Li dendrite growth in solid electrolytes
LLZTO solid electrolytes were obtained from Toshima Manufacturing Co., Ltd. The pellets were mechanically ground to a thickness of approximately 150 μm, with final polishing performed using a 0.05-μm alcohol-based colloidal silica suspension. To reduce the interfacial resistance between lithium and the solid electrolyte, the thin LLZTO discs were immersed in 1 M HCl for 30 s to remove surface contaminants, following the procedure demonstrated in ref. 51. Immediately after the acid treatment, the solid electrolyte discs were transferred into an argon-filled glovebox (O2 and H2O < 0.5 ppm). Lithium foil (MaTeck Material Technologie & Kristalle GmbH) was scraped using a plastic tweezer to expose a fresh, shiny surface. A 3-mm-diameter lithium pad was then punched out and stuck to the LLZTO disc. The assembled cell was placed on a hotplate and baked at 130 °C for 1 h.
To study the interaction between lithium dendrites and specific features of interest, a single lithium metal pad was used as the counter electrode. A tungsten probe was placed on the surface of the solid electrolyte to serve as the working electrode, where lithium dendrites nucleated and grew41. A constant current was applied between the lithium metal pad and the tungsten needle using a SP-200 potentiostat (Bio-Logic Science Instruments GmbH). Electrochemical impedance spectroscopy (EIS) data were recorded in the frequency range between 10 Hz and 7 MHz with an amplitude of 50 mV using a SP-200 impedance analyser (Bio-Logic). The growth of lithium dendrites was conducted entirely within the glovebox and monitored using a camera mounted on a stereo microscope (KERN & SOHN GmbH).
Lithium dendrite growth through the symmetric cell configuration was cycled using the same potentiostat equipped with a pressure stand (Imada Inc.). Before applying the bias, the symmetric cell was heated to 130 °C using a heating sleeve (RS Components Ltd.) to improve the interfacial contact between the lithium metal and the solid electrolyte. After short-circuiting, the lithium metal was removed using sandpaper with a grit size of 1,200. The short-circuited solid electrolyte was then taken out of the glovebox, soaked in epoxy overnight for curing and subsequently polished to the region in which features resembling lithium dendrites could be observed, as shown in Extended Data Fig. 1f,g. Extended Data Fig. 1e schematically illustrates the sample preparation procedure. Extended Data Fig. 1f shows the surface after rough polishing with 320-grit sandpaper and Extended Data Fig. 1g shows the result after fine polishing using a 0.1-μm SiO2 polishing suspension. The lithium metal on the plating side, where dendrite growth occurred, could be easily peeled off by hand, as shown in Supplementary Figs. 10, 11 and 17. Therefore, no sandpaper was used to remove the lithium electrode, in contrast to the procedure used for the samples shown in Extended Data Fig. 1 and Supplementary Fig. 6.
Cryogenic FIB, SEM and EBSD
Using an inert high-vacuum (< 10−7 mbar) cryogenic transfer suitcase (Ferrovac AG), hereafter referred to as the ‘suitcase’, the LLZTO disc was transferred from the argon-filled glovebox to a Thermo Fisher Scientific Helios 5 CX Ga FIB/SEM system. The Helios 5 is equipped with an Aquilos cryo-stage featuring free rotation capability and a Thermo Fisher Scientific EZ-Lift tungsten cryogenic micromanipulator. Both the cryo-stage and the manipulator were maintained at −190 °C using active heating control and a nitrogen flow rate of 190 mg s−1. All operations inside the FIB/SEM system—including SEM imaging, FIB cutting, TEM lamella preparation and EBSD—were conducted at a stable temperature of −190 °C. The TEM lamella was welded onto both the micromanipulator needle and a copper grid by means of redeposition induced by line cuts, as shown in Supplementary Fig. 33. Detailed lamella preparation procedures have been described in previous works52,53. Once thinned to below 150 nm, the lamella and the bulk sample were transferred back into the argon glovebox using the suitcase. The interaction between the electron beam and the solid electrolyte is strongly suppressed at cryogenic temperatures. No electron-beam-induced lithium nucleation was observed under cryogenic conditions, in contrast to the artefacts frequently encountered at room temperature11,54.
