Plasmids
Cloning was performed either by Gibson assembly with the NEBuilder HiFi DNA Assembly Master Mix (New England Biolabs (NEB), E2621) or by site-directed mutagenesis by direct PCR with the KAPA HiFi HotStart Ready Mix (Roche, 07958935001) or Q5 Master Mix (NEB, M0492S). Plasmids were purified using the Plasmid Plus Midi Kit (QIAGEN, 12945) and verified by Sanger or nanopore sequencing. Constructs for insect cell expression were cloned into pFLN to facilitate bacmid generation in DH10EmBacY61.
Cell culture
All mammalian cells were cultured at 37 °C with 5% CO2. Expi293F cells (Gibco, A14527) were cultured in Expi293 expression media (Gibco, A1435102) and passaged 1:10 every 3 d to maintain a maximal cell density of 3 million cells per ml. K562 cells harbouring a miR-7-sensitive GFP reporter were a gift from J. Mendell10. K562 cells were cultured in RPMI 1640 (Gibco, 11875119) supplemented with 10% (v/v) fetal bovine serum (FBS) (Takara Bio, 631367) and passaged 1:4 every 3 d to maintain a maximal cell density of 800,000 cells per ml. Mouse embryonic fibroblast cells (MEFs) and HEK293FT cells were cultured in DMEM (Corning, 17-207-CV) with 10% FBS. Except in the case of contact inhibition for AGO2 intracellular co-IP and small-RNA-sequencing (sRNA-seq) experiments, described below, MEFs were passaged 1:4 or 1:8 as needed to maintain a maximal confluency of approximately 70%. HEK293FT cells were passaged 1:4 or 1:8 as needed to maintain a maximal confluency of approximately 70%. For passaging, adherent cells were dissociated using 0.25% trypsin–EDTA (Gibco, 25200114). For Expi293F cells, transfections were performed as described in the section ‘AGO–miRNA complexes for in vitro co-IP assays’. For K562 and HEK293FT cells, transfections were performed with Lipofectamine 2000 (Invitrogen, 11668019) and Opti-MEM (Gibco, 31985062) according to the manufacturer’s instructions. Drosophila S2 cells were cultured at 26 °C in Schneider’s Drosophila Medium (Gibco, 21720001) supplemented with 10% heat-inactivated FBS (Gibco, 16140071). S2 cells were passaged every 3–6 d to maintain an approximate concentration between 1 and 10 million cells per ml. For insect cell expressions, baculoviruses were generated from bacmids in Spodoptera frugiperda Sf9 insect cells (Thermo Fisher Scientific, 11496015) and used to infect Trichoplusia ni High-Five insect cells (Thermo Fisher Scientific, B85502) for expression. Cell lines were not authenticated. All cell lines tested negative for mycoplasma contamination upon arrival to the lab.
Proteins and RNAs
Proteins for in vitro ubiquitylation assays and for cryo-EM
Unless otherwise stated, all proteins are of human origin. Sequences of proteins and associated tags are listed in Supplementary Table 3. Genes were synthesized by TwistBiosciences or obtained as stated. Ubiquitin was expressed untagged and purified as described62. Cys-UB, in which ubiquitin carries an N-terminal cysteine for protein labelling, the lysine-less UB version (Cys-UB K0), NEDD8, UBE2L3, UBE2R2, UBE2M, ARIH1 and ARIH2 were expressed as GST-TEV or GST-3C fusions in BL21(DE3) RIL bacterial cells, as described35,39,63,64. Following glutathione-affinity pull-down (Cytiva, 17075605) and TEV protease cleavage, the protein was further purified by ion-exchange and size-exclusion chromatography. SrtA-5M, CUL3(NTD), CUL3(NTD, 1–384, I342R, L346D)-Avi and CUL3(∆N, NTD, 25–384, I342R, L346D)-Avi were expressed in BL21(DE3) RIL cells and purified by immobilized metal-affinity chromatography (Ni-INDIGO, Cube Biotech, 75105), followed by ion-exchange chromatography and size-exclusion chromatography, as described65,66. CUL2, CUL3, CUL5, GST-TEV-RBX1(5–108), GST-TEV(5–113) and CUL2(NTD, 1–380, L344K, V3776D)-Avi were expressed in insect cells using two baculoviruses for assembly of CUL–RBX complexes and purified following reported procedures35,63. CUL2–RBX1, CUL3–RBX1 and CUL5–RBX2 were neddylated and purified as described35,63.
ZSWIM8-Twin-Strep was cloned into pFLN. ELOB and ELOC were cloned into pFLN and assembled into pBig1a using biGBac Assembly61. ZSWIM8–ELOB–ELOC complex (hereafter referred to as ZSWIM8–ELOB/C) was expressed in insect cells using two baculoviruses for 3 d at 27 °C. Cells were resuspended in lysis buffer (50 mM HEPES, 300 mM NaCl and 10% (v/v) glycerol, pH 7.5) supplemented with protease inhibitors and 1 mM tris(2-carboxyethyl)phosphine (TCEP). Cells were disrupted by sonication and cell debris was removed by centrifugation. ZSWIM8–ELOB/C was enriched using Twin-Strep affinity purification (Strep-Tactin, IBA Lifesciences, 2-1201-025) and eluted with lysis buffer supplemented with 3 mM desthiobiotin, followed by size-exclusion chromatography (HiLoad Superdex 200, Cytiva).
ZSWIM8mono was designed on the basis of our cryo-EM structure. We hypothesized that CUL3 binding to ZSWIM8 requires the presence of a folded D-domain. To functionally separate the requirement of ZSWIM8 dimerization for AGO2 binding and ubiquitylation from its impact on CUL3 binding, we designed a ZSWIM8 variant that retained a folded D-domain and retained binding to CUL3. We expected that transplanting a second copy of the D-domain into the native sequence would allow the D-domain to fold in cis and resemble the D-domain that is natively folded in trans. We inserted D-domain residues E222–P273 with two flanking GGSGGS linkers in between D-domain residues Q228 and R229 (Supplementary Table 2). AlphaFold3 structure predictions supported the expected assembly of the D-domain.
For structural studies and stepwise ubiquitin transfer assays, His6-TEV-AGO2 was cloned into pFLN and expressed in insect cells. AGO was purified following an adapted purification protocol67. In brief, insect cells were resuspended in 50 mM Tris, 100 mM KCl and 5 mM dithiothreitol (DTT), pH 8.0. Following sonication, polyethylenimine was added to a concentration of 0.2% (v/v), mixed by inversion and immediately centrifuged to remove cell debris. AGO was enriched by immobilized metal-affinity chromatography (Ni-INDIGO, Cube Biotech, 75105). Following affinity chromatography, a nucleic-acid-free AGO fraction (as judged by 260/280 ratio) was isolated by cation-exchange chromatography (Source 15S or MonoS, Cytiva) with a gradient of 0–500 mM KCl in 20 mM Tris, pH 8.0. Nucleic-acid-free AGO was incubated with synthetic single-stranded miRNA (Metabion, IDT; Supplementary Table 4) at room temperature for 30 min. Excess miRNA was removed by size-exclusion chromatography in 25 mM HEPES, 150 mM NaCl and 1 mM TCEP, pH 7.8 (Superdex 200, Cytiva).
To label AGO for in vitro ubiquitylation assays, we used a split-intein-mediated labelling approach because of its high efficiency and site selectivity. We used the split intein gp41-1, which consists of an N-terminal part, gp41-1-N, and a C-terminal part, gp41-1-C68,69. These two parts excise themselves after trans-splicing, leaving a minimal ligation scar on the protein consisting of two short extein motifs. gp41-1-N was cloned into pGEX downstream of a TEV cleavage site. Following the TEV cleavage site, an N-terminal GGG motif was incorporated for sortase labelling, along with the native extein sequence SGY. GST-gp41-1-N was expressed in Rosetta Escherichia coli by induction with IPTG. Following glutathione-affinity purification and TEV protease cleavage, the protein was further purified by size-exclusion chromatography. gp41-1-C was cloned downstream of a Twin-Strep site into pFLN to create Twin-Strep-gp41-1-C-AGO2 or Twin-Strep-gp41-1-C-AGO1. These constructs contained the native extein linker, SSS, inserted between the split intein and the AGO sequence. AGO was purified following the procedure described above.
