As a postdoctoral researcher, Jennifer Rabin spent her days studying how risk factors for heart disease corresponded to the chance of developing Alzheimer’s disease. In 2019, shortly after she started her own laboratory at the University of Toronto, Canada, she met with a PhD student who wanted to study women’s health. Together, they began to explore whether an obvious sex difference, the drop in the hormone oestradiol during the menopause transition, might be connected to higher rates of Alzheimer’s in women. Rabin was astounded by the research literature — or rather, its scarcity. “I just couldn’t believe how little we know,” she says.
The student’s project was a turning point for Rabin. Over the next few years, she realized that across many conditions, women’s health was uncharted scientific territory. Rabin thought that profound insights could emerge from dissecting large data sets for health differences between sexes. “It felt like this unknown area that could unlock a lot of answers,” she says.
Most health and medical data are drawn from research on male bodies. Until the 1990s, women were rarely included in clinical studies for fear of harming unborn children, and female animals were excluded from studies using model organisms because of a prevailing belief — now discredited — that hormonal fluctuations made their biology too complicated. This systematic exclusion has contributed to delays in diagnosis, drugs that are less effective or dangerous for women and the scarcity of evidence-based guidelines to address women’s health needs. The little research that exists has focused mostly on ‘bikini medicine’ — equating women’s health with their reproductive organs alone. This has left large data gaps in researchers’ understanding of nearly every other health condition, including cancer, heart disease and dementia.
The missing pieces of menopause science
In 2016, the US National Institutes of Health (NIH) instituted a policy to bridge this data gap. The policy mandated that NIH-funded preclinical and clinical research include sex as a biological variable. Similar policies and mandates emerged around the same time in the European Union and Canada. The spotlight, supported by increases in funding, has led to surprising — even transformational — changes in scientists’ understanding of women’s health.
One approach has been to separate data by sex, through a method known as disaggregation. Rabin and other researchers have applied this method to long-standing databases that track the health and disease of large groups of participants to reveal important differences that researchers can then dig into more deeply to understand the underlying mechanisms.
But disaggregating data is not enough. Although analysing large data sets can reveal the existence of health differences by sex, it alone cannot explain why women’s health differs from that of men, says Cara Tannenbaum, a physician at the University of Montreal in Canada, who has long studied gender and health. (This article uses ‘men’ and ‘women’ to reflect terms used by the research community and in the literature). Tannenbaum sees these differences as a starting point for scientific enquiry, one that should be followed by an exploration of the underlying reasons for sex differences. Disaggregating data, she says, “is the beginning of the story”.
Global differences
Gathering sufficient data specifically on female lab animals and women has been a challenge. Analyses of cell-culture-based studies have found that most such research fails to specify the sex of cell lines and, when it is reported, more than 70% study only male cells. A 2019 review highlighted that research using animal models had overwhelmingly studied only males1.

Neuroscientist Jennifer Rabin (left) administers neuropsychological tests for a study on women’s brain health.Credit: Doug Nicholson/Sunnybrook
Clinical research has a similar dearth of data from women. In 1977, the US Food and Drug Administration (FDA) issued guidelines that excluded women “with childbearing potential” from phase I and phase II clinical trials. This recommendation followed the thalidomide tragedy, in which a drug for morning sickness was not adequately tested in women and caused congenital disabilities in the children of individuals who took it during pregnancy. The situation began to change in 1993, when the NIH Revitalization Act was put in place to increase racial and gender diversity in clinical trials.
The 2016 NIH policy that sex be included in study designs as a biological variable was a further step towards inclusion — and an important signal to the broader research community. “It made it more palatable and acceptable” to discuss the topic, Tannenbaum says, and “sends a very strong signal that research is not rigorous or ethical unless we look at sex and gender”.
Although the NIH policies applied only to the United States, they had important ripple effects on the understanding of the health of women, globally. In a 2024 study2, public-policy researcher Vedavati Patwardhan at the University of Washington in Seattle and her colleagues looked at how, between 1990 and 2021, disease patterns varied globally between male and female participants aged 10 years and older across their lifespans.
The team used data from the Global Burden of Disease study, a project launched by the World Bank in the 1990s to understand how death and disease affect people in 204 countries and territories. The team found that men were more likely to die sooner than women from 13 of the top 20 causes of disease.
