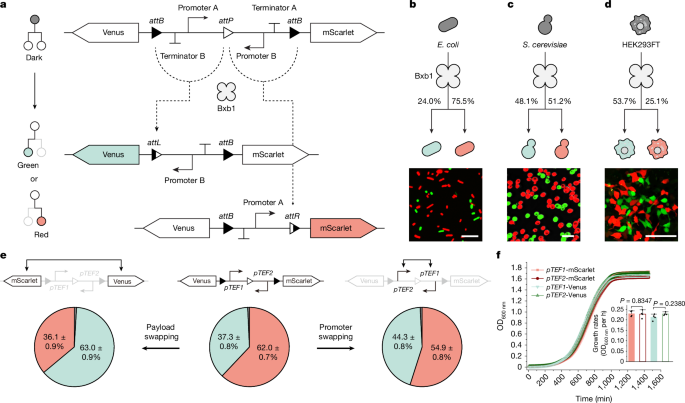
Bacterial strains and culture conditions
All cloning was performed in E. coli Trans1-T1 chemically competent cells (TransGen Biotech). Genetic circuit validation used E. coli DH10B (TSC-C09, Tsingke). Cultures were grown in lysogeny broth (LB) at 37 °C with shaking at 220 rpm. Plasmid maintenance was ensured by supplementing LB with chloramphenicol (34 μg ml−1), carbenicillin (100 μg ml−1), kanamycin (50 μg ml−1), spectinomycin (100 μg ml−1) or zeocin (100 μg ml−1), as appropriate.
Yeast strains and media
All yeast experiments used S. cerevisiae BY4741 (MATa his3Δ1 leu2Δ0 met17Δ0 ura3Δ0 sed1Δ; Horizon Discovery) as the standard chassis. Cells were cultured in YPD medium containing 1.5% (w/v) yeast extract (Angel Yeast), 2.0% (w/v) peptone (Angel Yeast) and 2% (w/v) glucose (Sangon). YPD was also used for circuit induction and growth curve measurements. Auxotrophic selection used SD minimal media (SD-Ura, SD-Leu, SD-His or SD-Ura-Leu-His) solidified with 2% agar (Coolaber). Antibiotic selection used SD supplemented with hygromycin (500 μg ml−1) or zeocin (300 μg ml−1). Snowflake yeast was generated by CRISPR–Cas9-mediated deletion of ACE2, as previously described42.
Mammalian cell culture
HEK293FT cells (Invitrogen, R70007) were maintained in high-glucose Dulbecco’s modified Eagle’s medium (DMEM) (Gibco) supplemented with 10% fetal bovine serum (FBS; Gibco) and 1% (v/v) penicillin–streptomycin (10,000 U ml−1 penicillin, 10 mg ml−1 streptomycin; Gibco) at 37 °C and 5% CO2. Cells were passaged at a 1:3 ratio every two days in 10-cm dishes. CHO-K1 cells (NICR) were cultured in F-12K medium (Kaighn’s modification; M&C Gene Technology) with 10% FBS and 2% (v/v) penicillin–streptomycin under identical conditions and passaged at a 1:6 ratio every three days. For selection, media were supplemented with hygromycin (800 μg ml−1), zeocin (400–1,000 μg ml−1), blasticidin S (20 μg ml−1) or puromycin (4 μg ml−1), as required.
Plasmid construction and single-copy maintenance in E. coli
To maintain the branching device at a single copy in E. coli, we used a bacterial artificial chromosome (BAC) backbone18. The intrinsically low copy number of BAC vectors minimizes copy-number-dependent variability and stabilizes circuit behaviour. All genetic components, including fluorescent reporters, promoters, terminators and insulators, were amplified via 2 × Phanta UniFi Master Mix (P526-02, Vazyme Biotech) and assembled into the BAC backbone using one-pot Gibson assembly (CU201-02, TransGen Biotech). Recombinase expression was controlled using a tightly regulated aTc-inducible system to drive the expression of Bxb1 recombinase53. Plasmids were verified by Sanger sequencing (Tsingke) and whole-plasmid sequencing (KBSeq, Sangon Biotech). Detailed genetic parts, protein sequences, plasmid backbones and representative constructs are provided in Supplementary Tables 4–7.
