. An exception was made for endogenous fluorescent reporters (RFP and tdTomato), for which thresholds were adjusted as necessary to enable clear visualization of labelled cells in representative images.
Cell quantification in tissue sections was performed manually using the CellCounter plugin or by automated detection of DAPI+ nuclei using the ‘Analyze particles’ function. Signal thresholds were manually adjusted to distinguish marker-positive and marker-negative cells, with identical settings applied to all images within the same experiment. For most analyses, representative images and quantifications were obtained from a minimum of seven fields of view per mouse sample or experimental condition. For analyses of tumours containing fibrotic fibroblasts, 10–20 individual mutant clones were analysed per mouse. Tumour burden was quantified by defining RFP+ signal thresholds and measuring total RFP+ area using the ‘Analyze particles’ function. The RFP+ area was normalized to the total area of the whole-lobe cross section, with two to four independent tissue sections analysed per mouse. For quantification of cell numbers in organoids, detection thresholds were manually adjusted, and DAPI+ and SOX9+ nuclei were quantified using the ‘Analyze particles’ function with a minimum particle size of 10 µm2.
Quantitative reverse transcription–polymerase chain reaction
Total RNA was isolated using a QIAGEN RNeasy Micro or Mini-plus Kit according to the manufacturer’s instructions. Equivalent quantities of total RNA were reverse transcribed with SuperScript IV complementary DNA (cDNA) Synthesis Kit (Life Technologies). Diluted cDNA was analysed by real-time polymerase chain reaction (StepOnePlus; Applied Biosystems). SYBR Green assays were used for human or mouse gene expression with SYBR Green Master Mix (2×; Thermo Fisher Scientific). The primer sequences are as follows:
Mouse Gapdh: F-AGGTCGGTGTGAACGGATTTG, R-TGTAGACCATGTAGTTGAGGTCA
Mouse Arg1: F-CTCCAAGCCAAAGTCCTTAGAG, R- AGGAGCTGTCATTAGGGACATC
Mouse Ym-1: F-TGGAATTGGTGCCCCTACAA, R- CCACGGCACCTCCTAAATTG
Mouse Tnf: F-CCCTCACACTCAGATCATCTTCT, R- GCTACGACGTGGGCTACAG
Human GAPDH: F-GGAGCGAGATCCCTCCAAAAT, R- GGCTGTTGTCATACTTCTCATGG
Human KRAS: F-AGTGCCTTGACGATACAGCT, R-CCTCCCCAGTCCTCATGTAC.
Single-cell transcriptomics
Library preparation and sequencing
Lung mesenchymal cells from Confetti and Red2Kras lungs
Two weeks after three doses of tamoxifen induction (0.2 mg g−1 body weight; administered every other day), lung tissues were collected, and mesenchymal cells (CD45–CD31–EpCAM–) were isolated from Sftpc–CreERT2;Confetti and Sftpc–CreERT2;Red2Kras mice. Cells from three mice of the same genotype were pooled into a single-cell suspension to generate two separate libraries (1× Confetti and 1× Red2Kras). Cell suspensions were spun down, counted and resuspended in 0.04% bovine serum albumin (BSA; Sigma; A3294) in PBS to achieve a cell concentration of approximately 345 cells µl−1. Single-cell 3′ RNA sequencing libraries were generated according to the manufacturer’s instructions (Chromium Single Cell 3′ Reagent v.3 Chemistry Kit; 10X Genomics), and cDNA quality was assessed. Libraries were sequenced to a minimum depth of approximately 20,000 reads per cell using Illumina NovaSeq X 1.5B.
Lung stromal and immune cells from Confetti and Red2Kras lungs
Two weeks after three doses of tamoxifen induction (0.2 mg g−1 body weight; administered every other day), lung tissues were collected, and stromal and immune cells (1:1 mixture of immune cells (CD45+EpCAM–) and stromal cells (CD45–EpCAM–)) were isolated from Sftpc–CreERT2;Confetti and Sftpc–CreERT2;Red2Kras mice. Cells from three mice of the same genotype were pooled into a single-cell suspension to generate two separate libraries (1× Confetti and 1× Red2Kras). Libraries were generated as described above and sequenced to a minimum depth of approximately 20,000 reads per cell using the Illumina NovaSeq 6000.
RFP+ mutant, mesenchymal and immune cells from Areg
flox/+ and Areg
flox/flox lungs
Two weeks after two doses of tamoxifen induction (0.2 mg g−1 body weight; administered every other day), lung tissues were collected from Sftpc–CreERT2;Red2Kras;Aregflox/+ and Sftpc–CreERT2;Red2Kras;Aregflox/flox animals. Lungs from three mice of the same genotype were dissociated and pooled into a single-cell suspension. Lineage-labelled epithelial cells (EpCAM+RFP+) and a mixed population of mesenchymal and immune cells (1:1 ratio of mesenchymal cells (CD45–CD31–EpCAM–) and immune cells (CD45+CD31–EpCAM–)) were then sorted. For each genotype, two separate libraries were generated, resulting in a total of four libraries (2× Aregflox/+ and 2× Aregflox/flox). Libraries were sequenced to a minimum depth of approximately 20,000 reads per cell using Illumina NovaSeq 6000.
Human lung alveolar organoids
Feeder-free organoids from control or KRASG12D-induced RFP+ cells were used for single-cell RNA sequencing (scRNA-seq) analysis by isolating cells on day 7 following doxycycline-mediated induction. For cell isolation, organoids were incubated with dispase (1 mg ml−1; 30–60 min), dissociated with TripLE (Gibco) for 5 min and washed with PBS. Libraries were prepared as described above and sequenced to a minimum depth of approximately 20,000 reads per cell using Illumina NovaSeq 6000.