Labelling and biotinylation of proteins
Peptides for sortase labelling were obtained from the Peptide Synthesis Facility (Max-Planck Institute of Biochemistry Core Facility). The sortase peptide had the following sequence: GSGGLPETGG, with an N-terminally linked carboxyfluorescein. Peptides were more than 90% pure as judged by HPLC and mass spectrometry.
gp41-1-N was fluorescently labelled at its N-terminal GGG using sortylation with a fluorescein-labelled acceptor peptide (fluorescein-GSGGLPETGG) (Supplementary Table 3). gp41-1-N (100 µM) was mixed with peptide (350 µM) in 25 mM HEPES, 150 mM NaCl and 10 mM CaCl2, pH 7.5. The reaction was initiated by addition of SrtA-5M (10 µM)70 and allowed to proceed for 1 h at room temperature. Ni-INDIGO beads (20 µl) (Cube Biotech, 75105) were added and excess peptide was removed using a PD MidiTRAP G-25 (Cytiva) followed by size-exclusion chromatography (Superdex 75) in 25 mM HEPES, 150 mM NaCl and 1 mM TCEP, pH 7.5.
For labelling AGO proteins with fluorescein-gp41-1-N, the nucleic-acid-free fraction containing Twin-Strep-gp41-1-C-AGO2 or Twin-Strep-gp41-1-C-AGO1, isolated through ion-exchange chromatography, was incubated with miRNA and fluorescein-gp41-1-N at 1.2× molar excess over AGO. The reaction was allowed to proceed for 1 h at room temperature. Excess miRNA and unreacted fluorescein-gp41-1-N were removed by size-exclusion chromatography in 25 mM HEPES, 150 mM NaCl and 1 mM TCEP, pH 7.5. Following labelling, AGO is modified at its N terminus with fluorescein-GGGSGYSSS (Supplementary Table 3). Fluorescently labelled AGO was used in all in vitro ubiquitylation assays, as described below.
For biotinylation of CUL-NTDs, Avi-tagged CUL-NTDs were incubated with 50 mM ATP, 25 mM MgCl2, 5 mM biotin and 0.5 mg ml−1 BirA at 4 °C for 18 h following ion-exchange chromatography. Biotinylated NTDs were purified by size-exclusion chromatography.
For fluorescent labelling of UB, Cys-UB and Cys-UB K0 (1 mM) were buffer-exchanged using a PD MidiTrap G-25 column (Cytiva, 28918008) into 25 mM HEPES, 150 mM NaCl and 0.5 mM TCEP and incubated with 5-(Iodoacetamido)fluorescein (5 mM, Sigma-Aldrich, I9271) for 18 h at 4 °C. Excess 5-(Iodoacetamido)fluorescein was removed using a PD MidiTrap G-25 column followed by size-exclusion chromatography (Superdex 75) in 25 mM HEPES and 150 mM NaCl to yield fluorescently labelled UB and UB K0 (UB* and UB K0*, respectively). UB* was used in stepwise ubiquitin transfer assays, and UB K0* was used in ubiquitylation site mapping, as described below.
AGO–miRNA complexes for in vitro co-IP assays
Plasmids expressing AGO2 variants were generated from the pcDNA3.3-3xFLAG-SUMOEu1-HsAGO2 plasmid (Addgene, 231372). The plasmid expressing AGO1 was subcloned into the same backbone from an existing AGO1-expressing plasmid. Sequences of proteins and associated tags are listed in Supplementary Table 3. AGO–miRNA complexes were prepared largely as described51,71. Guide and passenger RNAs were each chemically synthesized (IDT) and gel-purified on a urea-polyacrylamide gel, then annealed into duplexes at 5 µM final concentration in annealing buffer (30 mM Tris-HCl pH 7.5, 100 mM NaCl and 1 mM EDTA). Annealing reactions were heated to 90 °C and slowly cooled to 30 °C over 1.5 h, before chilling on ice and storage at −80 °C. To generate cell lysate overexpressing the appropriate AGO variant, 200 ml of Expi293F cells at about 2 million per ml cell density were transfected with 190 µg of the AGO expression plasmid, 10 µg of pMaxGFP (Lonza) and 600 µg of polyethylenimine (Polysciences, 23966) incubated in 10 ml of Opti-MEM at room temperature for 20 min. After culture for 40 h, cells were collected and lysed in 8 ml of hypotonic buffer (10 mM HEPES pH 8.0, 10 mM KOAc, 1.5 mM Mg(OAc)2, 2% (v/v) glycerol, 5 mM NDSB-256 and 0.5 mM TCEP, with 1 tablet of cOmplete Mini EDTA-free Protease Inhibitor Cocktail (Roche, 11873580001) per 10 ml of buffer) by a Dounce homogenizer. Cell lysate was clarified by centrifugation at 3,000g for 15 min at 4 °C, then again for 30 min. The supernatant was re-equilibrated by adding 25% volume of re-equilibration buffer (150 mM HEPES pH 8.0, 700 mM KOAc, 15 mM Mg(OAc)2, 42% (v/v) glycerol, 5 mM NDSB-256 and 0.5 mM TCEP, with 1 tablet of cOmplete Mini EDTA-free Protease Inhibitor Cocktail per 10 ml of buffer), then further clarified by centrifugation at 60,000g for 20 min at 10 °C. Lysate was either used immediately or flash-frozen as aliquots in liquid nitrogen for storage at −150 °C.
To assemble each AGO–miRNA complex, 5–10 ml of cell lysate was first incubated with 600 µl of 5 µM annealed guide–passenger duplex for 1–2 h at 25 °C, to allow the in-lysate assembly of complexes. Meanwhile, 1 ml of slurry of Dynabeads MyOne Streptavidin C1 (Invitrogen, 65002) was washed according to the manufacturer’s instructions, then pre-bound to 1 nmol of gel-purified biotinylated, 2′O-methylated capture oligonucleotide containing an 8mer seed-match site with complementarity to the loaded miRNA (Supplementary Table 4), at 25 °C for 30 min with end-over-end rotation, followed by four washes with ice-cold equilibration buffer (18 mM HEPES pH 7.4, 100 mM NaCl, 1 mM MgCl2, 10% (v/v) glycerol, 0.003% (v/v) IGEPAL CA-630, 0.033 mg ml−1 recombinant albumin (NEB, B9200) and 0.005 mg ml−1 yeast transfer RNA (tRNA) (Life Technologies, AM7119)), then set aside at 4 °C until use. The incubated lysate was centrifuged at 6,000g for 10 min at 4 °C, filtered through a 5-µm PVDF filter (Millipore, SLSV025LS) and then added to the prepared beads and incubated at 25 °C for 1 h with end-over-end rotation. Beads were then washed twice with 5 ml of equilibration buffer and twice with 5 ml of capture-wash buffer (18 mM HEPES pH 7.4, 2 M NaCl, 1 mM MgCl2, 10% (v/v) glycerol, 0.003% (v/v) IGEPAL CA-630, 0.033 mg ml−1 recombinant albumin and 0.005 mg ml−1 yeast tRNA), then incubated with 1 nmol 3′-biotinylated DNA competitor oligonucleotide (IDT) complementary to the capture oligonucleotide (Supplementary Table 4) in 18 mM HEPES pH 7.4, 1 M NaCl, 1 mM MgCl2, 10% (v/v) glycerol, 0.003% (v/v) IGEPAL CA-630, 0.033 mg ml−1 recombinant albumin and 0.005 mg ml−1 yeast tRNA, at 25 °C for 2 h with end-over-end rotation. The eluate was then incubated with 50 µl of anti-FLAG M2 magnetic bead slurry (Millipore, M8823) pre-equilibrated in equilibration buffer according to manufacturer’s instructions, at 25 °C for 2 h with end-over-end rotation. The beads were washed twice with 500 µl of equilibration buffer and twice with 500 µl of storage buffer (18 mM HEPES pH 7.4, 100 mM NaCl, 1 mM MgCl2, 5% (v/v) glycerol and 0.003% (v/v) IGEPAL CA-630), before incubation in 100 µl of SENPEuB elution solution (500 nM of in-house purified SENPEuB protease51 in storage buffer) at 4 °C with gentle rotation for 1 h. The eluate was combined with an extra 100-µl wash with SENPEuB elution solution, then supplemented with β-mercaptoethanol (BME) (Sigma-Aldrich, M6250) to a final concentration of 0.5 mM. Aliquots were flash-frozen in liquid nitrogen for storage at −80 °C. AGO2 loaded with a 14-nt miR-7 was purified using the same method as for purifying AGO–miRNA complexes for in vitro ubiquitylation assays, described above, but without fluorescent labelling.