Plasmid construction and genomic integration in S. cerevisiae
To ensure single-copy circuit presence in S. cerevisiae BY4741, all devices were integrated into the genome. Plasmids were assembled using the Yeast Toolkit (YTK)24 and MYT system54, both based on Golden Gate cloning. Assembly reactions contained 6.5 μl DNA inserts, 1.0 μl backbone, 1.0 μl T4 ligase buffer (B0202S, NEB), 0.5 μl T4 DNA ligase (M0202L, NEB) and 1.0 μl restriction enzyme, adjusted to a total volume of 10 μl. BsaI-HF v2 (R3733L, NEB) or BsmBI v2 (R0739L, NEB) were used. Thermocycling comprised 80 cycles of digestion (BsaI at 37 °C or BsmBI at 42 °C for 5 min) and ligation (16 °C for 5 min), followed by final digestion at 55 °C for one hour and heat inactivation at 80 °C for 10 min. Assembled plasmids were transformed into E. coli, selected on antibiotic plates and verified by Sanger and whole-plasmid sequencing. Genetic parts and protein sequences are listed in Supplementary Tables 4 and 5, and plasmid backbones and representative constructs in Supplementary Tables 6 and 7.
Plasmid construction and genomic integration in HEK293FT cells
For site-specific genomic integration in HEK293FT cells, we first generated a landing-pad plasmid containing a single PhiC31 attB site (CGCGCCCGGGGAGCCCAAGGGCACGCCCTGGCAC), flanked by Rogi1 homology arms and a puromycin resistance cassette. A sgRNA targeting the Rogi1 locus (TTAGTCCTAGTGCCATGAAG) was cloned into pSpCas9-(BB)-2A-GFP under the human U6 promoter to enable CRISPR–Cas9-mediated targeting55. Cells were co-transfected with the Cas9–sgRNA and landing-pad plasmids, selected with puromycin and subjected to single-cell dilution in 96-well plates (Extended Data Fig. 6). Clones were expanded and screened by digital PCR (Tsingke, China) to identify lines carrying a single-copy attB insertion, which were then used for downstream circuit integration.
For integration of the branching circuit, a donor plasmid (synthesized by GenScript Corporation) containing the full device, a PhiC31 attP site (CCCCCAACTGAGAGAACTCAAAGGTTACCCCAGTTGGGG) and a bleomycin resistance gene (BleoR) was co-transfected with a PhiC31 integrase expression vector. Stable integrants were selected by culturing cells in zeocin-containing DMEM for two to four weeks to eliminate episomal plasmids through dilution during cell division. Single-cell isolation and digital PCR were then repeated to identify clones with single-copy integration. This step was essential, because PhiC31-mediated recombination in mammalian cells frequently generates off-target or multicopy insertions, which would confound quantitative differentiation analyses.
To enable doxycycline-inducible Bxb1 expression, Bxb1 or DHFR–Bxb1 was cloned into a Tet-On lentiviral vector (LV-Tet-Bxb1) containing blasticidin resistance and an iRFP reporter. Transduced cells were enriched by antibiotic selection and flow cytometry and used for subsequent differentiation experiments. All plasmids were validated by antibiotic selection, Sanger sequencing and full-plasmid sequencing. Detailed component descriptions, protein sequences and plasmid information are provided in Supplementary Tables 4–8.
