Golanov, E. V., Yamamoto, S. & Reis, D. J. Spontaneous waves of cerebral blood flow associated with a pattern of electrocortical activity. Am. J. Physiol. 266, R204–R214 (1994).
Hudetz, A. G., Roman, R. J. & Harder, D. R. Spontaneous flow oscillations in the cerebral cortex during acute changes in mean arterial pressure. J. Cereb. Blood Flow Metab. 12, 491–499 (1992). This paper systematically investigates spontaneous oscillations in the cerebral cortex using laser Doppler flow imaging during physiological perturbations in mean arterial pressure.
Vern, B. A., Schuette, W. H., Leheta, B., Juel, V. C. & Radulovacki, M. Low-frequency oscillations of cortical oxidative metabolism in waking and sleep. J. Cereb. Blood Flow Metab. 8, 215–226 (1988).
Berger, H. Über das Elektrenkephalogramm des Menschen. Archiv für Psychiatrie und Nervenkrankheiten 87, 527–570 (1929).
Friston, K. Functional and effective connectivity in neuroimaging: a synthesis. Hum. Brain Mapp. 2, 56–78 (1994). This paper provides a conceptual framework of functional and effective connectivity in neuroimaging as approaches for understanding how different brain regions interact and influence one another.
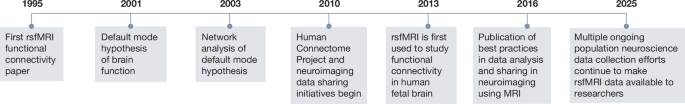
Biswal, B., Yetkin, F. Z., Haughton, V. M. & Hyde, J. S. Functional connectivity in the motor cortex of resting human brain using echo-planar MRI. Magn. Reson. Med. 34, 537–541 (1995).
Biswal, B., Hudetz, A. G., Yetkin, F. Z., Haughton, V. M. & Hyde, J. S. Hypercapnia reversibly suppresses low-frequency fluctuations in the human motor cortex during rest using echo–planar MRI. J. Cereb. Blood Flow Metab. 17, 301–308 (1997). This paper demonstrates using fMRI that hypercapnia reversibly suppresses low-frequency fluctuations and functional connectivity in the motor cortex at rest.
Mason, M. F. et al. Wandering minds: the default network and stimulus-independent thought. Science 315, 393–395 (2007).
Murphy, K., Birn, R. M. & Bandettini, P. A. Resting-state fMRI confounds and cleanup. Neuroimage 80, 349–359 (2013).
Snyder, A. Z. & Raichle, M. E. A brief history of the resting state: the Washington University perspective. Neuroimage 62, 902–910 (2012).
Bolt, T., Anderson, M. L. & Uddin, L. Q. Beyond the evoked/intrinsic neural process dichotomy. Netw. Neurosci. https://doi.org/10.1162/NETN_a_00028 (2018). This paper proposes an integrated view of brain activity that emphasizes the continuous interplay between externally driven and internally generated neural dynamics.
Thomason, M. E. et al. Cross-hemispheric functional connectivity in the human fetal brain. Sci. Transl. Med. 5, 173ra24 (2013).
Shen, H. H. Resting-state connectivity. Proc. Natl Acad. Sci. USA 112, 14115–14116 (2015).
Vincent, J. L. et al. Intrinsic functional architecture in the anaesthetized monkey brain. Nature 447, 83–86 (2007). This study used rsfMRI in anaesthetized monkeys to demonstrate the intrinsic functional architecture of the brain even in the absence of conscious awareness.
Raichle, M. E. et al. A default mode of brain function. Proc. Natl Acad. Sci. USA 98, 676–682 (2001).
Greicius, M. D., Krasnow, B., Reiss, A. L. & Menon, V. Functional connectivity in the resting brain: a network analysis of the default mode hypothesis. Proc. Natl Acad. Sci. USA 100, 253–258 (2003).
Seeley, W. W. et al. Dissociable intrinsic connectivity networks for salience processing and executive control. J. Neurosci. 27, 2349–2356 (2007).