Purified AGO–miRNA complexes for use in in vitro co-IP assays were quantified by filter-binding titration. A serial dilution series of limiting concentrations of complexes were incubated with 2 nM of a radiolabelled target RNA, in 18 mM HEPES pH 7.4, 100 mM NaCl, 1 mM MgCl2, 2.5% (v/v) glycerol, 0.003% (v/v) IGEPAL CA-630, 0.5 mM BME, 0.017 mg ml−1 recombinant albumin and 0.003 mg ml−1 yeast tRNA, at 37 °C for 1 h. Filter binding was conducted as described above, except membranes were pre-equilibrated in 18 mM HEPES pH 7.4, 100 mM NaCl and 1 mM MgCl2 and washed with 18 mM HEPES pH 7.4, 100 mM NaCl, 1 mM MgCl2 and 5 mM DTT. Quantified fractions of target RNA bound across AGO–miRNA dilutions were fit to a quadratic equation by nonlinear least-squares regression in R using the Levenberg–Marquardt algorithm (nlsLM from the R package minpack.lm):
$${F}_{{\rm{b}}{\rm{o}}{\rm{u}}{\rm{n}}{\rm{d}}}=\frac{DF[\text{stock}]+[{\text{target}}_{T}]+{K}_{{\rm{D}}}-\sqrt{{(DF[\text{stock}]+[{\text{target}}_{T}]+{K}_{{\rm{D}}})}^{2}-4DF[\text{stock}][{\text{target}}_{T}]}}{2[{\text{target}}_{T}]}{F}_{\text{max}}$$
where Fbound represents fraction of target bound, [targetT] represents the concentration of total target oligonucleotide, [stock] represents stock concentration of RISC, DF represents the dilution factor, KD represents the dissociation constant for the affinity between RISC and the target and Fmax represents the maximal fraction of target bound at the plateau. A range of bounds and initiation values were tested for [stock], KD and Fmax to ensure robust estimation of [stock]. Fmax was always limited to the range (0, 1). KD was fit in log-transformed space.
ZSWIM8 for in vitro co-IP assays
3xFLAG-SUMOEu1-ZSWIM8-3xHA was cloned into pDARMO_CMVT (a gift from K. Rogala)72. ELOB and ELOC were cloned into pRK5. Sequences of proteins and associated tags are listed in Supplementary Table 3. For 200 ml of culture, Expi293F cells at about 3 million per ml cell density were transfected with 95 µg of the ZSWIM8 expression plasmid, 47.5 µg of the ELOB expression plasmid, 47.5 µg of the ELOC expression plasmid, 10 µg of pMaxGFP and 600 µg of polyethylenimine incubated in 24 ml of Opti-MEM at room temperature for 20 min. After culture for 48 h, cells were pelleted, washed with PBS and resuspended in double the cell pellet volume of lysis buffer (18 mM HEPES pH 7.4, 150 mM KOAc, 10 µM Zn(OAc)2, 5% (v/v) glycerol, 0.5% (v/v) IGEPAL CA-630 and 0.5 mM TCEP, with 1 tablet of cOmplete Mini EDTA-free Protease Inhibitor Cocktail per 10 ml of buffer). The resuspension was passed through a 23-G 1 M 1.5-inch needle and incubated at 4 °C for 15 min with end-over-end rotation. Lysate was clarified by centrifugation at 1,000g for 2 min at 4 °C, followed by centrifugation at 40,000g for 30 min at 4 °C. Meanwhile, 1.2 ml of anti-FLAG magnetic beads (Genscript, L00835) were washed twice with wash buffer (18 mM HEPES pH 7.4, 150 mM KOAc, 5% (v/v) glycerol, 0.01% (v/v) IGEPAL CA-630 and 0.5 mM TCEP). Clarified lysate was filtered through a 5-µm PVDF filter, then incubated with the prepared beads at 4 °C for 1.5 h with end-over-end rotation. After incubation with lysate, beads were washed four times with 3 ml of wash buffer and eluted with 1 ml of elution buffer (5 µM of in-house purified SENPEuB protease51 in wash buffer) at 4 °C for 1.5 h with end-over-end rotation. The eluate was centrifuged at 17,000g for 5 min at 4 °C, and the supernatant was collected and quantified by nanodrop. Aliquots were flash-frozen in liquid nitrogen for storage at −80 °C.
T6B peptide
3xHA-tagged T6B peptide (6xHis-SUMO-TNRC6B(599–683)-3xHA73, otherwise referred to as T6B-3xHA peptide) was expressed and purified as described previously74, with some modifications. 3xFLAG-tagged T6B peptide (6xHis-SUMO-TNRC6B(599–683)-3xFLAG, otherwise referred to as T6B-3xFLAG peptide) used in sRNA-seq experiments, described below, was purified by an analogous method. Sequences of proteins and associated tags are listed in Supplementary Table 3.
6xHis-SUMO-TNRC6B(599–683)-3xHA was cloned into pET28a. The expression construct was transformed into BL21 (DE3)-competent E. coli (NEB, C2527H). A single transformant was used to seed an overnight culture in Luria broth supplemented with 50 µg ml−1 kanamycin (GoldBio, K-120-5) and 35 µg ml−1 chloramphenicol (GoldBio, C-105-25) and grown at 30 °C overnight with shaking. Each 1.5-l batch of Luria broth was supplemented with 50 µg ml−1 kanamycin and 35 µg ml−1 chloramphenicol and seeded with 15 ml of overnight culture. This culture was grown at 37 °C with shaking. Once the culture reached an optical density (OD)600 of around 0.2, it was transferred to 18 °C with shaking, and, once it reached an OD600 of around 0.6, 0.5 mM of IPTG (GoldBio, I2481C50) was added. Induced cells were grown further overnight at 18 °C with shaking.
Cells were collected by centrifuging at 8,980g for 6 min at 4 °C. Pellets were resuspended in lysis buffer (50 mM Tris pH 8.0, 500 mM NaCl, 5% (v/v) glycerol, 15 mM imidazole, 4 mM BME and 1 tablet of cOmplete EDTA-free Protease Inhibitor Cocktail (Roche, 11873580001)), with 1 ml of lysis buffer per 240 ml of overnight culture. Cells were lysed by sonication, and lysate was clarified by centrifuging at 41,400g for 30 min at 4 °C. Clarified lysate was added to Ni-NTA agarose beads (QIAGEN, 30210) that had been washed twice with lysis buffer (280 µl of slurry per 1 l of culture), and the slurry was incubated at 4 °C for 2 h with end-over-end rotation. The beads were collected by centrifugation at 100g for 2–5 min at 4 °C and washed three times with 40 bead volumes of lysis buffer. Following the last wash, beads were resuspended in 5 ml of lysis buffer and transferred to a gravity-flow column. Captured protein was eluted with elution buffer (50 mM Tris pH 8.0, 500 mM NaCl, 5% (v/v) glycerol, 250 mM imidazole and 4 mM BME).
The eluate was dialysed at 4 °C overnight in dialysis buffer (50 mM Tris pH 8.0, 100 mM NaCl, 5% (v/v) glycerol, 15 mM imidazole and 4 mM BME) using a 10-kDa molecular weight cutoff dialysis cassette (Thermo Scientific, 69570). On the following day, the dialysed eluate was purified by anion-exchange chromatography on either a Resource Q column (Cytiva, 17-1179-01) or a HiTrap Q XL column (Cytiva, 17515901) pre-equilibrated in buffer A (50 mM Tris pH 8.0, 100 mM NaCl, 5% (v/v) glycerol and 4 mM BME). T6B-3xHA peptide was eluted off the column with a linear gradient of buffer B (50 mM Tris pH 8.0, 2 M NaCl, 5% (v/v) glycerol and 4 mM BME). Fractions containing T6B-3xHA peptide were pooled and concentrated by centrifugal filtration using a concentrator pre-equilibrated with buffer A (Amicon Ultra 10-kDa cutoff, Millipore, UFC8010). Concentrated T6B-3xHA peptide was further purified using size-exclusion chromatography with a Superdex 200 Increase column (Cytiva, 28990944) in buffer A. Fractions containing T6B-3xHA peptide were pooled, then centrifuged at 17,000g for 5 min at 4 °C, and the supernatant was collected and quantified by nanodrop. Aliquots were flash-frozen in liquid nitrogen for storage at −80 °C.