Read alignment
Raw FASTQ files containing droplet-based sequencing data were preprocessed in CellRanger (v.6.0.2). Reads were aligned to the Ensembl Mus musculus GRCm38 reference genome or Homo sapiens GRCh38 (GENCODE v.38), empty droplets were filtered out and the number of unique molecular identifiers (UMIs) mapped to each protein-coding gene was quantified to generate the final count matrices.
Quality control
Analysis of count matrices was performed in R using the Seurat package59 or Scanpy60 pipeline (v.1.9.1). Quality control metrics for each library were first assessed and used to define thresholds for filtering out low-quality cells and possible doublets. Standard cutoffs of less than 10% mitochondrial genes, more than 1,000 detected genes, more than 2,000 UMIs and fewer than 50,000 UMIs were used for most cases. For the immune cell dataset from the Red2Kras versus Confetti experiment and human alveolar organoid, cells were filtered by custom cutoff (more than 500 and less than 7,000 detected genes and more than 2,000 UMI count).
Dimensionality reduction, clustering and analysis
Seurat pipeline was used for further data processing. Briefly, filtered data were log-normalized and scaled, and the top 2,000 highly variable genes were used for principal component analysis (PCA). The first 30 PCAs were used for downstream analyses. Nearest neighbours were calculated. Cells were clustered using Louvain algorithm and visualized using UMAP. Seurat objects were integrated using Harmony. After integration, count matrices were renormalized, and PCA-based dimensionality reduction, clustering and UMAP visualization were performed. Markers for each clusters were identified using the FindAllMarkers() function, and individual populations were annotated on the basis of previously described gene markers for immune, mesenchymal and epithelial lung cell types7,23,24. Unwanted cell types were manually removed where appropriate. Cell populations of interest were subset, reclustered and reprocessed. Gene expression between clusters was visualized using the DotPlot(), VlnPlot(), FeaturePlot() and Heatmap() functions. Gene Ontology terms for ‘biological processes’ were obtained using g:Profiler on the top differentially expressed genes, applying a significance threshold of P < 0.05. Selected biologically relevant terms were presented in the figures. Cell trajectory analysis for selected populations was performed using the Monocle 3 package61. Identification of communication networks and ligand–receptor pairs between epithelial cells and fibroblasts was performed using CellChat by following standard analysis protocols62.
Integration of oncogenesis and regeneration mesenchyme datasets
To compare mesenchymal transcriptional profiles during injury response and early oncogenesis, our scRNA-seq dataset was integrated with a previously published dataset of mesenchymal cells during lung regeneration24. Both datasets were processed and filtered as described above. A total of 10,000 anchor features were identified using the FindIntegrationAnchors() function, and the datasets were integrated using IntegrateData() function. Dimensional reduction, log normalization, scaling, clustering and downstream analyses were performed as described above.
Analysis of early-stage human LUAD scRNA-seq datasets
To investigate the presence of mutant epithelial states and fibrotic fibroblasts in early-stage human LUAD, we reanalysed a previously generated scRNA-seq dataset for primary LUAD tumours (stages I–III) and distant normal tissue45. Only samples with confirmed EGFR wild-type status were used for the analysis. For quality control, cutoffs of less than 20% mitochondrial genes, more than 200 detected genes and fewer than 3,000 detected genes were used. Dimensional reduction, log normalization, scaling, clustering and downstream analyses were performed as described above using the Seurat (v.5) package on R studio (v.4.4.2). From the resulting dataset, epithelial cells were reclustered on the basis of EPCAM expression, with ciliated cells (FOXJ1) excluded. Mesenchymal cells were reclustered on the basis of COL1A1 expression, and fibroblasts were further reclustered on the basis of PDGFRα, COL13A1 and COL14A1 expression. Gene expression between cell clusters was visualized as described above.
Statistical analysis and reproducibility
All in vivo experiments were performed in at least two independent experiments, with individual animals considered as biological replicates. All in vitro assays were performed in at least three independent experiments, and summary statistics were calculated from experiment-level mean values, unless otherwise stated. For most quantifications, 10–20 tumours or a minimum of seven fields of view were analysed per mouse or per experimental condition, and measurements were treated as nested within each mouse or condition and averaged to obtain mouse-level or experiment-level values for statistical analysis. For tumour burden, tumour area was normalized to the total lobe area across sections, with two to four independent tissue sections analysed per mouse, and section-level measurements were averaged to yield a single mouse-level value for statistical analysis. For individual tumour size, two slides per animal, spaced 100 µm apart, were evaluated. Total lobes and lesion areas were defined manually and measured using QuPath open-source software (v.0.6.0).
Data are presented as mean ± s.e.m. Statistical analyses were performed using Prism software (GraphPad; v.7.0) or R. Statistical significance was assessed using two-tailed unpaired Student’s t-test or two-tailed Mann–Whitney test, as indicated in the figure legends. The number of animals or in vitro assays is stated in the figure legends (n = x mice per group; n = x independent experiments per condition). Representative images are shown, and the corresponding quantifications are derived from the indicated numbers of animals and experiments. Nested statistical analyses that accounted for within-mouse or within-experiment variability yielded results consistent with analyses of mouse-level or experiment-level means. Therefore, these summary values were used for statistical comparisons, and the specific statistical tests and exact P values are reported in the figures and figure legends.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.