Damoiseaux, J. S. et al. Consistent resting-state networks across healthy subjects. Proc. Natl Acad. Sci. USA 103, 13848–13853 (2006).
De Luca, M., Beckmann, C. F., De Stefano, N., Matthews, P. M. & Smith, S. M. fMRI resting state networks define distinct modes of long-distance interactions in the human brain. Neuroimage 29, 1359–1367 (2006).
Power, J. D. et al. Functional network organization of the human brain. Neuron 72, 665–678 (2011).
Yeo, B. T. T. et al. The organization of the human cerebral cortex estimated by intrinsic functional connectivity. J. Neurophysiol. 106, 1125–1165 (2011).
Dosenbach, N. U., Fair, D. A., Cohen, A. L., Schlaggar, B. L. & Petersen, S. E. A dual-networks architecture of top-down control. Trends Cogn. Sci. 12, 99–105 (2008).
Uddin, L. Q., Thomas Yeo, B. T. & Spreng, R. Towards a universal taxonomy of macro-scale functional human brain networks. Brain Topogr. 32, 926–942 (2019).
Smith, S. M. et al. Correspondence of the brain’s functional architecture during activation and rest. Proc. Natl Acad. Sci. USA. 106, 13040–13045 (2009).
Fox, M. D. et al. The human brain is intrinsically organized into dynamic, anticorrelated functional networks. Proc. Natl Acad. Sci. USA. 102, 9673–9678 (2005). This paper demonstrates that the human brain is intrinsically organized into dynamic, anticorrelated functional networks, with the default and task-positive networks displaying opposing activity patterns during rest, suggesting a fundamental principle of brain organization.
Fransson, P. Spontaneous low-frequency BOLD signal fluctuations: an fMRI investigation of the resting-state default mode of brain function hypothesis. Hum. Brain Mapp. 26, 15–29 (2005).
Hampson, M., Driesen, N. R., Skudlarski, P., Gore, J. C. & Constable, R. T. Brain connectivity related to working memory performance. J. Neurosci. 26, 13338–13343 (2006).
Kelly, A. M., Uddin, L. Q., Biswal, B. B., Castellanos, F. X. & Milham, M. P. Competition between functional brain networks mediates behavioral variability. Neuroimage 39, 527–537 (2008).
Chang, S. E., Lenartowicz, A., Hellemann, G. S., Uddin, L. Q. & Bearden, C. E. Variability in cognitive task performance in early adolescence is associated with stronger between-network anticorrelation and future attention problems. Biol. Psychiatry Glob. Open Sci. 3, 948–957 (2023). This study demonstrates that variability in cognitive task performance during early adolescence is linked to stronger anticorrelations between brain networks, and that these brain network interactions predict attention-related issues.
Marek, S. et al. Reproducible brain-wide association studies require thousands of individuals. Nature 603, 654–660 (2022).
Cosío-Guirado, R. et al. A comprehensive systematic review of fMRI studies on brain connectivity in healthy children and adolescents: current insights and future directions. Dev. Cogn. Neurosci. 69, 101438 (2024).
Uddin, L. Q. Bring the noise: reconceptualizing spontaneous neural activity. Trends Cogn. Sci. 24, 734–746 (2020).
Finn, E. S. et al. Functional connectome fingerprinting: identifying individuals using patterns of brain connectivity. Nat. Neurosci. 18, 1664–1671 (2015).
Zhang, J. et al. What have we really learned from functional connectivity in clinical populations? Neuroimage 242, 118466 (2021).
Deco, G., Jirsa, V. K. & McIntosh, A. R. Resting brains never rest: computational insights into potential cognitive architectures. Trends Neurosci. 36, 268–274 (2013).
Uddin, L. Q., Castellanos, F. X. & Menon, V. Resting state functional brain connectivity in child and adolescent psychiatry: where are we now?. Neuropsychopharmacology 50, 196–200 (2025).
