In 2021, dermatologist David Ozog was on holiday with his family in the Bahamas, when his 18-year-old son had a massive stroke. The teenager was airlifted to Florida, and then to Chicago for surgery. As his son was lying partially paralysed in a hospital bed, Ozog got a call from a colleague who had an unconventional suggestion.
Forget SkinTok: the real science of skincare and why it matters for your healt
The colleague, a dermatologist at Harvard Medical School in Boston, Massachusetts, told Ozog about research he was conducting with the US Department of Defense. Early results hinted that red and near-infrared light applied to the head might protect neural tissue after brain injury. He urged Ozog to consider trying it on his son.
Ozog stayed up until 4 a.m. that night reading scientific papers and, ultimately, ordering several panels made of red and near-infrared light-emitting diodes (LEDs). “I started sneaking them into the hospital,” says Ozog, who works at Henry Ford Health in Grand Rapids, Michigan.
Today, his son is walking and back in university. Ozog cannot prove that light therapy made a difference, but he thinks that it helped. He has since become a convert to an idea that, at the time, was considered fringe. “I thought the same thing,” he says, “How could shining this thing on you possibly have any biologic effect?”
But what was at the margins of medicine just a few years ago is now edging towards the mainstream. Red-light devices are increasingly appearing in dermatology offices, wellness centres, locker rooms and homes. According to some projections, the global market will surpass US$1 billion by 2030, propelled by a surge of companies promising benefits for everything from ageing skin to attention deficit hyperactivity disorder (ADHD) — claims echoed widely across social media.
Experts warn that there is considerable hype about red-light therapy. But a growing body of legitimate science has been exploring the benefits for several conditions. Clinical studies have reported improvements in peripheral neuropathy1, retinal degeneration2 and certain neurological disorders3. For some indications, expert groups now recommend red-light regimens1.
Researchers are also uncovering how red and near-infrared light might exert these effects. Mitochondria — the power plants of the cell — are emerging as a central piece of the puzzle.
The science behind these benefits is growing at a time in which humans are exposed to less red light than ever before. People spend more time indoors away from the Sun, and efforts to conserve energy have narrowed the spectrum of indoor lighting, eliminating many red and near-infrared wavelengths (see ‘Light by the source’). Some scientists are now asking whether these factors might have biological consequences. “We’re literally being starved of something that, biologically, we’ve evolved to receive,” says Ozog.
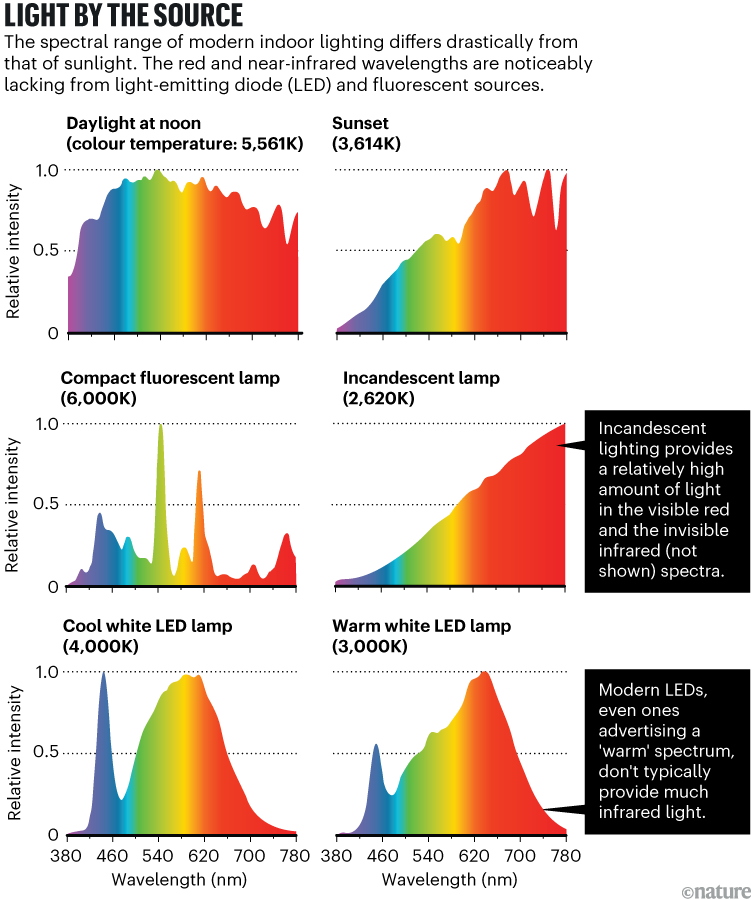
Source: K. M. Zielinska-Dabkowska/Asensetek Lighting Passport Pro Standard Spectrometer
From fringe to clinic
Light’s role in human health is hardly a new idea. For more than a century, scientists have known that ultraviolet light drives vitamin D production. The 1903 Nobel Prize in Physiology or Medicine recognized concentrated light as a treatment for skin tuberculosis. Bright-light therapy is standard care for seasonal affective disorder, and narrowband ultraviolet light remains a mainstay for psoriasis.