Plasmid integration in CHO-K1 cells
As shown in Extended Data Fig. 6, a PhiC31 attB–containing landing pad was first introduced into the CHO-K1 genome using CRISPR–Cas9. Single-cell cloning followed by digital PCR screening was used to isolate clones with a single-copy insertion. These cells were then co-transfected with a plasmid carrying the branching circuit, a BleoR cassette, a TagBFP reporter and a PhiC31 attP site, together with a PhiC31 integrase expression vector. This strategy enabled site-specific recombination between the genomic attB and plasmid-borne attP sites, resulting in precise genomic integration of the circuit. After transfection, cells were serially passaged in zeocin-containing F-12K medium for approximately one month to enrich stable integrants and dilute residual episomal plasmids. TagBFP-positive cells (405-nm excitation) were subsequently isolated by fluorescence-activated cell sorting, followed by single-cell dilution and digital PCR to identify clones containing a single-copy circuit insertion.
For doxycycline-inducible recombinase expression, Bxb1 or DHFR–Bxb1 was cloned into a Tet-On lentiviral vector (LV-Tet-Bxb1) containing a blasticidin resistance cassette and an iRFP reporter. Transduced cells were selected with blasticidin, sorted by flow cytometry based on iRFP fluorescence (640-nm excitation) and used for downstream differentiation assays. All plasmids were validated by antibiotic selection, Sanger sequencing (Tsingke) and full-plasmid next-generation sequencing (NGS) (Cwbio). Detailed genetic components, protein sequences and plasmid maps are provided in Supplementary Tables 4–8.
Transformation of E. coli
Plasmid DNA (1–10 μL) was mixed with 200 μl freshly thawed chemically competent E. coli and incubated on ice for 30 min. Cells were heat-shocked at 42 °C for 45 s, cooled on ice for 2 min and recovered in 500 μl antibiotic-free LB medium at 37 °C for one hour before plating on selective LB agar.
Yeast transformation
Plasmids were linearized with NotI in a reaction containing CutSmart buffer, NotI-HF (R3189L, NEB) and plasmid DNA, followed by incubation at 37 °C for one hour. Five microlitres of digested DNA was mixed with boiled salmon sperm ssDNA (CS9672, Coolaber) to a final volume of 10 μl. S. cerevisiae cells were cultured in YPD at 30 °C for one to two days, collected by centrifugation and washed three times with 0.1 M lithium acetate (LiOAc; ref. 56). Cells were resuspended in 0.1 M LiOAc, and 2-μl aliquots were used as competent cells. For transformation, plasmid–ssDNA was combined with competent cells, 50% PEG-3350 and 1 M LiOAc, mixed gently and incubated sequentially at 42 °C and 30 °C for 40 min each. Cells were then pelleted, resuspended in sterile water and plated on SD agar for auxotrophic or antibiotic selection.
Lentivirus production and transduction
Lentiviral particles (LV-Tet-Bxb1) were produced in HEK293FT cells using a three-plasmid system57. The transfer plasmid encoding Bxb1 or DHFR–Bxb1 was co-transfected with psPAX2 and pMD2.G using Lipofectamine 3000 (L3000008, Thermo Fisher Scientific) according to the manufacturer’s instructions. Viral supernatants were collected at 48 h and 72 h, filtered through 0.45-μm PES membranes and concentrated by ultracentrifugation.
For transduction, 1 × 106 HEK293FT cells or 8 × 105 CHO-K1 cells were seeded per well in 6-well plates and cultured for 24 h. The medium was replaced with 1 ml viral suspension for 18 h, followed by the addition of fresh medium and incubation for another 24 h. Cells were then transferred to 10-cm dishes, dissociated with 0.25% trypsin and cultured in selection medium. Fresh antibiotic-containing medium was replaced every four days until confluence was reached.
Digital PCR for copy-number analysis
Gene copy numbers in engineered E. coli, S. cerevisiae BY4741 and mammalian cells were determined using ratio-based digital PCR. Genomic DNA was extracted and analysed by Beijing Tsingke Biotech Co., Ltd (China). Absolute copy numbers were calculated by normalizing target concentrations to endogenous reference genes with known copy numbers in each chassis.