Target RNAs
Shorter target RNAs were chemically synthesized (IDT; Supplementary Tables 1 and 4) with 5′ hydroxyl chemistry, and gel-purified on a urea-polyacrylamide gel. Longer target RNAs were in vitro transcribed using chemically synthesized and gel-purified single-stranded DNA templates annealed to an oligonucleotide containing the T7 promoter sequence (IDT; Supplementary Tables 1 and 4). Transcription reactions using in-house purified T7 RNA polymerase were conducted in 5 mM ATP, 2 mM UTP, 5 mM CTP, 8 mM GTP, 5 mM DTT, 40 mM Tris pH 7.9, 2.5 mM spermidine, 26 mM MgCl2, 0.01% (v/v) Triton X-100, 5 mM DTT, SUPERase•In (1 U µl−1; Invitrogen, AM2694) and Thermostable Inorganic Pyrophosphatase (0.0083 U µl−1; NEB, M0296). After incubation at 37 °C for 3–4 h, DNA templates were digested with RQ1 DNase (Promega, M6101) at 37 °C for 30 min. RNA products were then gel-purified on a urea-polyacrylamide gel.
Gel-purified chemically synthesized RNAs were radiolabelled directly using T4 Polynucleotide Kinase (PNK) (NEB, M0201) and [γ-32P] ATP (Revvity, BLU035C005MC) in T4 PNK reaction buffer (NEB, B0201S) at 37 °C for 1–2 h, then desalted using Micro Bio-Spin P-6 columns (Bio-Rad, 7326221) and gel-purified on a urea-polyacrylamide gel. In vitro transcription products were first dephosphorylated using Quick CIP (NEB, M0525) at 37 °C for 15 min, followed by heat-inactivation at 80 °C for 3 min. Dephosphorylated RNAs were then immediately radiolabelled using T4 PNK and [γ-32P] ATP in rCutSmart buffer (NEB, B6004S) supplemented with 5 mM DTT (Invitrogen, 15508013) at 37 °C for 1–2 h, then desalted using Micro Bio-Spin P-30 columns (Bio-Rad, 7326250), Micro Bio-Spin P-6 columns or Amersham MicroSpin G-25 columns (Cytiva, 27532501) and gel-purified on a urea-polyacrylamide gel.
Biochemical assays
In vitro ubiquitylation assays
The identification of cullin–RING ligase subunits and the ARIH1 RBR-type E3 as TDMD effectors10 suggested that ZSWIM8 performs ubiquitylation by the previously defined E3–E3 mechanism, which is a variation on the canonical E1–E2–E3 pathway35,36. In the E3–E3 mechanism, ubiquitin is transferred from the E2 UBE2L3 to the active site of ARIH1 only after ARIH1 activation through binding a neddylated CRL. ARIH1 then ubiquitylates the substrate recruited to the CRL substrate receptor, and generates short ubiquitin chains. Optimal polyubiquitylation is achieved in collaboration with a UBE2R-family E2, also activated by the neddylated CRL. The design of our in vitro ubiquitylation assays was informed by this mechanism.
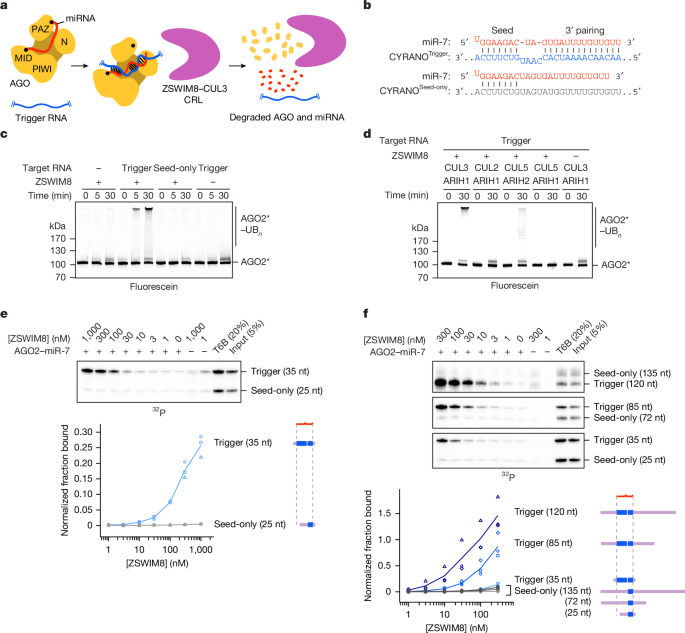
All concentrations stated refer to the final concentrations used. Fluorescently labelled AGO (0.2 µM) loaded with the indicated miRNA was incubated with a target RNA (0.22 µM) for 15 min at room temperature. In the meantime, CUL-NEDD8–RBX1 (0.5 µM), ZSWIM8–ELOB/C (0.5 µM), ARIH1 (0.4 µM), UBE2L3 (1.5 µM), UBE2R2 (1.5 µM), UB (50 µM) and BSA (0.5 mg ml−1) were mixed in 50 mM HEPES, 50 mM NaCl and 1 mM TCEP, pH 7.5. AGO–miRNA–target-RNA complex was added to the remaining components and incubated for 1 h at room temperature. ATP (5 mM) and MgCl2 (7.5 mM) were added, and the reaction was initiated by addition of UBA1 (0.1 µM). At indicated time points, samples were removed and quenched with an equal volume of 2 × SDS–PAGE sample buffer (100 mM Tris-HCl, 20% (v/v) glycerol, 30 mM EDTA, 4% (v/v) SDS and 4% BME) and incubated at 95 °C for 5 min. Samples were resolved on 4–22% SDS–PAGE gels and visualized on a Typhoon 9410 Imager by in-gel fluorescence, followed by gel staining with Coomassie blue. Assays were performed with n ≥ 2, and representative gels are shown. For reactions containing CUL5 and ARIH2, CUL-NEDD8–RBX1 was substituted for CUL5-NEDD8–RBX2, and ARIH1 was substituted with ARIH2. The assay setup as described above was used to generate the data shown in Figs. 1c,d and 3f and Extended Data Figs. 1e,h,i, 3m, 4i, 5g,h, 8a,e and 9b,g. Uncropped scans of fluorescent gels and gels stained with Coomassie blue are provided in Supplementary Figs. 1 and 2, respectively.
Stepwise ubiquitin transfer assays
Stepwise transfer assays were performed in a two-step process consisting of a loading reaction and a discharge reaction. In the loading reaction, the UB-carrying enzyme (UBE2L3) is charged by UBA1, and quenched by apyrase, which prevents further ubiquitin activation once UBE2L3 is discharged. The discharge reaction is initiated by addition of the remaining reaction components (CRL, substrate receptor, substrate and other E3s). Stepwise ubiquitin transfer assays follow the transfer of ubiquitin not just to the substrate but also to and from other proteins, such as E2 and E3 components. Substrate receptors that interact with CRLs are commonly autoubiquitylated and thus autoubiquitylation provides a functional readout of the ability of a substrate receptor to interact with a catalytically competent CRL.
All concentrations stated refer to the final concentrations used. AGO2 (0.2 µM) loaded with the indicated miRNA was incubated with a target RNA (0.22 µM) at room temperature for 15 min. In the meantime, CUL-NEDD8–RBX1 (0.5 µM), ZSWIM8–ELOB/C (0.5 µM), ARIH1 (0.4 µM) and BSA (0.5 mg ml−1) were mixed in 50 mM HEPES, 50 mM NaCl and 1 mM TCEP, pH 7.5, and incubated at room temperature for 15 min. In the meantime, UBE2L3 (10 µM) was mixed with fluorescein-UB (15 µM) in 25 mM HEPES, 100 mM NaCl, 2.5 mM MgCl2 and 1 mM ATP at room temperature. E2 charging was initiated by addition of UBA1 (0.4 µM) and allowed to proceed for 15 min. The reaction was quenched by addition of apyrase (NEB, 0.25 units) and incubated at 4 °C for 5 min. Assuming complete charging of UBE2L3, UBE2L3~UB* was further diluted to 2 µM in 25 mM HEPES and 150 mM NaCl, pH 7.5. Ubiquitylation was initiated by addition of UBE2L3~UB to the other components at a final concentration of 0.4 µM. For the sample at t0, an aliquot of UBE2L3~UB* was diluted to 0.4 µM in 25 mM HEPES and 150 mM NaCl, pH 7.5, and a sample was immediately removed and quenched with 2 × SDS–PAGE sample buffer (100 mM Tris-HCl, 20% glycerol, 30 mM EDTA and 4% SDS). Samples were removed and quenched at indicated time points. Samples were resolved on 4–22% SDS–PAGE gels and visualized on a Typhoon 9410 Imager by in-gel fluorescence, followed by gel staining with Coomassie blue. Assays were performed with n ≥ 2, and representative gels are shown. The assay setup as described above was used to generate the data shown in Extended Data Figs. 3l and 4f. Uncropped scans of fluorescent gels and gels stained with Coomassie blue are provided in Supplementary Figs. 1 and 2, respectively.