Mitra, A., Snyder, A. Z., Blazey, T. & Raichle, M. E. Lag threads organize the brain’s intrinsic activity. Proc. Natl Acad. Sci. USA 112, E2235–E2244 (2015).
Matsui, T., Murakami, T. & Ohki, K. Transient neuronal coactivations embedded in globally propagating waves underlie resting-state functional connectivity. Proc. Natl Acad. Sci. USA 113, 6556–6561 (2016).
Abbas, A. et al. Quasi-periodic patterns contribute to functional connectivity in the brain. Neuroimage 191, 193–204 (2019). This study demonstrates that quasi-periodic patterns are a key contributor to functional connectivity in the brain and provide a dynamic mechanism that helps explain the variability and organization of ICNs.
Gu, Y. et al. Brain activity fluctuations propagate as waves traversing the cortical hierarchy. Cereb. Cortex 31, 3986–4005 (2021).
Raut, R. V. et al. Global waves synchronize the brain’s functional systems with fluctuating arousal. Sci. Adv. 7, eabf2709 (2021).
Bolt, T. et al. A parsimonious description of global functional brain organization in three spatiotemporal patterns. Nat. Neurosci. 25, 1093–1103 (2022).
Greene, A. S., Gao, S., Scheinost, D. & Constable, R. T. Task-induced brain state manipulation improves prediction of individual traits. Nat. Commun. 9, 2807 (2018).
Matthews, P. M. & Jezzard, P. Functional magnetic resonance imaging. J. Neurol. Neurosurg. Psychiatry 75, 6–12 (2004).
Glover, G. H. Overview of functional magnetic resonance imaging. Neurosurg. Clin. N. Am. 22, 133–139 (2011).
Horovitz, S. G. et al. Decoupling of the brain’s default mode network during deep sleep. Proc. Natl Acad. Sci. USA 106, 11376–11381 (2009). This study demonstrates that functional connectivity within the default mode network is significantly reduced during slow-wave sleep, suggesting that deep sleep disrupts the coordinated activity of this network.
Kiviniemi, V. et al. Slow vasomotor fluctuation in fMRI of anesthetized child brain. Magn. Reson. Med. 44, 373–378 (2000).
Peltier, S. J. et al. Functional connectivity changes with concentration of sevoflurane anesthesia. Neuroreport 16, 285–288 (2005).
Lu, H. et al. Rat brains also have a default mode network. Proc. Natl Acad. Sci. USA 109, 3979–3984 (2012).
Gozzi, A., Schwarz, A., Crestan, V. & Bifone, A. Drug-anaesthetic interaction in phMRI: the case of the psychotomimetic agent phencyclidine. Magn. Reson. Imaging 26, 999–1006 (2008).
Logothetis, N. K., Pauls, J., Augath, M., Trinath, T. & Oeltermann, A. Neurophysiological investigation of the basis of the fMRI signal. Nature 412, 150–157 (2001).
Scholvinck, M. L., Maier, A., Ye, F. Q., Duyn, J. H. & Leopold, D. A. Neural basis of global resting-state fMRI activity. Proc. Natl Acad. Sci. USA 107, 10238–10243 (2010).
Schulz, K. et al. Simultaneous BOLD fMRI and fiber-optic calcium recording in rat neocortex. Nat. Methods 9, 597–602 (2012).
Chen, T.-W. et al. Ultrasensitive fluorescent proteins for imaging neuronal activity. Nature 499, 295–300 (2013).
Marvin, J. S. et al. An optimized fluorescent probe for visualizing glutamate neurotransmission. Nat. Methods 10, 162–170 (2013).
Sun, F. et al. A genetically encoded fluorescent sensor enables rapid and specific detection of dopamine in flies, fish, and mice. Cell 174, 481–496.e19 (2018).
Patriarchi, T. et al. Ultrafast neuronal imaging of dopamine dynamics with designed genetically encoded sensors. Science 360, eaat4422 (2018).