EBSD patterns of the LLZTO pellet were collected at cryogenic temperature (−190 °C) using a direct electron detector (Clarity Plus, EDAX LLC). Kikuchi patterns were acquired under an accelerating voltage of 10 kV and a beam current of 2.8 nA. To analyse diffraction from a lithium dendrite within the solid electrolyte, a lamella was prepared following the same procedure described above, except the final lamella thickness was maintained at approximately 1 μm. Supplementary Fig. 34 shows the TKD lamella, which maintains its mechanical integrity without any observable bending or distortion induced by ion-milling preparation. Moreover, because the sample was prepared using Ga+ FIB at cryogenic temperature, strain rearrangement during ion milling is expected to be strongly suppressed and therefore experimentally negligible, as reported in several previous studies37,38,55. TKD patterns of the lithium dendrite were also acquired using the same direct electron EBSD detector. The diffraction patterns of the lithium dendrite were analysed using spherical indexing56—a new technique that enables improved pattern recognition and orientation determination for low-symmetry or low-quality patterns. In contrast to the classical analysis technique that uses a Hough transform for detection of the Kikuchi bands in Kikuchi patterns57, spherical indexing is an advanced image matching technique, in which the experimental pattern is compared with a theoretical master pattern. The comparison is done by developing both the experimental and the master pattern into a series of spherical harmonic functions and comparing them by a spherical cross-correlation function. Because spherical indexing matches the whole pattern, this technique can be applied very robustly with weak diffraction patterns, typically obtained from lithium. Furthermore, because the master pattern can be calculated for any diffraction voltage and because the matching is executed directly on the diffraction sphere, the technique is independent of the acceleration voltage of pattern generation and can also be applied to low-voltage patterns. The classical Hough transform, which detects straight lines, fails in this case because of the high curvature of low-energy Kikuchi lines. Spherical indexing, together with the necessary image preprocessing (static and dynamic background subtraction and contrast enhancement) were done using an early build of the software OIM Analysis 9.1 produced by Ametek EDAX. The master pattern was calculated for 10 kV and 20° of sample tilt in transmission. The bandwidth, a parameter that describes the amount of details that is matched in the pattern, was set to 127.
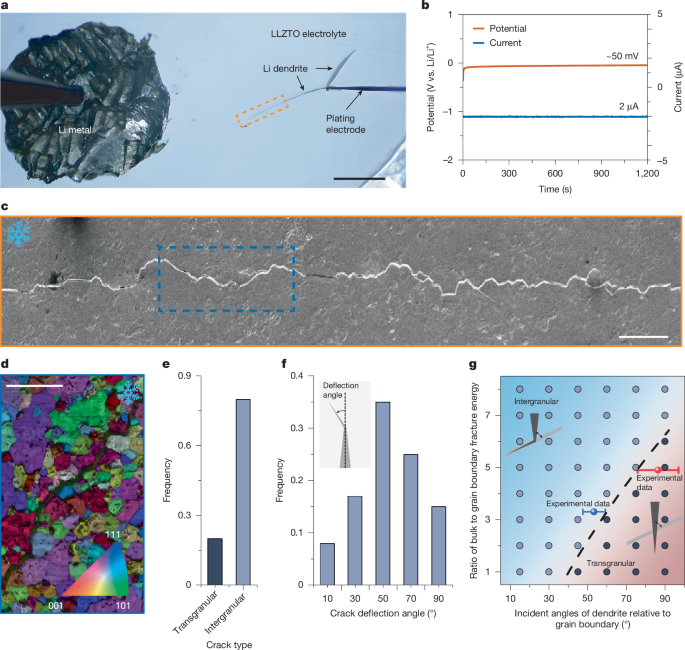
The incident angles between the dendrite and grain boundaries were measured from EBSD results for both intergranular and transgranular fractures. In both cases, the incident angle values follow a normal distribution. The mean values, along with the 95% confidence intervals extracted from Fig. 1f and Supplementary Fig. 4b, were fitted and plotted in Fig. 1g. The positions of the red and blue dots were placed such that their error bars just begin to intersect the boundary between intergranular and transgranular regions, as indicated by the dashed line.
