Skin cells can be turned into bioinks, which can then be 3D printed to form tissues that emulate skin.Credit: Wake Forest Institute for Regenerative Medicine
The woman’s burns are severe, essentially eradicating the skin from more than half of her body. Every layer of this complex tissue must therefore be replaced with grafts. These need to be harvested gradually from intact parts of the body, expanded in a laboratory, and then transplanted to cover the injured sites over the course of several months — assuming sufficient healthy tissue is available.
Researchers such as Anthony Atala, a specialist in regenerative medicine at the Wake Forest University School of Medicine in Winston-Salem, North Carolina, have a different vision for burn treatment, in which grafts are made to order in the lab. Clinicians already use engineered single-layered epidermal grafts to protect tissue while it heals, but this is just a temporary measure to prevent infection during recovery. By contrast, Atala aims to convert skin cells taken from patients into ‘bioinks’, which can then be 3D printed into donor-matched, multilayered tissues that replicate core functions of natural skin. “Why don’t we engineer skin that is full thickness, so you can actually use that as a permanent graft — just like you would a patient’s own graft?” he asks. In 2023, he and his colleagues took an important step towards this goal, showing that they could print three-layered human skin grafts that included pigment, vasculature and even hair follicles. When this approach was tested in pigs, the resulting grafts efficiently integrated themselves into the surrounding skin1.
Nature Outlook: Skin
This is just one way to engineer tissue grafts that replicate key features of skin. Several groups have achieved promising clinical results by using conventional cell-culture methods to produce biologically faithful skin substitutes. This work is also fuelling the development of improved skin models for toxicology and disease research, which could reduce the need for animal experiments and deliver more-informative predictions. “It’s just getting harder and harder to ignore the fact that we are not mice,” says Pankaj Karande, a chemical engineer at the Rensselaer Polytechnic Institute in Troy, New York.
As a flat, neatly layered tissue that lacks the architectural sophistication of the kidney or lung, skin is an obvious and appealing target for tissue engineering efforts. But skin still exhibits remarkable cellular-scale complexity — and researchers underestimate that at their peril. “We’re transplanting an organ,” says Angela Gibson, a surgeon specializing in burn treatment at the University of Wisconsin–Madison. “The challenge is, we have to create an organ.”
A layered problem
When people think about skin, they usually focus on the epidermis. This outer layer of tightly interconnected keratinocyte cells is a protective barrier against infection and injury, and the point of contact for cosmetics, topical medications and kisses. It is reasonably straightforward to culture keratinocytes into thin epidermal layers, and engineered epidermis is now commercially available for both clinical and research applications.
But the epidermis is just the skin’s outermost shell. Many of the skin’s essential physiological functions occur in the thick, underlying dermal layer, which supports and sustains the epidermis. As a result, says Gibson, monolayer epidermal cultures are “super-fragile”. Researchers have therefore spent the past few decades developing models that couple a keratinocyte-based epidermal layer with a dermal layer comprising cells known as fibroblasts. These cells can be readily harvested from a person, multiplied in culture and then assembled into two-layered models that are genetically and immunologically matched to that person’s skin.
Diego Velasco, a bioengineer at Carlos III University of Madrid, favours an approach in which both layers are constructed simultaneously. His team creates the dermis by embedding fibroblasts in a gel that incorporates fibrin — a protein with a crucial role in clotting and tissue repair — and then layering keratinocytes atop the gel. Velasco’s group then exposes the topmost cell layer to the air while leaving the rest of the assembly submerged in liquid culture medium, which triggers the formation of a mature epidermal layer that includes the tough outer surface of skin, the stratum corneum. The dermis takes longer to mature than this layer does, and the fibrin-gel matrix is an important factor. “We use fibrin because, in the end, we are mimicking the wound-healing phase,” Velasco says.
Véronique Moulin, a surgeon at Laval University in Quebec, Canada, and her colleagues prefer to let the cells do most of the work. For the team’s ‘self-assembled skin substitute’ (SASS), fibroblasts and keratinocytes are cultured in separate stages. Initially, the fibroblasts are raised under conditions that allow them to produce their own matrix and form functional dermal tissue without needing any synthetic gel. Once the dermal layer is assembled, keratinocytes are seeded on top to initiate formation of the epidermis. “The problem is that it is a long-time process — we have to wait two months,” says Moulin. That’s about twice as long as Velasco’s approach takes.
With a robust culture and assembly process in place, epidermal–dermal assemblies can be readily produced that replicate key histological, molecular and mechanical features of human skin. “I would say, anybody who has reasonable experience in culture and a good standard operating procedure — and preferably can visit a lab that does it — can do it,” says Sue Gibbs, a cell biologist at Amsterdam University Medical Center in the Netherlands. But these models can still fall short of the real thing, depending on the extent of the tissue repair or the desired complexity of the in vitro model.