Feng, J. et al. A genetically encoded fluorescent sensor for rapid and specific in vivo detection of norepinephrine. Neuron 102, 745–761.e8 (2019).
Gong, Y. et al. High-speed recording of neural spikes in awake mice and flies with a fluorescent voltage sensor. Science 350, 1361–1366 (2015).
Tian, L. et al. Imaging neural activity in worms, flies and mice with improved GCaMP calcium indicators. Nat. Methods 6, 875–881 (2009).
Mitra, A. et al. Spontaneous infra-slow brain activity has unique spatiotemporal dynamics and laminar structure. Neuron 98, 297–305.e6 (2018). This paper demonstrates that infra-slow brain activity has distinct spatiotemporal dynamics and is organized in specific layers of the brain.
Gozzi, A. & Zerbi, V. Modeling brain dysconnectivity in rodents. Biol. Psychiatry 93, 419–429 (2023).
Drew, P. J. et al. Ultra-slow oscillations in fMRI and resting-state connectivity: neuronal and vascular contributions and technical confounds. Neuron 107, 782–804 (2020).
Cole, D. M., Smith, S. M. & Beckmann, C. F. Advances and pitfalls in the analysis and interpretation of resting-state FMRI data. Front. Syst. Neurosci. 4, 8 (2010).
Karunakaran, K., Wolfer, M. & Biswal, B. B. in Brain Network Dysfunction in Neuropsychiatric Illness (eds Diwadkar, V. A. & Eickhoff, S. B.) 45–74 (2021).
Uddin, L. Q. Mixed signals: on separating brain signal from noise. Trends Cogn. Sci. 21, 405–406 (2017).
Chen, J. E. et al. Resting-state ‘physiological networks’. Neuroimage 213, 116707 (2020). This paper introduces the concept of physiological networks and demonstrates that ICNs reflect patterns of physiological processes, such as heart rate and respiration, that are intertwined with neuronal activity and can influence interpretations of functional connectivity.
Bolt, T. et al. Autonomic physiological coupling of the global fMRI signal. Nat. Neurosci. https://doi.org/10.1038/s41593-025-01945-y (2025).
Power, J. D., Barnes, K. A., Snyder, A. Z., Schlaggar, B. L. & Petersen, S. E. Spurious but systematic correlations in functional connectivity MRI networks arise from subject motion. Neuroimage 59, 2142–2154 (2012).
Parkes, L., Fulcher, B., Yücel, M. & Fornito, A. An evaluation of the efficacy, reliability, and sensitivity of motion correction strategies for resting-state functional MRI. Neuroimage 171, 415–436 (2018).
Ciric, R. et al. Mitigating head motion artifact in functional connectivity MRI. Nat. Protoc. 13, 2801–2826 (2018).
Power, J. D., Plitt, M., Laumann, T. O. & Martin, A. Sources and implications of whole-brain fMRI signals in humans. Neuroimage 146, 609–625 (2017).
Li, J. et al. Topography and behavioral relevance of the global signal in the human brain. Sci. Rep. 9, 14286 (2019). This paper demonstrates that the global signal, which is often treated as noise, is spatially organized and linked to behavioural states, suggesting that it carries meaningful information about the brain.
Nomi, J. S. et al. Systematic cross-sectional age-associations in global fMRI signal topography. Imaging Neurosci. https://doi.org/10.1162/imag_a_00101 (2024).
Saad, Z. S. et al. Trouble at rest: how correlation patterns and group differences become distorted after global signal regression. Brain Connect. 2, 25–32 (2012).
Murphy, K. & Fox, M. D. Towards a consensus regarding global signal regression for resting state functional connectivity MRI. Neuroimage 154, 169–173 (2017). This paper discusses the ongoing debate about GSR and reviews the arguments for and against applying GSR.
Yan, C.-G., Wang, X.-D., Zuo, X.-N. & Zang, Y.-F. DPABI: Data Processing & Analysis for (Resting-State) Brain Imaging. Neuroinformatics 14, 339–351 (2016).