Cryo-STEM
The STEM lamella was loaded in a Mel-Build holder inside an argon-filled glovebox and then kept under inert argon atmosphere during sample transfer. All analysis was performed at cryogenic conditions (−150 °C). STEM was performed on a Titan Themis microscope (Thermo Fisher Scientific) operated at 300 kV. The aberration-corrected probe has a convergence semiangle of 23.8 mrad. High-angle annular dark-field and annular bright-field STEM micrographs were collected using respective angular ranges of 73–200 and 8–16 mrad. STEM energy-dispersive X-ray spectroscopy spectrum imaging was acquired using a Super-X detector. STEM-EELS spectrum imaging was performed using a Quantum ERS spectrometer (Gatan) with a collection angle of 35 mrad. To facilitate comparison with EELS spectra reported in the literature, we opt to show raw EELS spectra from selected areas in Supplementary Figs. 12b and 14d,e. Multivariate statistical analysis was performed on the spectrum imaging datasets to separate backgrounds and signals from different lithium-containing phases29,58,59. For lithium count maps shown in Fig. 2f,g, power law background was modelled for components 1 and 2, with respective fitting windows of (45, 50) eV and (45, 57) eV. The integration window was kept to (57, 67) eV. As evidenced in Supplementary Fig. 12, the Li K-edge onsets of the LLZTO and the Li/LiOH phases are different. The quantification of lithium is hence facilitated by multivariate statistical analysis29, for which most of the spatial variance in EELS signal can be expressed in components 1 and 2. As shown in Supplementary Fig. 12c–f, component 1 is mainly located in the dendrite area and the spectral feature is LiOH-like; component 2 relates to the LLZTO area surrounding the dendrite, with LLZTO-like spectral feature. Component 3 no longer resembles a physical spectrum, as it represents small differential signals to modify the two leading components. This observation confirms the dominance of the Li/LiOH and LLZTO phase in this area. Four-dimensional STEM diffraction imaging was recorded using the pixelated detector Electron Microscope Pixel Array Detector (EMPAD, Thermo Fisher Scientific) and a probe convergence semiangle of 0.65 mrad.
Small-scale mechanical testing on LLZTO solid electrolyte
LLZTO solid electrolytes with a thickness of 1 mm were mechanically ground and polished, with the final step performed using a 0.05-μm alcohol-based colloidal silica suspension. The samples were immersed in 1 M HCl for 30 s to remove surface contaminants. Immediately after the acid treatment, the nanoindentation experiments were conducted using an iMicro Nanoindenter (KLA Inc.) under ambient environment. A constant indentation strain rate of \(\dot\varepsilon =0.1\,\rms^-1\) was applied using a Berkovich diamond pyramidal indenter. Four independent sets of experiments were performed to verify repeatability (Supplementary Fig. 30), with a total testing duration of approximately 30 min. Although a surface carbonate layer can form on LLZTO on exposure to ambient conditions, its thickness within about 0.5 h of air exposure is expected to be negligible compared with the indentation depth60.
Phase-field fracture modelling
Finite strain kinematics
A microstructural domain \(\mathcalB_\subset \mathbbR^3\) undergoing deformation is described by a mapping \(\boldsymbol\mathcalX(\bfx):\mathcalB_\to \mathcalB\), which correlates each material point \(\bfx\in \mathcalB_\) to its corresponding position \(\boldsymbol\mathcalX\) within the deformed domain \(\mathcalB\). The deformation gradient is denoted by \(\bfF=\frac\partial \boldsymbol\mathcalX\partial \bfx=\nabla \boldsymbol\mathcalX\).