Ubiquitylation site mapping
To identify ubiquitylated lysines on AGO2, we adapted the stepwise ubiquitin transfer assay described above. Final concentrations were increased to CUL3-NEDD8–RBX1 (1.6 µM), ZSWIM8–ELOB/C (1.6 µM), ARIH1 (1.2 µM), AGO2 (2 µM) and UBE2L3~UB K0* (0.8 µM), and the reaction volume was increased to 50 µl. Instead of fluorescently labelled WT UB, we used fluorescently labelled UB K0, in which all lysines have been mutated to arginine to prevent unwanted ubiquitin chain formation. The reaction was allowed to proceed for 5 min at room temperature and was quenched by addition of 20 mM DTT.
Hereafter, 50 µl of SDC buffer (1% sodium deoxycholate, 40 mM 2-chloroacetamide and 10 mM TCEP in 100 mM Tris, pH 8.0) was added. Samples were incubated for 20 min at 37 °C and subsequently diluted 1:1 with water. Proteins were digested overnight at 37 °C using 0.5 µg of Lys-C and 1 µg of trypsin. Following digestion, peptides were acidified with trifluoroacetic acid to a final concentration of 1%. Approximately 200 ng of peptide material was loaded onto Evotips (Evotip Pure, Evosep) for liquid chromatography–tandem mass spectrometry analysis.
Evotips were eluted onto a 15-cm C18 analytical column (PepSep C18, 15 cm × 150 μm, 1.5 µm; Bruker Daltonics) using the Evosep One HPLC system. The column temperature was maintained at 50 °C, and peptides were separated using the 30 SPD method. Eluting peptides were directly introduced into a timsTOF HT mass spectrometer (Bruker Daltonics) via a nanoelectrospray ionization source. The mass spectrometer was operated in data-dependent PASEF (parallel accumulation serial fragmentation) mode, comprising one survey TIMS (trapped ion mobility spectrometry) mass spectrometry scan followed by ten PASEF tandem mass spectrometry scans per acquisition cycle. Data were acquired over a mass range of m/z 100–1,700 and an ion mobility range of 1/K0 = 0.85–1.35 Vs cm−2, using equal ion accumulation and ramp times of 100 ms in the dual TIMS analyser, corresponding to a spectral rate of 9.43 Hz. Precursor ions were isolated using a 2-Th window for m/z < 700 and a 3-Th window for m/z > 700 by synchronized quadrupole switching during TIMS elution. Collision energy was applied as a function of ion mobility, ranging from 45 eV at 1/K0 = 1.3 Vs cm−2 to 27 eV at 0.85 Vs cm−2, with linear interpolation between these values. Singly charged precursor ions were excluded using a polygon filter mask, and further m/z and ion mobility criteria were applied for dynamic exclusion.
Raw mass spectrometry data were processed using FragPipe (v.23.0) with default parameters from the LFQ-UB workflow. Spectra were searched against a database containing common contaminants and the proteins of interest. Carbamidomethylation of cysteine was set as a fixed modification, whereas methionine oxidation, lysine ubiquitylation, and protein N-terminal acetylation were included as variable modifications. StrictTrypsin was specified as the protease. The identified ubiquitylation sites are shown in Supplementary Fig. 3.
In vitro co-IP assays
All concentrations stated refer to the final concentrations used. Pierce anti-HA magnetic bead slurry (Thermo Scientific, 88837, 15 µl per reaction) was washed twice with blocking buffer (18 mM HEPES pH 7.4, 150 mM KOAc, 1 mM Mg(OAc)2, 5% (v/v) glycerol, 0.01% (v/v) IGEPAL CA-630, 0.1 mg ml−1 recombinant albumin and 0.01 mg ml−1 yeast tRNA) and incubated in blocking buffer at 4 °C for 2 h with end-over-end rotation. Blocking buffer was then removed immediately before use. Radiolabelled target RNAs were prepared as described above and pre-mixed. Pre-mixed RNA concentrations were calculated on the basis of a conservative estimate of 0–50% loss during radiolabelling. AGO–miRNA complex was pre-diluted in storage buffer (18 mM HEPES pH 7.4, 100 mM KOAc, 1 mM Mg(OAc)2, 20% (v/v) glycerol, 0.003% (v/v) IGEPAL CA-630, 0.5 mM BME, 0.033 mg ml−1 recombinant albumin and 0.005 mg ml−1 yeast tRNA). Unless other concentrations are specified, 0.75 nM of pre-diluted AGO–miRNA complex was pre-incubated with approximately 0.25 nM target RNAs in co-IP buffer (18 mM HEPES pH 7.4, 150 mM KOAc, 2 mM Mg(OAc)2, 5% (v/v) glycerol, 0.01% (v/v) IGEPAL CA-630 and 0.5 mM BME) supplemented with blocking reagents (0.1 mg ml−1 recombinant albumin and 0.01 mg ml−1 yeast tRNA) at 37 °C for 30–60 min. Where specified, heparin (Sigma-Aldrich, H3393), which reduces non-specific binding of RNA (especially longer RNA) to the beads, was also included in the blocking buffer and co-IP buffer. Heparin at 1 µg ml−1 was included in assays shown in Figs. 1f, 3g and 4e and Extended Data Figs. 6d and 9c,f. Heparin at 10 µg ml−1 was included in assays shown in Extended Data Figs. 1c and 8d. Heparin was not included in assays shown in Fig. 1e and Extended Data Figs. 1d,g, 2a–d and 8f–h. In the meantime, bait proteins ZSWIM8-3xHA and T6B-3xHA were pre-diluted in ZSWIM8 buffer (18 mM HEPES pH 7.4, 150 mM KOAc, 5% (v/v) glycerol, 0.01% (v/v) IGEPAL CA-630 and 0.5 mM TCEP).
Pre-incubated AGO–miRNA–target complex and pre-diluted bait proteins (or ZSWIM8 buffer) were mixed at a 1:1 ratio and incubated at 25 °C for 1 h, at a final volume of 30 µl per reaction. To reduce non-specific binding during the subsequent immunoprecipitation, any aggregation-prone species were removed by centrifugation at 17,000–21,000g for 5 min at 4 °C. Then, 5% of the supernatant was taken from the no-bait control to serve as input. Supernatants were otherwise resuspended with pre-blocked beads and incubated at 4 °C for 1.5 h, with intermittent shaking at 1,200 rpm for 15 s every 2 min. Following incubation, reactions were transferred to fresh tubes, and beads were washed four times with 125 µl of 18 mM HEPES pH 7.4, 150 mM KOAc, 1 mM Mg(OAc)2, 5% (v/v) glycerol, 0.01% (v/v) IGEPAL CA-630 and 0.5 mM BME, with a tube change during the fourth wash. Immunoprecipitated species were eluted by resuspension of beads with gel loading buffer (8 M urea, 25 mM EDTA, 0.025% (w/v) xylene cyanol, 0.025% (w/v) bromophenol blue) and shaking at 1,000 rpm at 65 °C for 10 min. Eluates were run on a urea-polyacrylamide gel. The gel was frozen at −20 °C while exposing a phosphorimager plate, and radioactivity was subsequently imaged on a phosphorimager. Band intensities were quantified using ImageQuant TL (v.10.2); image background was estimated either by subtracting the mean signal of equal-sized rectangles drawn at regions with no bands, or, where indicated, by additionally examining the lane profile tool drawn across each lane. Signal from each band was then background-subtracted using the 0 nM ZSWIM8 sample and normalized to that of the T6B sample. The assay setup as described above was used to generate the data shown in Figs. 1e,f, 3g and 4e and Extended Data Figs. 1c,d,g, 2a–d, 6d,e, 8d,f–h and 9c,d,f. Uncropped scans of gels are provided in Supplementary Fig. 1.