But the researchers also found that female participants experienced a greater toll from ailments that reduce health and well-being. In other words, although women lived longer, they did so with poorer health. In particular, women are more likely than men to have conditions such as depression, back pain and other musculoskeletal disorders. This difference in disease burden emerged between the ages of 10 and 24 and persisted across the entire lifespan in female participants. Men, however, tended to be generally healthier early in life, but conditions such as heart disease, lung cancer or kidney disease took a greater toll as they aged.
Why these variations exist is unclear. “The next step is for the research community to analyse why these differences may manifest in particular contexts,” Patwardhan says.
But doing so will be a challenge — many national health databases don’t include sex-disaggregated data, says Angela Chang, a public-health researcher at the University of Southern Denmark in Odense.
Around the world, it’s typical to record a person’s sex by ticking a box when they walk into a health-care facility. But that information doesn’t always make it into research efforts, says global-health researcher Sarah Hawkes at Monash University Malaysia, who is based in Kuala Lumpur. For example, when COVID-19 hit, Hawkes and Chang found it nearly impossible to understand whether the pandemic was affecting men and women differently.
To address this problem, Hawkes and her colleagues went on to develop the world’s largest sex-disaggregated database on COVID-19. They have found that only 140 of 206 countries reported disaggregated data on deaths, and only 18 had the same details about rates of COVID-19 testing3. In those 140 countries, the team found stark differences. Men were less likely to be tested and more likely to be hospitalized. Women were less likely to die: for every 10 women who lost their lives, 13 men died. “Men and women were having very different pandemic experiences,” Hawkes says.
But those differences were not reflected in how countries developed policies to respond to people’s health needs. In the 76 countries for which the researchers could find data, more than 90% “did not incorporate a sex or gender lens into their clinical guidelines”, Hawkes says.
Recognizing these trends can help to direct resources and to design policies to reduce disparities between men and women. But these analyses are possible only when high quality information is available. “That is something where we have a lot of work to do,” says Sanne Peters, an epidemiologist at University Medical Center Utrecht in the Netherlands. “Even if there are no sex differences, that’s also important to know,” she adds. “At the moment, we are always playing this card of, ‘oh, we can’t be sure’.”
Sex-sorted data save lives
Differences between sexes are not just limited to risk factors or rates of disease. Medications and treatments can work differently, too. Demonstrating whether interventions are equally effective in men and women is the first step towards meaningful change, Peters says.
Analyses of chemotherapies for cancer, for example, have shown that women tend to experience more severe side effects than do men (see ‘Sex differences in cancer treatments’). A few years ago, health-systems researcher Joseph Unger at the Fred Hutch Cancer Center in Seattle, Washington, began to explore whether newer cancer treatments, such as targeted therapies and immune treatments, had reduced this disparity. “No one had looked at whether these sex differences persisted in these modern treatments,” he says.
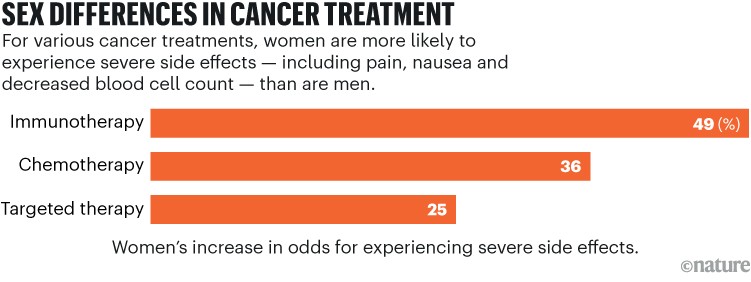
Source: ref. 4
Unger and his colleagues analysed data from 202 trials, representing more than 23,000 individuals4. The researchers excluded cancers that are more prevalent in one sex, such as ovarian, breast or prostate cancer. They found that women endured more severe and more frequent side effects from cancer treatment than did men who received the same treatments, especially from chemotherapy and therapies that target the immune system.
Some side effects of cancer treatment, such as nausea or pain, can be difficult to evaluate because they are subjective. But anaemia, low platelet counts and other blood biomarkers can be measured in lab tests. The researchers found that women had worse subjective and biomarker-based adverse events than did men. Although the mechanisms are unclear, the differences might be a result of how male and female hormones affect drug metabolism, the gut microbiome, inflammation or immunity. In other words, these differences are not simply because women are more sensitive to side effects or complain about them more, which had been commonly assumed in the research literature5 , Unger says. “It puts to rest the idea that these are simply differences in reporting side effects,” he says.