Whitfield-Gabrieli, S. & Nieto-Castanon, A. Conn: a functional connectivity toolbox for correlated and anticorrelated brain networks. Brain Connect. 2, 125–141 (2012).
Nichols, T. E. et al. Best practices in data analysis and sharing in neuroimaging using MRI. Nat. Neurosci. 20, 299–303 (2017).
Dafflon, J. et al. A guided multiverse study of neuroimaging analyses. Nat. Commun. 13, 3758 (2022).
Laumann, T. O. & Snyder, A. Z. Brain activity is not only for thinking. Curr. Opin. Behav. Sci. 40, 130–136 (2021).
Barttfeld, P. et al. Signature of consciousness in the dynamics of resting-state brain activity. Proc. Natl Acad. Sci. USA 112, 887–892 (2015).
Gonzalez-Castillo, J., Kam, J. W. Y., Hoy, C. W. & Bandettini, P. A. How to interpret resting-state fMRI: ask your participants. J. Neurosci. 41, 1130–1141 (2021).
Birn, R. M. et al. The influence of physiological noise correction on test–retest reliability of resting-state functional connectivity. Brain Connect. 4, 511–522 (2014).
Birn, R. M. et al. The effect of scan length on the reliability of resting-state fMRI connectivity estimates. Neuroimage 83, 550–558 (2013).
Calhoun, V. D., Miller, R., Pearlson, G. & Adalı, T. The chronnectome: time-varying connectivity networks as the next frontier in fMRI data discovery. Neuron 84, 262–274 (2014). This paper introduces addresses time-varying functional connectivity using fMRI and argues that studying these dynamic networks is important for understanding disorders and cognitive processes.
Hutchison, R. M. et al. Dynamic functional connectivity: promise, issues, and interpretations. Neuroimage 80, 360–378 (2013).
Specht, K. Current challenges in translational and clinical fMRI and future directions. Front. Psychiatry 10, 924 (2019).
Lurie, D. J. et al. Questions and controversies in the study of time-varying functional connectivity in resting fMRI. Netw. Neurosci. 4, 30–69 (2020).
Uddin, L. Q. Salience processing and insular cortical function and dysfunction. Nat. Rev. Neurosci. 16, 55–61 (2015).
Corbetta, M., Patel, G. & Shulman, G. L. The reorienting system of the human brain: from environment to theory of mind. Neuron 58, 306–324 (2008).
Uddin, L. Q. et al. Controversies and progress on standardization of large-scale brain network nomenclature. Netw. Neurosci. 7, 864–905 (2023).
Vanderwal, T., Eilbott, J. & Castellanos, F. X. Movies in the magnet: naturalistic paradigms in developmental functional neuroimaging. Dev. Cogn. Neurosci. 36, 100600 (2019).
Finn, E. S. Is it time to put rest to rest? Trends Cogn. Sci. 25, 1021–1032 (2021).
Makowski, C. et al. Leveraging the Adolescent Brain Cognitive Development Study to improve behavioral prediction from neuroimaging in smaller replication samples. Cereb. Cortex 34, bhae223 (2024).
Spisak, T., Bingel, U. & Wager, T. D. Multivariate BWAS can be replicable with moderate sample sizes. Nature 615, E4–E7 (2023).
Gratton, C., Nelson, S. M. & Gordon, E. M. Brain–behavior correlations: two paths toward reliability. Neuron 110, 1446–1449 (2022).
Ward, H. B. et al. Robust brain correlates of cognitive performance in psychosis and its prodrome. Biol. Psychiatry 97, 139–147 (2025).
Pagani, M., Gutierrez-Barragan, D., de Guzman, A. E., Xu, T. & Gozzi, A. Mapping and comparing fMRI connectivity networks across species. Commun. Biol. 6, 1238 (2023).
Ma, Z., Zhang, Q., Tu, W. & Zhang, N. Gaining insight into the neural basis of resting-state fMRI signal. Neuroimage 250, 118960 (2022).