For example, many groups are looking to incorporate vasculature into the dermal layer. “This is actually the most important thing for wound healing,” says Agnes Klar, a tissue engineer at the University of Zurich in Switzerland. There is an important third layer beneath the dermis, the hypodermis, which produces metabolic factors and signalling molecules that are essential for sustaining the skin. Klar’s team is one of a handful that have succeeded in cultivating three-layered skin substitutes that include a working vasculature2. Klar says it remains challenging, however, to select culture conditions and media that sustain all these cell types while preserving their identity.
Skin also contains nerves, pigment-providing melanocytes, immune cells, sweat glands and hair follicles — all of which are needed for the organ to perform its essential duties. “Hair follicles really provide a house for the stem cells,” says Karande. “They become the agents to help with wound healing.” And as the number of cells involved multiplies, so does the complexity of the tissue-engineering task.
3D bioprinting can help. Karande has worked on this approach for more than 15 years, and notes that it was initially a struggle to transform cells into bioinks that can go through printers originally designed for use with rugged, heat-tolerant polymers. But he, Atala and others have shown that the approach can enable precise placement of many cell types in a complex tissue. “We started out just doing a few cell types, and we then just kept increasing the number of cells that we could isolate and expand,” says Atala, whose 2023 model incorporated six donor-derived cell types1.
Grafts, made to order
Numerous clinical scenarios would benefit from physiologically faithful skin substitutes — for example, to address the persistent ulcers that can form in people with poorly managed diabetes. But Gibson and others see the treatment of severe burns as the area with the greatest short-term opportunities.
Current treatment entails applying ‘autografts’, collected from the patient’s own body; even small patches of skin can be stretched slowly, over time, to produce larger grafts. But the graft has to match the thickness of the injured tissue, and if the burns are too extensive, the options are dire. “If you are burned over 50% of your body, it’s always difficult to find enough non-burnt skin to graft,” says Moulin. “Usually, patients remain at the burn unit for several months.”
Current tissue substitutes are limited. Vericel, a company in Cambridge, Massachusetts, offers Epicel grafts — patient-derived epidermal sheets that can repair superficial burns. Epicel can accelerate autograft-mediated repair of full-thickness burns in which the dermal layer is seriously damaged, but is not an adequate skin substitute on its own. Gibson likens these deep burns to a lawn that has been mowed so heavily that the underlying dirt and roots are damaged; new sod will be needed for regrowth to happen. The wounds can be covered with human-tissue grafts, such as Apligraf, developed by Organogenesis in Canton, Massachusetts, that feature both dermal and epidermal layers. However, these are not donor-matched, and serve as only temporary coverings until autografts become available.
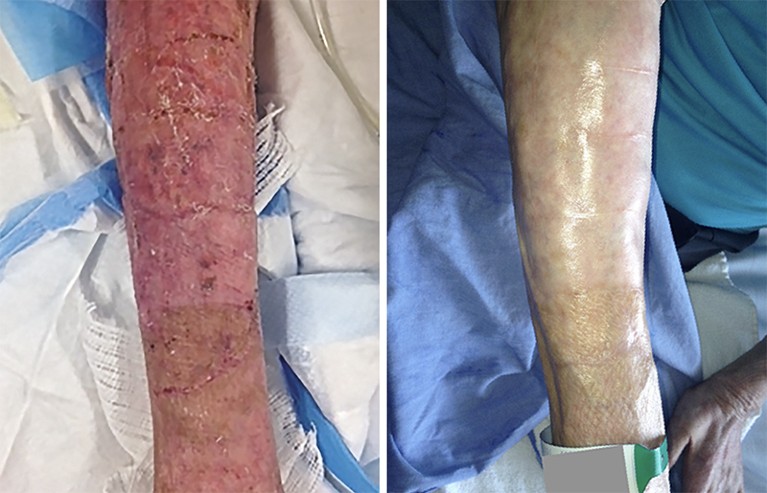
Severe burns (left) can be treated with ‘SASS’ grafts, which blend in with surviving skin (right).Credit: REF. 3
A few research groups have brought more-sophisticated, patient-derived dermal–epidermal grafts to the clinic. For example, SASS is now undergoing clinical testing in people in Canada with extensive, severe burns. In a 2018 study of 14 people with burns covering at least half of their body, the researchers showed that virtually every patch of lab-generated SASS tissue successfully integrated into surrounding skin, with an average success rate of 95% across all grafts3. Importantly, all of these grafts remained intact for up to eight years, with minimal scarring, and physical and mechanical properties similar to those of real skin. “The plastic surgeons are very happy, and the patients, too,” says Moulin, whose team is now in the middle of a larger trial with 52 people with burns.