Digital PCR enables absolute nucleic acid quantification with high sensitivity and accuracy58 by partitioning reactions into thousands of nanolitre-scale droplets, each acting as an independent PCR microreactor. After amplification, fluorescence was measured to classify droplets as positive or negative, and target concentrations were calculated using Poisson statistics. TaqMan probes were used, with FAM–BHQ1 for target detection and VIC–MGB for reference detection. Reactions were performed using 2× T5 Fast qPCR Mix (probe-based, Tsingke) according to the manufacturer’s protocol.
Copy number (N) was calculated as
$$N=C({\rm{t}}{\rm{a}}{\rm{r}}{\rm{g}}{\rm{e}}{\rm{t}}{\rm{e}}{\rm{d}}\,{\rm{g}}{\rm{e}}{\rm{n}}{\rm{e}})/C({\rm{r}}{\rm{e}}{\rm{f}}{\rm{e}}{\rm{r}}{\rm{e}}{\rm{n}}{\rm{c}}{\rm{e}}\,{\rm{g}}{\rm{e}}{\rm{n}}{\rm{e}})$$
where C denotes absolute concentration (copies per μl). Concentrations were derived using
$$C=-\frac{1}{v}{\rm{l}}{\rm{n}}\,\left(1-\frac{k}{n}\right)$$
where n is the total number of partitions, k is the number of positive partitions and v is the partition volume. For the SinoGene DQ24 digital PCR system (Tsingke Biotechnology), v = 0.6 × 10−3 μl. The resulting copy-number data are shown in Supplementary Fig. 3.
Flow cytometry
For most yeast analyses, strains were inoculated into 1 ml YPD supplemented with inducers (1 mM aTc or 1 μM β-oestradiol) and cultured overnight at 30 °C with shaking at 800 rpm in 2.2 ml deep-well plates. The following day, cultures were diluted 1:50 in 200 μl ddH2O in 96-well flat-bottom plates and analysed on an Attune NxT flow cytometer (Thermo Fisher Scientific). For each sample, 10,000 events were collected and analysed using CytExpert 2.4. For doublet analysis, cultures were diluted 1:5 in ddH2O before acquisition.
For mammalian cell sorting, confluent cells from 10-cm dishes were dissociated with 0.25% trypsin, centrifuged and resuspended in 2 ml F-12K medium containing 4% penicillin–streptomycin. Cell suspensions were filtered through a 75-μm strainer and sorted on a FACSAria Fusion (BD Biosciences). Approximately 2 × 105 sorted cells were collected into 6-well plates for downstream culture.
Growth curves
Single yeast colonies were grown overnight in 1 ml YPD at 30 °C with shaking at 800 rpm in 2.2 ml deep-well plates. Cultures were diluted the next day to an OD600 nm of around 0.06 in 200 μl fresh YPD in clear, flat-bottom 96-well plates. OD600 nm was measured every 5 or 10 min for 30 h at 30 °C using a microplate reader (Synergy H1, BioTek) with a 10-s shake before each read. Maximum exponential growth rates were calculated as: LN (OD600 nm(t + 5)/OD600 nm(t))/5, where t is time in hours.
Consortium passaging
To assess the long-term stability of differentiated yeast consortia, cells were mixed at defined ratios and cultured in 5 ml YPD at 30 °C for 24 h. Population composition was quantified by flow cytometry, after which cultures were diluted 1:1,000 into fresh YPD and grown for another 24 h. This cycle was repeated for seven rounds to track compositional drift.
To evaluate circuit stability without induction, yeast cultures were serially passaged at a 1:1,000 dilution for 15 rounds (around 15 days) and monitored for spontaneous differentiation due to recombinase leakiness by flow cytometry. In CHO-K1 cells, stability was similarly assessed over 5 passages (around 15 days) at a 1:6 dilution by monitoring unintended differentiation events.