Because Unger’s study relied on data from clinical trials, all participants had equal access to cancer care. But outside the context of research studies, people’s sex and society’s gender norms can cause stark health-care disparities.
Researchers have known for decades that the risk of heart disease is gendered. Smoking, for example, disproportionately increases men’s risk of the disease, whereas women’s risk spikes at lower levels of excess weight or elevated blood glucose compared with men. Symptoms of heart disease appear 5–10 years earlier in men than they do in women, and men are more likely to experience coronary heart disease, whereas women are more likely to have strokes.
The earlier incidence in men is partly why heart disease has long been viewed as a male disease, according to Peters. When women have heart attacks, they typically experience a greater number and variety of symptoms than men do. These disparities have led to the prevalent belief that women’s set of symptoms for heart attacks are different — a misconception that delays diagnosis and care and ultimately harms women, Peters says. Women’s heart attacks are more likely to be misdiagnosed, she says, and women receive guideline-directed treatments less often than do men, which leads to a higher death toll from heart disease in women.
When researchers disaggregate data, the differences — and similarities — they uncover can be helpful in achieving more equitable health outcomes. In a 2020 meta-analysis, Peters and her colleagues reported that the top symptoms of heart attacks — chest pain, excessive sweating, shortness of breath and arm or shoulder pain — are the same in men and women6. However, women also seem to experience other symptoms, such as dizziness or nausea or vomiting, more often than men do. Recognizing this can help to reduce misdiagnoses, Peters says.

Women’s health specialist Cara Tannenbaum says that sex- and gender-disaggregated data are crucial for more equitable health care and better outcomes for women.Credit: Morteza Momenirad
In 2014, as part of an effort to address the disparities in treatment of heart disease, researchers conducted a meta-analysis of more than 68,000 individuals and found that compared with men, women were 48% more likely to die in hospital after a heart attack7.
To improve outcomes for women, researchers at the Cleveland Clinic in Ohio implemented a standard four-step protocol. This involved immediately sending anyone who arrived with certain heart attack symptoms to a specialized lab for the insertion of a balloon catheter.
When they analysed five years of data after they implemented the protocol, researchers reported that there were no significant differences in hospital deaths or rates of serious adverse events between men and women. Women’s chances of dying in hospital after certain kinds of heart attack decreased by more than half8.
These life-saving changes to treatment were the direct result of researchers disaggregating data and finding differences in outcomes, Tannenbaum says. If they had not done so, she says, “they never would’ve known that women truly were being disadvantaged by gendered processes”.
Creating a disaggregated future
Policy changes mean that more women now participate in clinical research and there has been a boost in the use of female cell lines and animals in preclinical studies. But sorting and analysing biomedical data by sex is still not routine, says neurobiologist Bronwyn Graham at the University of New South Wales in Sydney, Australia. “If you’re not disaggregating that data, then the inclusion of females alone doesn’t take us much further,” she says.

Neurobiologist Bronwyn Graham speaks about sex biases in research and health care at the Festival of Dangerous Ideas in Sydney in 2024.Credit: Maria Boyadgis
When Peters started studying heart disease, she began by reviewing the literature for studies that reported sex-specific data. She recalls excluding hundreds of studies because they did not disaggregate data. In hindsight, she says, that number “should have been my biggest finding”. Another challenge, according to Hawkes, is that databases and studies rarely distinguish between sex and gender. As a result, intersex people and those who are transgender or from other gender minorities are often not captured in databases or are left out of statistical analyses because their numbers are too small.
Health differences between men and women are the result of both biological differences as well as social norms around gendered behaviours, Hawkes says, so both sex and gender data are necessary for a complete picture. Unhealthy ideas of masculinity, for example, might underlie the greater risk of high blood pressure in men, as well as higher rates of death in road accidents. And social norms that can prevent women from leaving their homes unaccompanied can restrict access to health care and even prevent healthy behaviours such as taking a walk. Removing gender from the scientific lexicon makes the social and cultural factors that shape people’s health invisible, she explains. “Gender is important to the health of absolutely everybody,” Hawkes says.
Despite studies that show the value of incorporating sex and gender in scientific research, data disaggregation is still uncommon, researchers say. But it is a valuable tool both to identify where differences exist, and to direct resources or shape public-health policies to decrease health inequities, Hawkes explains. “If you don’t disaggregate your data, you’re assuming all human bodies are the same,” she says. “You’ll never be able to figure out who’s benefiting and who is being left behind.”