A start-up company called Cutiss, in Schieren, Switzerland, is commercializing bilayered skin substitutes developed by Klar and her colleagues. In February, Cutiss reported positive interim results from two phase II clinical trials of its engineered denovoSkin grafts, treating children and adults after either severe burns or reconstructive surgery. The results have not yet been published, but Klar says that participants end up with “very good skin, with very little scarring”. A handful of people in the United States and Europe have obtained this treatment through compassionate-use exemptions, and the company is now embarking on a phase III European trial.
The time required to produce a graft remains a major bottleneck. For the SASS grafts, it is now feasible to generate roughly 1,000 square centimetres of engineered skin every two weeks — although Moulin notes that gearing up for that level of production takes two months. 3D bioprinting could speed things up. Atala says it takes about six weeks to get from a biopsy to sufficient cellular bioink to print a skin substitute, with another week required before implantation. Turning this into a stable and reproducible manufacturing process will take some effort, but Karande sees opportunities for scaling up. “If you can multiplex this, you don’t have to wait on one printer printing out a full-sized sheet of skin,” he says. “You could have ten printers do it at the same time.”
A gap remains between the sophistication that can be achieved with engineered skin in the lab and what is feasible in the clinic. This is partly the result of a conscious decision by clinical researchers, who have generally focused on relatively simple tissue constructs, to deliver regenerative interventions as quickly as possible. Take the extreme example of a person with burns affecting 95% of their body. In such a case, “the most important thing is that the surgeon wants to close the wound”, says Thomas Biedermann, a tissue engineer at the University of Zurich who is coordinating the effort by Klar and her colleagues. But the team has big plans for future clinical programmes, including triple-layered grafts that incorporate the hypodermal layer along with blood vessels, and pigmentation that matches the donor’s skin.
Clinical development is especially difficult in this realm because of the inherent challenges of manufacturing safe and effective cell-based therapies reproducibly, particularly as the interventions become more complex. For example, Klar and Biedermann are grappling with how to convince regulators to allow grafts that contain melanocytes, which give skin its natural coloration and protect against damage from the Sun. “Everybody’s afraid of getting melanoma,” says Klar. “And with vascularization, there could be even faster melanoma if you put them both together.” Such safety concerns make the upgrading of engineered graft technologies a slow and deliberate process.
An advanced culture
The challenges of clinical development have spurred some researchers to focus on in vitro work. Gibbs spent more than a decade developing engineered skin substitutes for use in people with long-term wounds through an Amsterdam-based start-up company called A-Skin. When funding ran dry, she shifted to developing next-generation lab models.
In a research setting, tissue engineers have more flexibility to tinker with their skin constructs, incorporating the functional components most relevant to the scientific question and ignoring or streamlining the rest. “You make them as simple as possible and as complex as required,” says Gibbs. For example, introducing fully functional hair follicles is difficult, but Velasco’s team has identified a potential alternative. Instead, the researchers are working with gel-encapsulated follicle precursor cells known as dermal papilla cells, which allows them to get a sense of how chemical exposures affect early follicular development and hair growth.
More from Nature Outlooks
Researchers then have the option to graft these tissues onto animals. Moulin, for example, is performing rodent studies with the SASS model to learn more about the scarring process. But multilayered skin models can also be incorporated into ‘body-on-a-chip’ systems, in which engineered versions of multiple tissues are cultivated in a single device, with fluid-filled channels connecting the various compartments. Such systems allow tight control over a simplified version of the body, and can be combined with sensors that enable real-time monitoring of the response to disease or to treatment.
Gibbs is developing a chip in which multilayered skin samples are linked to models of the lymph nodes through the use of engineered, cell-lined channels that mimic the vasculature and lymphatic systems. One goal is to use this model to study melanoma and the mechanisms by which the tumours progress and spread, or metastasize. But she thinks that a similar body-on-a-chip design could be broadly useful for exploring a range of dermatological conditions. “Nearly all skin disease is immune-mediated,” she says. “You need an immune system in your models, and you can’t use the animal models because they’re not representative enough of human reactions.”
These two domains of skin tissue engineering — growing next-generation grafts for wound repair and engineering sophisticated models for research — are complementary. Many groups focused on clinical applications also see clear value for their models in the lab, and time spent developing and optimizing these models in vitro and in vivo could hasten their therapeutic use.
The ability to rapidly assemble elaborate, patient-specific grafts in large quantities might still be years away, but the wheels are in motion. Atala’s team is now working on manufacturing processes that are more reproducible and efficient, as a prelude to testing its 3D-printed tissues in humans. Once a successful approach is established, the idea of building up a tissue factory is no longer so far-fetched. “We are doing this on everything from semiconductor chips to car parts,” says Karande. “So why can’t it be done in biology?”