NGS analysis
Amplicon-based NGS was performed on yeast containing differentiation circuits. Cultures were grown in 5 ml YPD and induced with 1 mM aTc or 1 μM β-oestradiol. Genomic DNA was extracted using a TIANamp Yeast DNA Kit (DP307, TIANGEN). A 300-bp region spanning the recombinase-mediated rearrangement was amplified by PCR using the following primers: forward 5′-CGACGGCGATCACAGACATTAA-3′ and reverse 5′-CTCACACTGACGAATCATGT-3′. PCR products were purified and submitted to Tsingke for sequencing and analysis.
Confocal microscopy
For liquid cultures, 5 μl yeast suspension was placed on a microscope slide and covered with a 1 cm × 1 cm coverslip. Samples were imaged using a Nikon AX confocal microscope. For colony imaging, SD medium containing 0.7% agar and appropriate inducers or antibiotics was prepared, and 2 μl of yeast diluted to an OD600 nm of around 0.06 was spotted onto plates. After incubation at 30 °C for three to five days, colonies were imaged by confocal microscopy.
For time-lapse imaging of differentiation (Supplementary Videos 1–3), uninduced yeast were diluted 1:1,000 (OD600 nm ≈ 0.01) and seeded into glass-bottom dishes containing induced YPD. Cultures were maintained at 30 °C on a temperature-controlled stage, and fluorescence images were acquired every 10 min to capture aggregate formation.
For pattern-formation imaging (Supplementary Videos 4 and 5), engineered progenitors were diluted to around 100 cells and seeded into 96-well round-bottom ultra-low-attachment plates (Corning, 7007) containing induced YPD. Plates were centrifuged at 100g for 10 min to promote cell contact and incubated at 30 °C. Time-lapse imaging was performed every 10 min using a Nikon AX microscope.
For CHO-K1 time-lapse imaging, 100–1,000 cells were seeded into ultra-low-attachment 96-well plates with F-12K medium, with doxycycline and TMP added as required. Plates were incubated at 37 °C with 5% CO2, and confocal images were acquired every 24 h.
Aggregate area analysis
Ten confocal images of each engineered yeast aggregate shown in Supplementary Fig. 84 were acquired. Aggregate areas were quantified using ImageJ (v1.54g). Objects smaller than 400 μm2 were excluded, and the remaining measurements were used to generate aggregate size distributions.
Sedimentation assay
Yeast containing genetic circuits were cultured overnight in 1.5 ml induced YPD at 30 °C and 800 rpm in deep-well plates, then adjusted to an OD600 nm of 1.7. One millilitre of culture was transferred to a cuvette, and OD600 nm was recorded every 30 min for 210 min using a UV spectrophotometer to determine sedimentation rates.
Protein secretion assay
Protein secretion was assessed using the Nano-Glo HiBiT Extracellular Detection System (Promega), which detects complementation between the 11-amino-acid HiBiT tag (VSGWRLFKKIS) and LgBiT to generate bioluminescence59. HiBiT was fused to target proteins, and 100 μl culture supernatant was mixed with 1 μl LgBiT, 97 μl reaction buffer and 2 μl substrate. After 5 min of incubation at room temperature, luminescence was measured with a BioTek plate reader. Non-secreting BY4741 cells served as a negative control.
α-Galactosidase activity
X-α-Gal stock (40 mg ml−1; Sigma-Aldrich, 16555) was prepared in dimethyl sulfoxide (DMSO). For activity assays, 100 μl stock was added to 3 ml of YPD inoculated with yeast at a 1:1,000 dilution. Cultures were incubated for 24 h at 30 °C, then collected and imaged to assess α-galactosidase activity.
Secreted laccase activity
Laccases are copper-dependent enzymes and therefore require copper supplementation60. Stock solutions of 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS; Sigma-Aldrich, A1888) and copper sulfate were prepared at final concentrations of 0.1 M and 1 M, respectively. To assay secreted laccase activity, 125 µl ABTS and 25 µl CuSO4 were added to 3 ml YPD medium inoculated with yeast cells (1:1,000). Cultures were incubated at 30 °C for 24 h and subsequently imaged.