Filter-binding assays
To measure intrinsic affinity of ZSWIM8 for RNAs, a dilution series of purified ZSWIM8-3xHA was incubated in excess with radiolabelled RNAs in 18 mM HEPES pH 7.4, 150 mM KOAc, 1 mM Mg(OAc)2, 5% (v/v) glycerol, 0.01% (v/v) IGEPAL CA-630, 0.25 mM BME, 0.25 mM TCEP (Supelco, 646547), 0.1 mg ml−1 recombinant albumin and 0.01 mg ml−1 yeast tRNA, at 25 °C for 2 h.
Nitrocellulose (Amersham Protran, 0.45-µm pores; Cytiva, 10600062) and nylon (Amersham Hybond-XL; Fisher Scientific, 45001147) membrane filters were cut with a circle punch into discs of 0.5-inch diameter each and pre-equilibrated at room temperature for at least 20 min in 18 mM HEPES pH 7.4, 100 mM KOAc and 1 mM Mg(OAc)2. For each sample, a nitrocellulose disc was stacked on top of a nylon disc, atop a circular pedestal mounted on a Visiprep SPE Vacuum Manifold (Supelco, 57250-U), set at approximately −20 kPa. Then, 10 µl of reaction was promptly applied to the stacked discs, followed by 100 µl of ice-cold wash buffer (18 mM HEPES pH 7.4, 100 mM KOAc, 1 mM Mg(OAc)2 and 5 mM DTT). Filter membrane discs were separated with tweezers, air-dried, then exposed on a phosphorimager plate. Radioactivity was imaged on a Typhoon phosphorimager (Cytiva), and spot intensities were quantified using ImageQuant TL (v.10.2). The assay setup as described above was used to generate the data shown in Extended Data Fig. 2e.
Analytical size-exclusion chromatography
ZSWIM8–ELOB/C, ZSWIM8mono–ELOB/C and ZSWIM8∆dimer–ELOB/C (10 µM) were each incubated with CUL3–RBX1 (12 µM) in 25 mM HEPES, 150 mM NaCl and 1 mM TCEP, pH 7.5, at 4 °C for 30 min before analysis by size-exclusion chromatography (Superose 6, Cytiva). Samples were resolved on 4–22% SDS–PAGE gels, and gels were stained with Sypro Ruby (Sigma-Aldrich, S4942) before analysis by in-gel fluorescence. Analytical size-exclusion chromatograms and corresponding SDS–PAGE analyses are shown in Extended Data Fig. 3k. Uncropped scans of gels are provided in Supplementary Fig. 1.
Bio-layer interferometry
Bio-layer interferometry measurements were performed on an Octet K8 system (Sartorius) at 30 °C with shaking at 1,000 rpm. Concentrated proteins were diluted into BLI buffer (25 mM HEPES, 150 mM NaCl, 0.5 mg ml−1 BSA and 1 mM TCEP, pH 7.5). Biotinylated cullin-NTDs were immobilized on streptavidin sensors (Sartorius, 18-5019). ZSWIM8 served as the analyte and was measured in seven dilutions from 650 nM to 0.9 nM. Raw data were processed in Octet Data Analysis HT software (v.13.0.1). As no dissociation was detected, the KD was estimated through fitting Rmax of association. Processed data were plotted and fit using a single-site binding model in GraphPad Prism (v.10.4.0).
Sequence alignments of protein homologues
Protein sequences were downloaded from UniProt75. Multiple-sequence alignment was conducted with the MUSCLE algorithm76. Maximum-likelihood phylogeny was calculated using FastTree 2.2 (ref. 77).
Cryo-EM
Sample preparation
All concentrations stated refer to the final concentrations used. AGO2–miR-7 (7 µM) was mixed with a 120-nt fragment of the CYRANO trigger (8 µM) (Supplementary Table 1) and incubated at room temperature for 15 min. In the meantime, ZSWIM8–ELOB/C (6 µM) was mixed with CUL3(NTD) (6 µM) in 25 mM HEPES, 50 mM NaCl and 1 mM TCEP, pH 7.8. AGO2–miR-7–CYRANO was added and incubated on ice until plunge-freezing. Cu holey-carbon grids (R1.2/1.3, 200 mesh, Quantfoil, Q250-CR1.3) were freshly glow-discharged. Then, 3.5 µl of complex was applied, incubated for 5 s and blotted with a blot force of 2 and blot time of 2 s using a Vitrobot Mark IV (4 °C, 95% humidity) and plunge-frozen in liquid ethane.
Electron microscopy
A screening dataset was recorded on a Glacios cryo-transmission electron microscope at 200 kV using a Gatan Vista Alpine detector in counting mode and SerialEM software (v.4.1). The dataset consisted of 3,998 videos with a pixel size of 1.871 Å. Total exposure was set to 60 electrons per Å2 (40 frames), and with a defocus range between −0.5 and −2.5 µm. A high-resolution dataset was recorded on a Titan Krios transmission electron microscope at 300 kV using a Gatan K3 detector in counting mode and Serial EM software. The dataset (26,355 micrographs) was recorded at a magnification of 105,000, pixel size 0.8512 Å, total exposure 58 electrons per Å2 and a defocus range between −0.5 and −2.0 µm. A representative micrograph can be found in Supplementary Fig. 4.
Data processing
All datasets were processed in CryoSparc (v.4.6.2)78 (Extended Data Table 1). Raw video files were motion-corrected and dose-weighted, followed by estimation of contrast-transfer functions. Exposures were manually curated to remove micrographs with damage or ice contamination. Particles were picked in CryoSparc without a template and with a template generated from the screening dataset. Heterogeneous refinements, 3D classification, global and local refinements, particle subtraction and post-processing were performed in CryoSparc. For all heterogeneous refinements, one decoy class was included. All final maps were post-processed using manual sharpening or DeepEMhancer. The processing scheme is shown in Supplementary Figs. 4 and 5. 3D variability analysis was performed in CryoSparc with six modes and a filter resolution of 10 Å. Examples of map-to-model fits are shown in Supplementary Fig. 6.
Model building
The high-resolution map of CUL3(NTD)–ZSWIM8–ELOB/C–AGO2–miR-7–CYRANO (map A, sharpened with a B factor of 89.8) was further refined using three focused refined maps showing high-resolution features (maps B, C and D). A composite map was constructed on the basis of focused refined and DeepEMhancer-sharpened maps (map E). Each focused map has been deposited, along with a composite map. Maps A, B, C and E were used to build ZSWIM8NPAZ and ZSWIM8MID interactions with AGO2, RNA and C-terminal HEAT repeats. Maps A, C, D and E were used to build CUL3(NTD), ELOB, ELOC and the ZSWIM8 E3 superdomain. Structurally flexible RBEs were modelled using a combination of unsharpened maps (map F) and low-pass-filtered maps (map G). An initial model was generated by docking de novo-folded ZSWIM8 (AlphaFold3), CUL3 (PDB 5NLB), ELOB/C (PDB 1LM8) and AGO2 (PDB 6NIT) into the composite map. Iterative manual model building in COOT and real-space refinement using Phenix.refine were performed until a satisfactory map-to-model correlation was achieved79,80. Owing to the structural heterogeneity of CUL3, only the first 150 residues were modelled. For ZSWIM8, side chains were built for residues with interpretable density. For CUL3 and ELOB/C, side-chain residues were included on the basis of input models. Three long unstructured regions in ZSWIM8 were not modelled owing to the absence of any interpretable density. The TDMD sensor domain of ZSWIM8NPAZ includes a disconnected density that was modelled to contain ZSWIM8 residues Q39–R45 on the basis of AlphaFold3 predictions. RBEs 1–3 on ZSWIM8NPAZ were modelled with side chains on the basis of sharpened and unsharpened maps. RBEs 1–3 on ZSWIM8MID were modelled without side chains owing to inferior densities. C-terminal HEAT repeats were modelled on the basis of AlphaFold3 predictions without side chains owing to a lack of interpretable side-chain densities. Trigger RNA base U44 was poorly resolved in sharpened maps; its flipped-out position was modelled on the basis of unsharpened maps. The density for trigger RNA base A43 indicates a syn conformation and was modelled as such. All figures were generated using ChimeraX (v.1.8–v.1.9)81. All densities shown in figures were prepared using the composite map, unless otherwise stated.