In the present work, the total deformation gradient is multiplicatively decomposed as:
$$\bfF=\bfF_\rme\bfF_\rmi\bfF_\rmp,$$
(1)
in which Fe represents the elastic deformation, Fi captures the deformation induced by the electromechanical reaction (that is, lithium plating) and Fp accounts for the plastic deformation within the lithium dendrite. The volumetric change from lithium plating, Ji, can be given by the following equation:
$$J_\rmi=\textdet\bfF_\rmi.$$
(2)
We define the plastic and volumetric velocity gradient tensors in the intermediate configurations as Lp and Li, respectively. The evolution equations for Fp and Fi can be derived as:
$$\dot\bfF_\rmp=\bfL_\rmp\bfF_\rmp,$$
(3a)
$$\dot\bfF_\rmi=\bfL_\rmi\bfF_\rmi.$$
(3b)
Lithium deposition electromechanics
The volumetric expansion of the solid electrolyte lattice, resulting from the reduction of lithium ions at the reaction site, is captured through the swelling model proposed by Liu et al.61 and Narayan et al.62. The volumetric change is described by the following equation:
$$J_\rmi=1+\varOmega \eta _\textmax\bar\eta .$$
(4)
Here \(\bar\eta \) is the normalized quantity of deposited metallic lithium and ηmax denotes the concentration of metallic lithium under fully lithiated conditions. The parameter \(\bar\eta \) serves as an order parameter that captures the emergence of deposited lithium in the solid electrolyte. The parameter Ω in equation (4) is the molar volume of lithium and is taken to be constant.
The local volume change rate arising from the deposition of lithium is given by:
$$\dotJ_\rmi=J_\rmi\rmtr\bfL_\rmi,$$
(5)
in which tr refers to the trace operation.
Substituting equation (4) into equation (5) results in:
$$\rmtr\bfL_\rmi=\frac\varOmega \eta _\textmax\dot\bar\eta 1+\varOmega \eta _\textmax\bar\eta .$$
(6)
Provided that the volume expansion occurs isotopically, we can derive the velocity gradient as:
$$\bfL_\rmi=\frach(\bar\eta )3\frac\varOmega \eta _\textmax\dot\bar\eta 1+\varOmega \eta _\textmax\bar\eta \bfI,$$
(7)
in which I is a second-order identity tensor. The interpolation function h acts as a regulator to ensure that deposition only takes place in regions in which electrons are available for the reduction process, that is, in the vicinity of the dendrite. The interpolation function h is taken as:
$$h(\bar\eta )=\bar\eta ^3(6\bar\eta ^2-15\bar\eta +10).$$
(8)
Phase-field fracture model
In phase-field damage models, sharp cracks are treated as diffuse regions with gradually degraded material properties. This approach eliminates the necessity of explicitly tracking the crack interface. In this work, the energy formulation is based on the Griffith criterion. Therefore, the total free energy can be obtained as follows:
$$\psi =\int _\mathcalB_((1-\varpi )\psi _\rmLLZTO^\rmE+\varpi \psi _\rmLi^\rmE+\psi _\rmD)\rmd\bfX,$$
(9)
in which \(\psi _\rmLLZTO^\rmE\) and \(\psi _\rmLi^\rmE\) delineate the elastic energy density contributions from LLZTO and lithium, whereas ΨD accounts for the surface energy density associated with the newly formed crack surfaces. In the phase-field damage model, the order parameter d ∈ [0, 1] represents the degree of material degradation, in which d = 1 corresponds to a fully intact state and d = 0 indicates complete material failure. The interpolation parameter \(\varpi \) is introduced to distinguish between the energy contributions in the solid electrolyte and the lithiated region of the dendrite within the solid electrolyte.
To ensure that the crack can only initiate and propagate under tensile stresses, the following decomposition is used:
$$\psi _\rmLLZTO^\rmE=g(d)\psi _\rmLLZTO^\rmE++\psi _\rmLLZTO^\rmE-.$$
(10)
According to the above expression, only the tensile contribution to the energy \(\psi _\rmLLZTO^\rmE+\) is diminished by the degradation function g(d), typically defined as g(d) = d2, whereas the compressive component \(\psi _\rmLLZTO^\rmE-\) remains unaffected.