Secreted lipase activity
Yeast strains were cultured in YPD medium supplemented with 1% soybean oil for 48 h. Fermentation broth (5 ml) was collected by centrifugation (12,000 rpm, 15 min). Lipids in the supernatant were extracted by sequential mixing with 500 µl acetic acid, 500 µl 12% (w/v) NaCl and 2 mL ethyl acetate. After vortexing for 15 min and centrifugation (12,000 rpm, 15 min), the organic phase was recovered, dried under nitrogen and resuspended in chloroform. Lipids were separated by thin-layer chromatography using hexane: diethyl ether: acetic acid (70:30:1) and visualized with iodine vapour.
Violacein and β-carotene extraction
Violacein and β-carotene were extracted from cultured yeast by collecting cells (1 ml, OD600 nm = 2.0) via centrifugation (4,000 rpm, 1 min). Cell pellets were resuspended in 500 µl DMSO and incubated at 37 °C for 30–60 min with grinding to enhance lysis and extraction efficiency. After centrifugation (4,000 rpm, 1 min), supernatants containing the pigments were analysed by full-spectrum scanning (400–650 nm, 5-nm intervals) using a BioTek microplate reader.
Quantifying pigment-producing cell ratios
Yeast strains containing pZ-driven violacein and pPhlF-regulated β-carotene pathways were transformed with a branching circuit using Z3EV and PhlF-VP16 as reporters. Colonies were grown on agar plates for three days, inoculated into 5 ml YPD supplemented with 1 mM aTc and 1 nM β-oestradiol and cultured at 30 °C with shaking (1,000 rpm) for eight hours. Notably, 1 nM β-oestradiol was sufficient for robust induction, substantially lower than the commonly used 1 µM. Cultures were diluted 10,000-fold, plated onto YPD agar containing 1 nM β-oestradiol and incubated for three days. Plates were imaged, and colonies producing orange (β-carotene) or purple (violacein) pigments were counted.
Recombinase-guided pigment library
A library of 25 synthetic differentiation circuits was constructed by Golden Gate assembly24. Each construct contained a unique engineered attP variant with specific point mutations that tune recombination efficiency with Bxb1 recombinase, spanning a continuum from fast to slow recombination rates (Supplementary Fig. 80). These differences modulate the activation of downstream modules (Z3EV or PhlF-VP16), generating variable proportions of violacein- and β-carotene-producing subpopulations. The plasmid library was transformed into S. cerevisiae BY4741 engineered with both pigment pathways under distinct synthetic transcriptional control. Ninety-six colonies were randomly selected and cultured in 2.2 ml deep-well plates at 30 °C and 800 rpm in the presence of inducers (1 mM aTc and 1 nM β-oestradiol) to initiate differentiation.
DNS assay for cellulase activity
Glucose concentrations were measured using a modified DNS method61. Samples (50 µl) were mixed with 100 µl DNS buffer (Macklin), heated at 100 °C for 5 min and cooled on ice for 1 min. After adding 150 µl ddH2O, 200 µl of each sample was transferred to a microplate, and absorbance at 540 nm was measured. Glucose concentrations were calculated from a standard curve (0.125–5 g l−1) (Supplementary Fig. 81). Cellulase activity was assessed using 1 ml culture supernatant (OD600 nm = 2.0) incubated with 2% Avicel PH-101 (Sigma) in sealed deep-well plates at 35 °C with shaking (around 300 rpm) for 24 h, followed by glucose quantification using the DNS assay.
Snowflake yeast construction
Snowflake yeast strains were generated by deleting ACE2 using CRISPR–Cas9, as previously described42. The plasmid pMYT095, containing Cas9 and gRNA expression cassettes, was used for genome editing54. The guide RNA (TTATTCAAAATATAATTGTCGGG) was designed using the Yeastriction online tool. Plasmids for ACE2 deletion were constructed and validated by Sanger sequencing (Sangon Biotech). Donor DNA fragments were synthesized, purified (Vazyme DNA Extraction Mini Kit) and co-transformed with plasmids into yeast using the lithium acetate method. Transformants were confirmed by PCR and Sanger sequencing.