“The whole spectrum is doing a range of things beneficial to us,” says Glen Jeffery, a neuroscientist at University College London. Modern photobiomodulation — the use of red and near-infrared light comprising wavelengths, from about 600 nanometres to 1,100 nanometres, to influence cellular processes — emerged in the 1960s after Hungarian scientists accidentally discovered that low-level red light stimulated hair growth in rodents4. Interest accelerated in the 1990s, when NASA scientists experimenting with using red LEDs to grow plants in space noticed that small cuts on their hands healed unusually quickly under the lights5.
Smart brain-zapping implants could revolutionize Parkinson’s treatment
Over the past decade, the evidence has solidified in several clinical niches. In 2025, Ozog joined more than 20 specialists in a major consensus review1, which concluded that the therapy was safe and effective for several types of ulcer, peripheral neuropathy, acute radiation dermatitis and androgenic alopecia, a type of pattern hair loss. Last year, the US Food and Drug Administration approved a red-light device for dry age-related macular degeneration. And since 2020, red-light therapy in the mouth has been included in clinical guidelines for preventing and treating cancer-therapy-related oral mucositis — painful mouth ulcers that can limit treatment and disrupt nutritional intake.
Ozog laments that the therapy isn’t more widely applied in cancer care: “Here is a simple, safe, inexpensive treatment that’s probably used in about 10% of treatment centres.”
Beyond these footholds, researchers are exploring broader effects of the therapy. Clinical trials report improved muscle recovery in athletes6, as well as reductions in depression symptoms7 and in pain8 for people with osteoarthritis and fibromyalgia. Small human trials and animal studies also suggest potential benefits for metabolic9 and cardiovascular disease10. A small randomized study in Brazil, published in 2022, found that people with severe COVID-19 who received daily light therapy left the hospital an average of nearly four days earlier than controls did11.

Once used just in clinical contexts, red-light therapy has become a booming wellness trend.Credit: Abbie Parr/AP Photo/Alamy
Still, for some scientists, the most striking early results involve the brain. In mouse models of Parkinson’s disease (PD), photo- biomodulation applied atop the head preserved dopamine-producing neurons deep in the brain12 — cells whose loss drives the disorder’s progression.
Researchers have seen benefits in animal models of PD last for weeks after treatment, and early human trials are under way, using optical fibres that put the light close to the diseased cells. “The holy grail of neuroscience research is finding an effective neuroprotective treatment that protects the cells from dying,” says John Mitrofanis, a neuroscientist at the University of Grenoble Alpes in France.