Tsai, P.-J. et al. Converging structural and functional evidence for a rat salience network. Biol. Psychiatry 88, 867–878 (2020).
Lu, H. & Yang, Y. Leveraging large-scale brain networks in rats to understand neurological and psychiatric disorders in humans. Neuropsychopharmacology 50, 337–338 (2025).
Hadjiabadi, D. H. et al. Brain tumors disrupt the resting-state connectome. Neuroimage Clin. 18, 279–289 (2018).
Hyder, F. & Rothman, D. L. Advances in imaging brain metabolism. Annu. Rev. Biomed. Eng. 19, 485–515 (2017).
Herman, P., Sanganahalli, B. G., Blumenfeld, H., Rothman, D. L. & Hyder, F. Quantitative basis for neuroimaging of cortical laminae with calibrated functional MRI. Proc. Natl Acad. Sci. USA 110, 15115–15120 (2013).
Wise, R. G., Ide, K., Poulin, M. J. & Tracey, I. Resting fluctuations in arterial carbon dioxide induce significant low frequency variations in BOLD signal. Neuroimage 21, 1652–1664 (2004).
Gati, J. S., Menon, R. S., Ugurbil, K. & Rutt, B. K. Experimental determination of the BOLD field strength dependence in vessels and tissue. Magn. Reson. Med. 38, 296–302 (1997).
Bandettini, P. A. & Wong, E. C. A hypercapnia-based normalization method for improved spatial localization of human brain activation with fMRI. NMR Biomed. 10, 197–203 (1997).
Kannurpatti, S. S. & Biswal, B. B. Detection and scaling of task-induced fMRI-BOLD response using resting state fluctuations. Neuroimage 40, 1567–1574 (2008). This paper demonstrates the use of resting-state fluctuations in fMRI signals to detect and scale task-induced BOLD responses, which offers improved detection and interpretation of fMRI BOLD signals during cognitive tasks.
Liu, P. et al. Cerebrovascular reactivity mapping using intermittent breath modulation. Neuroimage 215, 116787 (2020).
Deco, G., Tononi, G., Boly, M. & Kringelbach, M. L. Rethinking segregation and integration: contributions of whole-brain modelling. Nat. Rev. Neurosci. 16, 430–439 (2015). This paper reviews the balance between segregation and integration in brain function through the lens of whole-brain modelling.
Pathak, A., Roy, D. & Banerjee, A. Whole-brain network models: from physics to bedside. Front. Comput. Neurosci. 16, 866517 (2022).
Jirsa, V. K., Sporns, O., Breakspear, M., Deco, G. & McIntosh, A. R. Towards the virtual brain: network modeling of the intact and the damaged brain. Arch. Ital. Biol. 148, 189–205 (2010).
Deco, G. & Kringelbach, M. L. Great expectations: using whole-brain computational connectomics for understanding neuropsychiatric disorders. Neuron 84, 892–905 (2014).
Khosla, M., Jamison, K., Ngo, G. H., Kuceyeski, A. & Sabuncu, M. R. Machine learning in resting-state fMRI analysis. Magn. Reson. Imaging 64, 101–121 (2019).
Hong, S.-J. et al. Toward neurosubtypes in autism. Biol. Psychiatry 88, 111–128 (2020).
Uddin, L. Q., Dajani, D. R., Voorhies, W., Bednarz, H. & Kana, R. K. Progress and roadblocks in the search for brain-based biomarkers of autism and attention-deficit/hyperactivity disorder. Transl. Psychiatry 7, e1218 (2017).
Li, J. et al. Cross-ethnicity/race generalization failure of behavioral prediction from resting-state functional connectivity. Sci. Adv. 8, eabj1812 (2022).
Kopal, J., Uddin, L. Q. & Bzdok, D. The end game: respecting major sources of population diversity. Nat. Methods 20, 1122–1128 (2023).
Schumann, G. et al. Precision medicine and global mental health. Lancet Glob. Health 7, e32 (2019).