Cellular assays
Cell lines
For generating clonal ZSWIM8-knockout K562s, parental K562 cells harbouring a miR-7-sensitive GFP reporter10 were transiently transfected with a plasmid derived from PX458 (Addgene, 48138) expressing pSpCas9-2a-BFP and either one or two single guide RNAs (sgRNAs) targeting the ZSWIM8 gene (Supplementary Table 4), followed by single-cell sorting of BFP-positive cells. Clones were screened for disruption of all ZSWIM8 alleles by amplification of the locus followed by nanopore sequencing of the PCR amplicon.
When generating clonal Zswim8-knockout MEFs, we observed substantial heterogeneity in clonal MEF populations derived from polyclonal parental populations, which was consistent with previous observations82. To minimize this clone-to-clone variability, we first generated a clonal parental population of WT MEFs by single-cell sorting of GFP-positive cells from MEFs83 that had been transiently transfected with a plasmid expressing pSpCas9-2a-GFP and a non-targeting sgRNA (Addgene, 48138; Supplementary Table 4). After isolating clonal lines, these parental cells were transiently transfected with a plasmid expressing pSpCas9-2a-GFP and an sgRNA targeting the Zswim8 gene (Addgene, 48138; Supplementary Table 4), followed by single-cell sorting of GFP-positive cells. Clones were screened for disruption of all Zswim8 alleles by amplification of the locus followed by Sanger sequencing of the PCR amplicon. Clonal WT MEFs were generated in parallel from the same parental clonal lines, except using a plasmid expressing pSpCas9-2a-GFP and a non-targeting sgRNA (Supplementary Table 4).
Polyclonal knock-in K562 lines stably expressing doxycycline-inducible ZSWIM8 transgenes were generated by PiggyBac transposition into clonal ZSWIM8-knockout K562 cells containing a miR-7-sensitive GFP reporter, described above. The WT ZSWIM8 CDS was cloned into PB-TetON (Addgene, 97421), and ZSWIM8 variants were generated from this WT plasmid. For each well in a six-well plate, approximately 600,000 cells were transfected with 1 µg of the PiggyBac transposon plasmid and 0.4 µg of the Super PiggyBac transposase plasmid (a gift from A. Hansen) using Lipofectamine 2000 and Opti-MEM according to the manufacturer’s instructions. After 48 h, cells were passaged 1:2 into fresh media containing 3 µg ml−1 puromycin (Gibco, A1113803). Subsequently, selection medium was refreshed every 3 d as cells were expanded up to 10-cm plates.
ZSWIM8 intracellular rescue assay
Following generation of ZSWIM8-knockout reporter K562 cells stably expressing doxycycline-inducible ZSWIM8 transgenes, as described above, about 400,000 cells were plated per well in a six-well plate with medium containing 3 µg ml−1 puromycin and 33.3 ng ml−1 doxycycline (Takara Bio, 631311). After 48 h, cells were expanded to 10-cm plates and medium containing 3 µg ml−1 puromycin and 33.3 ng ml−1 doxycycline was refreshed. After another 48 h, about 75% of the culture was set aside for northern analysis and about 12.5% was set aside for immunoblotting. The remaining 12.5% was concentrated to 2 million cells per ml in media and subjected to flow cytometry. For each sample, approximately 20,000 live cells were analysed using a BD LSR Fortessa instrument (BD Biosciences) and BD FACSDiva software (v.9.0). Subsequent analysis was performed using FlowJo (v.10.10.0). Gating strategies for flow cytometry analysis are shown in Supplementary Fig. 7. Quantified results of the ZSWIM8 intracellular rescue assay are shown in Figs. 3c and 4c and Extended Data Figs. 3f,n and 4b,j. Immunoblots of ZSWIM8 protein variants are shown in Supplementary Fig. 8.
RNA isolation
Total RNA was extracted with TRI Reagent (Invitrogen, AM9738) according to the manufacturer’s instructions. K562 cells were pelleted by centrifugation, washed with PBS and resuspended in 1 ml of TRI Reagent. GlycoBlue (Invitrogen, AM9516) was added to a final 25 µg ml−1 to facilitate precipitation and visualization of RNA pellets. After isopropanol precipitation and one 75% ethanol wash, pellets were resuspended in water and quantified by nanodrop.
Northern blots
Unless otherwise specified, 10 μg of total RNA was resolved on a denaturing 15% polyacrylamide gel and transferred onto a Hybond-N+ membrane (Cytiva, RPN303B) using a semi-dry transfer apparatus (Bio-Rad). The membrane was then incubated at 65 °C for 1–2 h with EDC (N-(3-dimethylaminopropyl)-N′-ethylcarbodiimide; Thermo Scientific, 22980) diluted in 1-methylimidazole, which chemically crosslinked 5′ phosphates to the membrane. Blots were hybridized to radiolabelled oligonucleotide probes (DNA or LNA) (Supplementary Table 4). Stripping of probes before re-probing was performed by incubation in boiling 0.04% (v/v) SDS with shaking. A detailed protocol for northern-blot analysis of small RNAs is available at http://bartellab.wi.mit.edu/protocols.html. Results were analysed on a Typhoon phosphorimager, using ImageQuant TL (v.10.2) for quantification of band intensities.
Immunoblots
K562 cells were pelleted by centrifugation, washed with PBS and lysed using Pierce IP Lysis Buffer containing 1 tablet of cOmplete Mini EDTA-free Protease Inhibitor Cocktail per 10 ml of buffer. Following 15 min of incubation in lysis buffer at 4 °C, lysates were clarified by centrifugation at 17,000g for 15 min at 4 °C and quantified by Bradford assay. Clarified lysates were incubated at 65 °C for 10 min in the presence of 1 × NuPAGE LDS sample buffer (Invitrogen, NP0007) and 2.5% (v/v) BME. Next, 8–12 µg of lysate was resolved on NuPAGE 4–12% Bis-Tris protein gels (Invitrogen, WG1403) with 1 × MES SDS running buffer (Invitrogen, NP0002) at 180 V for 50 min, and then transferred onto 0.45-μm PVDF membranes (Invitrogen, STM2006) at 25 V for 1 h on ice using a Midi Gel Tank (Invitrogen, STM1001) and Midi Blot Module (Invitrogen, STM2001). Membranes were then blocked with 5% (w/v) BSA (RPI, A30075-100.0) dissolved in PBS containing 0.1% (v/v) Tween 20 (Sigma-Aldrich, P1379) (PBS-T) for 1 h at room temperature, and then incubated with primary antibodies overnight at 4 °C. The following day, membranes were washed with 0.1% PBS-T at room temperature and incubated with near-infrared fluorescent secondary antibodies (LI-COR) for 1 h at room temperature, followed by washes and detection on an Odyssey CLx imager (LI-COR). Primary antibodies and their dilutions included: rabbit polyclonal anti-ZSWIM8 (1:400; Invitrogen, PA5-59492), rabbit monoclonal anti-HA (1:5,000; Cell Signaling Technology, C29F4, 3724) and mouse monoclonal anti-GAPDH (1:2,000; Invitrogen, GA1R, MA5-15738). Near-infrared fluorescent secondary antibodies and their dilutions included: IRDye 680RD goat anti-rabbit (1:10,000, LI-COR, 926-68071) and IRDye 800CW goat anti-mouse (1:10,000, LI-COR, 926-32210). Protein levels were quantified in ImageStudio (LI-COR, v.6.1.0.79).