Image-based cell–cell interaction analysis
Cell–cell interactions in engineered yeast populations were quantified using a five-step image-processing workflow based on fluorescence microscopy (Supplementary Fig. 86). Raw images were first manually enhanced in Adobe Photoshop to improve contrast and delineate cell boundaries. Enhanced images were then segmented in CellProfiler (v.4.2.1) by splitting RGB channels and identifying primary objects with diameters of 10–160 pixels using adaptive Otsu thresholding in three-class mode. Segmented data were imported into MATLAB (v.2024b) for downstream analysis. Individual cells were classified by dominant RGB fluorescence intensity, corresponding to specific engineered cell types. Interactions were defined by spatial proximity, with cell pairs within 35 pixels considered interacting. Interaction networks were visualized by connecting adjacent cells, and normalized contact-frequency heat maps were generated to quantify self- and heterotypic interaction preferences across the population.
In vitro morphogenesis of engineered mammalian cells
To engineer and analyse cellular morphologies in CHO-K1 cells, two complementary in vitro culture strategies were used. In the first, CHO-K1 cells pre-differentiated by doxycycline and TMP induction were collected and diluted to around 100–1,000 cells per well, then seeded into 96-well ultra-low attachment plates containing 100 μl F-12K medium supplemented with 10% FBS, 2% penicillin–streptomycin, 4 μg ml−1 puromycin, 20 μg ml−1 BSD and 400 μg ml−1 zeocin. Cells were maintained at 37 °C with 5% CO2, and morphological evolution was monitored by confocal fluorescence microscopy at 2, 12, 24, 48, 72 and 96 h after seeding.
In the second strategy, around 100–1,000 CHO-K1 cells containing a single-copy differentiation circuit and an inducible Bxb1 recombinase system were seeded under identical culture conditions. In situ differentiation was initiated by adding 1 μg ml−1 doxycycline and 0.1 mM TMP at seeding. Morphogenesis and structure formation were tracked by confocal microscopy at the same time points. Together, these time-course analyses enabled controlled observation of genetically programmed, inducer-driven morphological dynamics in the culture environment.
Predictive modelling of circuit-driven differentiation
To enable predictive design of differentiation patterns, we developed a simplified mathematical model describing Bxb1-mediated excision of the engineered circuit, accounting for conditions with and without steric hindrance from transcription machinery. We hypothesized that strong promoters recruit more transcription factors, thereby reducing Bxb1 cleavage efficiency relative to weak promoters. In the absence of steric effects, the left and right arms are cleaved at rates kA and kB, respectively, which depend on arm length. This dependence was modelled using power laws: \({k}_{{\rm{A}}}={k}_{A}^{0}{h}_{{\rm{A}}}^{-\beta },\) \({k}_{{\rm{B}}}={k}_{{\rm{B}}}^{0}{h}_{{\rm{B}}}^{-\beta },\) where hA and hB denote arm lengths, and β is a positive constant.