Several transcranial devices are being developed that could offer more practical delivery of photons, to treat a variety of psychiatric and neurological diseases. Unpublished results from Mitrofanis’s team hint that transcranial light “makes an older brain look more like a younger brain”, he says. Yet getting enough photons through the human skull to produce a meaningful effect remains a challenge. Brian Pryor, chief executive at BWtek Medical, a medical-device company in Newark, Delaware, says his team found that higher photon doses have greater impacts on the brain. Devices with such strong outputs “may be too powerful to sell over the counter”, he says. Several more clinical trials are planned or are currently under way.
Much remains unresolved: optimal wavelengths, intensities, timing, delivery methods and even pulse rates for different indications. Also unclear is whether people’s age or skin colour should determine the dose they receive. And beneath it all lies a deeper question: how might light produce such wide-ranging biological effects?
Turning light into cellular fuel
Converging lines of biophysical and biochemical evidence have led researchers to focus on mitochondria, the energy-producing organelles found in most cells of the body.
Red to near-infrared wavelengths of light scatter much less than do shorter blue and ultraviolet wavelengths. As a result, some photons — mainly those in the near-infrared range — can pass through clothing, and a fraction can penetrate centimetres into tissue, raising the possibility that they affect cells well beneath the skin. Wavelengths between around 600 and 700 nanometres and 760 and 940 nanometres are often reported to produce biological responses. These ranges closely match the wavelengths most readily absorbed by cytochrome c oxidase, a key enzyme in the mitochondrial electron transport chain that contributes to the formation of the cellular fuel known as adenosine triphosphate (ATP).
Can flashing lights stall Alzheimer’s? What the science shows
Evidence suggests that cells can absorb these wavelengths, and that the light nudges the electron transport chain into a more active state, boosting production of ATP13. Downstream effects include improved blood flow and changes in inflammation and oxidative stress. Some researchers have proposed an extra mechanism: red and near-infrared light reduces the viscosity of water, allowing energy-producing machinery to move more easily14. “For the mitochondrial engine to work properly, it needs a lubricant,” says Robert Fosbury, an astronomer-turned-photobiomodulation researcher at University College London.
Many investigations have concentrated on cells with densely packed mitochondria, including those in human embryos and the eye. A new clinical trial sponsored by Columbia University in New York City will test whether brief exposure to red light can improve the quality of embryos produced during in vitro fertilization. Jeffery’s lab studies the ageing retina, in which functional decline correlates with mitochondrial deterioration. Work from his group and others suggests that photo- biomodulation could help to protect retinal health15. Jeffery’s team’s work has even suggested that the light might not need to be delivered directly to the eyes to have an effect.
In earlier research, his group found that 15 minutes of red-light exposure on the back blunted post-meal spikes in blood sugar levels14. Jeffery speculates that mitochondria might communicate with one another and “are behaving like a community across the body”.
Such findings have generated not just excitement, but also debate. Juanita Anders, a photobiomodulation researcher at the Uniformed Services University in Bethesda, Maryland, who spoke independently of her government role, questions the premise and says that larger, more carefully controlled studies are needed to determine potential distal or systemic impacts. One hypothesized route involves biophotons, or faint light produced by the cells themselves. Mitochondria are a major source of these particles and might use them to signal cellular health, says Mitrofanis. He co-authored a 2025 study that found that photobiomodulation altered biophoton output, particularly in stressed cells16.
A pattern might be emerging: when cells are healthy, external light often has little effect. But during illness or metabolic stress, in which mitochondrial dysfunction is common, its impact seems to be stronger. That distinction could help to explain why results vary across studies.
Still, mitochondria probably do not tell the whole story. “Even if you use a mitochondrial inhibitor, we still see a therapeutic response,” says Praveen Arany, a dentist and oral biologist at the University of Buffalo in New York, who studies the effects of red light.
Dosage seems crucial. Many researchers point to a biological ‘sweet spot’ between too little and too much light. Others argue that wavelengths should not be considered in isolation, emphasizing the importance of a broad spectrum of light — the way nature delivers it. “I like to think about ratios of light,” says Elke Buschbeck, an evolutionary biologist at the University of Cincinnati in Ohio.