Lentiviral transduction
Lentiviral production and transduction were performed as described11. In brief, transfer plasmids expressing WT AGO2, as well as R40 and RC5+6 variants (Supplementary Table 2), were prepared as described11. Transfer plasmids expressing new AGO2 variants were generated from the WT pLJM1-3xHA-AGO2 plasmid and prepared from NEB Stable Competent E. coli (NEB, C3040). For each well in a six-well plate, about 170,000 HEK293FT cells per cm2 were reverse-transfected with 1.4 μg of transfer plasmid, 0.94 μg of pCMV-dR8.91 packaging plasmid (a gift from J. Weissmann) and 0.47 μg of pMD2.G envelope plasmid (Addgene, 12259) using Lipofectamine 2000 and Opti-MEM. After 16 h, medium was refreshed. After another 48 h, medium was collected and centrifuged at 500g for 10 min to pellet debris. To infect MEFs, 500 μl of virus-containing supernatant (approximately 40% of the total) was combined with 1 ml of media and 12 μg ml−1 polybrene (Santa Cruz Biotechnology, SC-134220), and then added to one well of a six-well plate containing MEFs that were plated 24 h before at around 15,600 cells per cm2, for a final 8 µg ml−1 polybrene. Plates were centrifuged at 1,200g for 1.5 h at about 30 °C, and then returned to the 37 °C incubator. The following day, cells were expanded to 10-cm plates with media containing 4 µg ml−1 puromycin. Puromycin selection continued for another 6 d, refreshing selection media every 2 d. Cells typically reached confluency after 3 d of selection and were left confluent for another 3 d.
AGO2 intracellular co-IP assays
Experiments testing the effect of AGO2 mutations on intracellular levels of associated miRNAs were performed as described11. In brief, lentiviral production and transduction of MEFs with pLJM1-3xHA-AGO2 constructs were performed as described above. Following 6 d of puromycin selection (which included 3 d of contact inhibition), approximately 5 million cells per sample were washed with PBS, dissociated using 1 × TrypLE Express Enzyme (Gibco, 12604039), washed with PBS again and lysed with 600 μl of Pierce IP Lysis Buffer (Thermo Scientific, 87788) containing 1 tablet of cOmplete Mini EDTA-free Protease Inhibitor Cocktail per 10 ml of buffer. Following 15 min of end-over-end rotation in lysis buffer at 4 °C, lysate was clarified by centrifugation at 17,000g for 15 min at 4 °C. After setting aside a portion for immunoblotting, each clarified lysate was incubated with 50 μl of Pierce anti-HA magnetic beads with end-over-end rotation at 4 °C for 2 h. Beads were then washed three times with 250 µl of lysis buffer, and co-immunoprecipitating RNA was extracted with TRI Reagent added directly to the beads. Following RNA isolation, one-third of the total volume was used for northern analysis. Representative northern blots are shown in Extended Data Fig. 6b, and the corresponding quantification is shown in Extended Data Fig. 6c. Immunoblots of AGO2 protein variants are shown in Supplementary Fig. 11.
sRNA-seq
AGO protein, along with bound miRNAs, was isolated from MEFs and S2 cells using a FLAG-tagged T6B peptide73, as described74, except that a 3xFLAG-SUMO-tagged peptide (hereafter referred to as T6B-3xFLAG peptide) was used instead of the original 1xFLAG-GST. Its purification is described above. Zswim8-knockout and control MEF cell lines, generated as described above, were allowed to reach confluency, at which point medium was refreshed and cells were left confluent for 5 d before collection. Zswim8-knockout and control S2 cells11 were collected once they reached confluency. Both MEFs and S2 cells were flash-frozen as cell pellets before lysis. Anti-FLAG M2 magnetic beads were pre-coupled to T6B-3xFLAG peptide, washed to remove unbound peptide and then incubated with clarified lysate from MEFs or S2 cells that were lysed in 50 mM Tris pH 8.0, 150 mM NaCl, 5 mM EDTA, 10% (v/v) glycerol and 0.5% (v/v) NP-40, with 1 tablet of cOmplete Mini EDTA-free Protease Inhibitor Cocktail per 10 ml of buffer. Beads were washed three times with lysis buffer and once with PBS, and bound RNA was extracted directly from beads with TRI Reagent, as described above.
sRNA-seq libraries were prepared as described (http://bartellab.wi.mit.edu/protocols.html), with the following changes. sRNA-seq libraries were generated using 40% of the RNA isolated from a T6B pull-down. Instead of an 18-nt size marker, a 12-nt size marker (Supplementary Table 4) was used in all size-selection steps to capture shorter miRNA isoforms. For libraries generated from S2 cells, 2S ribosomal RNA was depleted from samples after the initial size selection using subtractive hybridization with a biotinylated antisense oligo, as described74. Libraries were sequenced on the Illumina NovaSeq 6000 platform with 100-nt single-end reads.
Processing of sequencing reads was done by trimming adaptors with cutadapt (v.4.8)84, filtering out reads with a quality score of 30 or below with FASTX Toolkit v.0.0.14 and then string-matching the first 13 nt of each read against a dictionary of miRNA names and sequences derived mainly from TargetScanFly7 and TargetScanMouse7 (ref. 85). For miRNAs whose sequences in the TargetScan annotations differed from those in miRGeneDB 3.0 annotations86, which was most common for lowly expressed and/or non-conserved miRNAs, sequences from miRGeneDB were added to the mapping dictionaries as processing variants (for example, mmu-let-7g-3p.1 and mmu-let-7g-3p.2). miRNAs sharing the same first 13 nt were collapsed into a single entry, and the full names of the collapsed species are provided in Supplementary Table 5. The length of each mapped miRNA was recorded, generating a table of read counts for each miRNA at each size, ranging from 13 to 31 nt.
To identify miRNAs accumulating in Zswim8-knockout lines compared with control lines, reads associated with all isoforms of a given miRNA were summed, filtered on the basis of a read cutoff of greater than five reads per library for at least two of the knockout lines and 30 reads across all six lines (three knockouts and three controls) and normalized for sequencing depth using the estimateSizeFactors function of DESeq2 (v.1.38.3)87. Fold-changes between Zswim8-knockout and control lines were then estimated using summed count values with DESeq2 v.1.38.3 without use of the lfcShrink function87. The mean of normalized abundances in the control lines was taken as the expression in WT cells. To identify miRNAs that accumulated significantly after loss of ZSWIM8, we applied a modified BBUM model (false discovery rate-adjusted P < 0.05)88.
Three approaches were used to identify miRNAs that were not classified as significantly up-regulated but nonetheless were potentially ZSWIM8 sensitive, with the goal of stringently removing from subsequent analyses any miRNAs whose dominant length isoform (typically 22 nt) could be subject to TDMD. First, using the fold-changes calculated by summing the counts associated with all isoforms of a given miRNA, miRNAs with Padj less than 0.05, in which the P values were adjusted by the Benjamini–Hochberg method by DESeq2, and fold-changes (log2) greater than zero were considered potentially ZSWIM8-sensitive. Second, the fold-change of each miRNA was compared with that of the co-produced passenger miRNA derived from the same precursor hairpin, as described12. miRNAs were classified as potentially ZSWIM8-sensitive if their log2 fold-changes and those of their passenger strands were separated beyond their respective standard errors, as calculated by DESeq2, as would be expected if ZSWIM8 altered the degradation rate of the miRNA and not the production rate. For miRNAs with more than one potential passenger strand, read counts attributed to each passenger were summed, and the log2 fold-change of the summed counts was used in the comparison. For this analysis, miRNAs were not subject to the read cutoff described previously, as many passenger miRNAs were lowly expressed. Third, miRNAs in MEFs were annotated as potentially ZSWIM8-sensitive if they were previously called ZSWIM8-sensitive in mouse tissues12, and miRNAs in S2 cells were annotated as potentially ZSWIM8-sensitive if they were called sensitive in S2 cells11 or Drosophila embryos20. The isoforms of miRNAs meeting any of these three conditions were excluded from the analyses in Extended Data Fig. 7. Analogous analyses for these miRNAs whose levels are significantly or potentially affected by ZSWIM8 are shown in Supplementary Fig. 15.
To investigate the relationship between miRNA length and ZSWIM8-dependent degradation, fold-changes observed after loss of ZSWIM8 were calculated for individual length isoforms of the remaining miRNAs that had not been excluded owing to potential ZSWIM8 sensitivity of their major isoforms. To this end, read counts corresponding to each isoform were first normalized using the size factors previously computed on the basis of the summed counts, and fold-changes (log2) were calculated with DESeq2. Finally, the median log2 fold-change of all 22-nt species was calculated and subtracted from the log2 fold-changes of all species. This normalization centres the distribution of log2 fold-changes for the 22-nt isoforms at zero, as would be expected if they were not sensitive to loss of ZSWIM8.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.