Promoter-driven recruitment of transcription machinery introduces steric hindrance that reduces recombination efficiency. Binding on the left arm decreases the cleavage rate from kA to vA, whereas binding on the right arm decreases kB to vB. We assumed that steric hindrance on one arm does not influence recombination on the other. Hindrance strength was quantified by α, defined as: \(\alpha =\frac{{v}_{{\rm{A}}}}{{k}_{{\rm{A}}}}=\frac{{v}_{{\rm{B}}}}{{k}_{{\rm{B}}}}.\) Under these assumptions, circuit-mediated differentiation dynamics were described by the following ordinary differential equation system:
$$\frac{{\rm{d}}[D]}{{\rm{d}}t}=-{(k}_{{\rm{A}}}+{k}_{{\rm{B}}}+{r}_{{\rm{A}}}+{r}_{{\rm{B}}})[D]$$
$$\frac{{\rm{d}}[{L}_{{\rm{A}}}]}{{\rm{d}}t}={k}_{{\rm{A}}}[D]+{v}_{{\rm{A}}}[{M}_{{\rm{A}}}]+{k}_{{\rm{A}}}[{M}_{{\rm{B}}}],$$
$$\frac{{\rm{d}}[{L}_{{\rm{B}}}]}{{\rm{d}}t}={k}_{{\rm{B}}}[D]+{v}_{{\rm{B}}}[{M}_{{\rm{B}}}]+{k}_{{\rm{B}}}[{M}_{{\rm{A}}}]$$
$$\frac{{\rm{d}}[{M}_{{\rm{A}}}]}{{\rm{d}}t}={r}_{{\rm{A}}}[D]-{v}_{{\rm{A}}}[{M}_{{\rm{A}}}]-{k}_{{\rm{B}}}[{M}_{{\rm{A}}}]$$
$$\frac{{\rm{d}}[{M}_{{\rm{B}}}]}{{\rm{d}}t}={r}_{{\rm{B}}}[D]-{v}_{{\rm{B}}}[{M}_{{\rm{B}}}]-{k}_{{\rm{A}}}[{M}_{{\rm{B}}}]$$
Here, [D] denotes the population-level concentration of the ‘naked’ circuit; [LA] and [LB] represent recombination products generated by left- or right-arm excision, respectively; MA and MB denote circuit states bound by transcription machinery; and rA and rB are binding rates.
Although full parameterization would allow direct prediction of excision ratios, many parameters are experimentally inaccessible. We therefore estimated parameters by fitting the model to experimental data, minimizing the discrepancy between predicted and observed ratios. The objective function was defined as \(\phi ={\sum }_{i=1}^{m}{({y}_{i}-{x}_{i})}^{2},\) where xi is the measured ratio, yi the predicted value and i indexes the data points. Optimal parameters were obtained by minimizing ϕ over the defined parameter space.
Experimentally, we varied left and right arm lengths and quantified Venus-to-mScarlet cell ratios across combinations (Fig. 2d), which were used for model fitting (Supplementary Figs. 39 and 40). Parameters were screened over the ranges \(0 < {k}_{{\rm{A}}}^{0} < 0.3\,{{\rm{h}}}^{-1},\) \(0 < {k}_{{\rm{B}}}^{0} < 0.3\,{{\rm{h}}}^{-1},\) 0 < rA < 0.3 h−1, 0 < rB < 0.3 h−1, 0 < α < 0.1, 0 < β < 2. The optimal fit yielded \({k}_{{\rm{A}}}^{0}=0.38\,{{\rm{h}}}^{-1},\) \({k}_{{\rm{B}}}^{0}=0.14\,{{\rm{h}}}^{-1},\) rA = 0.56 h−1, rB = 0.01 h−1, α = 0.01, β = 1.3.
These parameters were subsequently used to infer \({k}_{{\rm{A}}}^{0}\) or \({k}_{{\rm{B}}}^{0}\) values for specific attP variants by fitting new measurements while holding other parameters constant. Corresponding estimates for left- or right-arm variants are summarized in Supplementary Table 1. The same framework was applied to estimate rA and rB for different promoters (Supplementary Table 2). Once condition-specific parameters (arm length, attP variant and promoter type) were determined, the model enabled prediction of Venus-to-mScarlet cell ratios across diverse configurations (Supplementary Table 3). All simulations were performed in Python (v.3.8.5).
Statistics and reproducibility
All experiments included at least three independent biological replicates (n ≥ 3). Data processing was performed using Excel (Microsoft Office 2019) and Origin 2019b (OriginLab). Error bars denote s.d., as specified in the figure legends, which also report replicate numbers. Two-group comparisons were done using the unpaired two-tailed Student’s t-test. P < 0.05 represents statistical significance (*P < 0.05, **P < 0.01, ***P < 0.001 and ****P < 0.0001; P ≥ 0.05, not significant).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.