The equations pertaining to the tensile and compressive components of LLZTO elastic energy can be established through the following relations:
$$\psi _\rmLLZTO^\rmE+=\frac12\bfS_\rmLLZTO^+:\bfE,$$
(11a)
$$\psi _\rmLLZTO^\rmE-=\frac12\bfS_\rmLLZTO^-:\bfE,$$
(11b)
in which E = (FeTFe − I)/2 is the Green–Lagrange strain and \(\bfS_\rmLLZTO^+\) and \(\bfS_\rmLLZTO^-\) are obtained through the following:
$$\bfS_\rmLLZTO^+=\mathbbP^+:\bfS_\rmLLZTO^$$
(12a)
$$\bfS_\rmLLZTO^-=\mathbbP^-:\bfS_\rmLLZTO^.$$
(12b)
Here \(\bfS_\rmLLZTO^\) is the second Piola–Kirchhoff stress within the solid electrolyte. The fourth-order projection tensors \(\mathbbP^+\) and \(\mathbbP^-\), derived within a thermodynamically consistent framework, are formulated as:
$$\beginarrayl\mathbbP^\pm =\frac\partial \bfS_\rmLLZTO^\pm \partial \bfS_\rmLLZTO^=\mathop\sum \limits_i=1^3\mathop\sum \limits_j=1^3\frac\partial \gamma _i^\pm \partial \lambda _j\bfn_i\otimes \bfn_i\otimes \bfn_j\otimes \bfn_j\\ \,\,+\mathop\sum \limits_i=1^3\mathop\sum \limits_j\ne i^3\frac\gamma _i^\pm -\gamma _j^\pm \lambda _i-\lambda _j\bfn_i\otimes \bfn_j(\bfn_i\otimes \bfn_j+\bfn_j\otimes \bfn_i),\endarray$$
(13)
in which, for i = 1, 2, 3, λi and \(\gamma _i^\pm \) correspond to the eigenvalues of \(\bfS_\rmLLZTO^\) and \(\bfS_\rmLLZTO^\pm \), respectively. On the other hand, the tangent modulus is computed using the hybrid scheme proposed by Ambati et al.63, yielding the following expression:
$$\mathbbC=g(d)(1-\varpi )\mathbbC_\rmLLZTO^+\varpi \mathbbC_\rmLi^.$$
(14)
We define the binary interpolation parameter \(\varpi \) based on the following relation:
$$\varpi =\left\{\beginarrayc1,\,\rmif\,g(d)^2\mathbbC_\rmLLZTO^\rmVoigt:\mathbbC_\rmLLZTO^\rmVoigt\le \mathbbC_\rmLi^\rmVoigt:\mathbbC_\rmLi^\rmVoigt\\ 0,\,\rmif\,g(d)^2\mathbbC_\rmLLZTO^\rmVoigt:\mathbbC_\rmLLZTO^\rmVoigt > \mathbbC_\rmLi^\rmVoigt:\mathbbC_\rmLi^\rmVoigt\endarray\right..$$
(15)
The surface energy is given by:
$$\psi _\rmD=\frac\mathcalG_\rmcl_(1-d)+\frac12\mathcalG_\rmcl_^2,$$
(16)
in which l0 is damage characteristic length and \(\mathcalG_\rmc\) corresponds to the critical energy release rate.
In this study, we assume that cracks within the solid electrolyte remain fully lithiated. This implies that crack propagation and the formation of metallic lithium coincide. This assumption enables the following relationship: \(\bar\eta =1-d\).