Gordon, E. M. et al. Precision functional mapping of individual human brains. Neuron 95, 791–807.e7 (2017). This paper presents a method for precision functional mapping of individual human brains and highlights how functional brain networks vary significantly across individuals.
Poldrack, R. A. et al. Long-term neural and physiological phenotyping of a single human. Nat. Commun. 6, 8885 (2015).
Yip, S. W. & Konova, A. B. Densely sampled neuroimaging for maximizing clinical insight in psychiatric and addiction disorders. Neuropsychopharmacology 47, 395–396 (2022).
Downar, J., Siddiqi, S. H., Mitra, A., Williams, N. & Liston, C. Mechanisms of action of TMS in the treatment of depression. Curr. Top. Behav. Neurosci. 66, 233–277 (2024).
Mitra, A., Raichle, M. E., Geoly, A. D., Kratter, I. H. & Williams, N. R. Targeted neurostimulation reverses a spatiotemporal biomarker of treatment-resistant depression. Proc. Natl Acad. Sci. USA 120, e2218958120 (2023).
Siddiqi, S. H. et al. Brain stimulation and brain lesions converge on common causal circuits in neuropsychiatric disease. Nat. Hum. Behav. 5, 1707–1716 (2021).
Cash, R. F. H. & Zalesky, A. Personalized and circuit-based transcranial magnetic stimulation: evidence, controversies, and opportunities. Biol. Psychiatry 95, 510–522 (2024).
Schaefer, A. et al. Local-global parcellation of the human cerebral cortex from intrinsic functional connectivity MRI. Cereb. Cortex 28, 3095–3114 (2018).
Gordon, E. M. et al. Generation and evaluation of a cortical area parcellation from resting-state correlations. Cereb. Cortex 26, 288–303 (2016).
Kong, R. et al. A network correspondence toolbox for quantitative evaluation of novel neuroimaging results. Nat. Commun. 16, 2930 (2025).
West, K. L. et al. BOLD hemodynamic response function changes significantly with healthy aging. Neuroimage 188, 198–207 (2019).
Lindquist, M. A., Meng Loh, J., Atlas, L. Y. & Wager, T. D. Modeling the hemodynamic response function in fMRI: efficiency, bias and mis-modeling. Neuroimage 45, S187–S198 (2009).
Yan, W., Rangaprakash, D. & Deshpande, G. Estimated hemodynamic response function parameters obtained from resting state BOLD fMRI signals in subjects with autism spectrum disorder and matched healthy subjects. Data Brief. 19, 1305–1309 (2018).
Bandettini, P. A. Neuronal or hemodynamic? Grappling with the functional MRI signal. Brain Connect. 4, 487–498 (2014).
Zang, Y., Jiang, T., Lu, Y., He, Y. & Tian, L. Regional homogeneity approach to fMRI data analysis. Neuroimage 22, 394–400 (2004).
Jiang, L. et al. Toward neurobiological characterization of functional homogeneity in the human cortex: regional variation, morphological association and functional covariance network organization. Brain Struct. Funct. 220, 2485–2507 (2015).
Jiang, L. & Zuo, X.-N. Regional homogeneity: a multimodal, multiscale neuroimaging marker of the human connectome. Neuroscientist 22, 486–505 (2016).
Beckmann, C. F., DeLuca, M., Devlin, J. T. & Smith, S. M. Investigations into resting-state connectivity using independent component analysis. Philos. Trans. R. Soc. B 360, 1001–1013 (2005). This paper used ICA applied to rsfMRI data to demonstrate the presence of multiple spatially independent brain networks without requiring predefined regions of interest.
Huettel, S. A. Functional Magnetic Resonance Imaging (Sinauer Associates, 2004). This textbook introduces the physiological basis of fMRI and data analysis methods.
Bullmore, E. & Sporns, O. Complex brain networks: graph theoretical analysis of structural and functional systems. Nat. Rev. Neurosci. 10, 186–198 (2009).
Bassett, D. S. & Sporns, O. Network neuroscience. Nat. Neurosci. 20, 353–364 (2017).
Rubinov, M. & Sporns, O. Complex network measures of brain connectivity: uses and interpretations. Neuroimage 52, 1059–1069 (2010).
van den Heuvel, M. P. & Hulshoff Pol, H. E. Exploring the brain network: a review on resting-state fMRI functional connectivity. Eur. Neuropsychopharmacol. 20, 519–534 (2010). This paper reviews the use of rsfMRI to describe the intrinsic network organization of the brain in healthy individuals and in those with psychiatric and neurological disorders.
van den Heuvel, M. P., Stam, C. J., Boersma, M. & Hulshoff Pol, H. E. Small-world and scale-free organization of voxel-based resting-state functional connectivity in the human brain. Neuroimage 43, 528–539 (2008).
Fornito, A., Zalesky, A. & Breakspear, M. Graph analysis of the human connectome: promise, progress, and pitfalls. Neuroimage 80, 426–444 (2013).
Canario, E., Chen, D. & Biswal, B. A review of resting-state fMRI and its use to examine psychiatric disorders. Psychoradiology 1, 42–53 (2021).
Biswal, B. B. et al. Toward discovery science of human brain function. Proc. Natl Acad. Sci. USA 107, 4734–4739 (2010).
Van Essen, D. C. et al. The WU-Minn Human Connectome Project: an overview. Neuroimage 80, 62–79 (2013).
Casey, B. J. et al. The Adolescent Brain Cognitive Development (ABCD) study: imaging acquisition across 21 sites. Dev. Cogn. Neurosci. 32, 43–54 (2018).
Di Martino, A. et al. The autism brain imaging data exchange: towards a large-scale evaluation of the intrinsic brain architecture in autism. Mol. Psychiatry 19, 659–667 (2014). This paper describes a grassroots data sharing effort involving pooling rsfMRI data from multiple sites performing studies of autism spectrum disorder.
HD-200 Consortium. The ADHD-200 Consortium: a model to advance the translational potential of neuroimaging in clinical neuroscience. Front. Syst. Neurosci. 6, 62 (2012).
Thompson, P. M. et al. ENIGMA and global neuroscience: a decade of large-scale studies of the brain in health and disease across more than 40 countries. Transl. Psychiatry 10, 100 (2020).
Dhamala, E., Yeo, B. T. T. & Holmes, A. J. One size does not fit all: methodological considerations for brain-based predictive modeling in psychiatry. Biol. Psychiatry 93, 717–728 (2023).
Milham, M. P. et al. Assessment of the impact of shared brain imaging data on the scientific literature. Nat. Commun. 9, 2818 (2018).
Uddin, L. Q. Accessible computing platforms democratize neuroimaging data analysis. Nat. Methods 21, 754–755 (2024).
Renton, A. I. et al. Neurodesk: an accessible, flexible and portable data analysis environment for reproducible neuroimaging. Nat. Methods 21, 804–808 (2024).
Hayashi, S. et al. brainlife.io: a decentralized and open-source cloud platform to support neuroscience research. Nat. Methods 21, 809–813 (2024).
Kiar, G. et al. Align with the NMIND consortium for better neuroimaging. Nat. Hum. Behav. 7, 1027–1028 (2023).
Ricard, J. A. et al. Confronting racially exclusionary practices in the acquisition and analyses of neuroimaging data. Nat. Neurosci. 26, 4–11 (2023). This paper discusses racially exclusionary practices in neuroimaging research and emphasizes the need for more inclusive methodologies to more accurately reflect diverse populations and improving the generalizability of findings in neuroscience.
Brown, S. A. et al. Responsible use of population neuroscience data: Towards standards of accountability and integrity. Dev. Cogn. Neurosci. 72, 101466 (2025).
Gao, P. et al. A Chinese multi-modal neuroimaging data release for increasing diversity of human brain mapping. Sci. Data 9, 286 (2022).
Tanaka, S. C. et al. A multi-site, multi-disorder resting-state magnetic resonance image database. Sci. Data 8, 227 (2021).