By considering isothermal and adiabatic processes, the evolution of the damage order parameter can be derived through an Allen–Cahn type of relation given below:
$$\dotd=-M\left[2d\mathcalH_\rmLLZTO-\frac\mathcalG_\rmcl_-\mathcalG_\rmcl_\rmDiv\nabla d\right],$$
(17)
in which the parameter M denotes the damage mobility parameter, which controls the rate of damage promotion in the simulation. The history field function \(\mathcalH_\rmLLZTO\) is introduced to ensure the irreversibility of the damage, which is expressed as:
$$\mathcalH_\rmLLZTO(\bfX,t)=\mathop\textmax\limits_t\in [0,T]\psi _\rmLLZTO^\rmE+(\bfE(\bfX,t)),$$
(18)
in which E(X, t) refers to Green–Lagrange strain.
Mechanical model of lithium
In the present model, we assume that lithium can undergo isotropic plastic deformation. On plastic deformation, the isochoric response of the material is connected to the deviatoric stress \(\bfM_\rmdev^\rmp=\bfM_\rmp-\frac13\rmtr\bfM_\rmp\bfI\), in which the Mandel stress Mp serves as the work-conjugate measure to the plastic velocity gradient Lp and acts as the driving force that governs its evolution.
On the basis of this plasticity model, the strain rate can be computed by the following relation:
$$\dot\gamma ^\rmp=\dot\gamma ^{\left(\sqrt\frac32\frac\parallel \bfM_\rmp^\rmdev\parallel _\rmFM\xi \right)}^n,$$
(19)
in which the internal variable ξ is akin to the slip resistance in the phenomenological crystal plasticity model. In equation (19), \(\dot\gamma ^\) denotes the reference strain rate and M is the Taylor factor. Consequently, the associated plastic velocity gradient Lp, which operates within the lattice configuration, is expressed as:
$$\bfL_\rmp=\frac\dot\gamma ^\rmpM\frac\bfM_\rmp^\rmdev\parallel \bfM_\rmp^\rmdev\parallel _\rmF.$$
(20)
The value of ξ is set to approach a stationary value ξ∞ asymptotically from its initial value ξ0 according to the following hardening rule:
$$\dot\xi =\dot\gamma ^\rmph_^a\rmsgn\left(1-\frac\xi \xi _\infty ^\ast \right),$$
(21)
in which h0 is the initial hardening and a indicates the stress sensitivity exponent. In equation (21), \(\xi _\infty ^\ast \) is the modified saturation hardening value and takes the following form:
$$\xi _\infty ^\ast =\xi _\infty +\frac(\sinh ^-1(\dot\gamma ^\rmp/c_1))^1/c_2c_3(\dot\gamma ^\rmp/\dot\gamma ^)^1/n.$$
(22)
This formulation introduces a dependence of the saturation hardening value on the shear strain rate, enabling controlled adjustment through the parameters ci. Last, the Mandel stress Mp can be related to the second Piola–Kirchhoff stress S through the following expression:
$$\bfM_\rmp=\bfF_\rmi^\rmT\bfF_\rmi\bfS.$$
(23)
Stress equilibrium
The balance of linear momentum requires satisfying the following relation:
$$\rmDiv\bfP=\bf,$$
(24)
in which P is the first Piola–Kirchhoff stress.
Simulation set-up
The two-dimensional simulation is conducted under plane strain boundary conditions on a 128 × 256 computational grip, with no applied external mechanical deformation. A pre-existing notch is introduced at the start to represent imperfections at the lithium anode–solid electrolyte interface. To study lithium-plating-induced crack propagation across grain boundaries, we use a bicrystal geometry in which the grain boundary is assigned a range of fracture energies and deflection angles to account for variations in grain boundary fracture behaviour. To investigate the interaction between lithium dendrite and engineered voids in LLZTO, circular and transverse voids are introduced ahead of the lithium dendrite within the LLZTO electrolyte. To examine the influence of residual stress on lithium dendrite propagation, an external compression stress of 1 MPa is applied to the model (Supplementary Fig. 29d,e). In all other simulations, no external mechanical loading is applied. Moreover, three-dimensional simulations are performed with a grid of 128 × 256 × 10, in which the pre-existing notch extends through the thickness of the geometry. These three-dimensional simulations (Fig. 3e) verify the two-dimensional results and confirm that the predicted fracture behaviour remains consistent across both geometries.