Expression and purification of G protein heterotrimer
G protein heterotrimer was expressed as described14 in Tni cells (Expression Systems; isolated from Trichoplusia ni; no validation or mycoplasma testing) with viruses for Gα, Gβγ and Ric-8a and snap frozen in liquid nitrogen for later use. To purify wild-type G protein, cell pellets were thawed and resuspended in a lysis buffer containing 20 mM HEPES pH 7.5, 1 mM EDTA, 5% glycerol, 1 mM MgCl2, 5 mM β-mercaptoethanol, 100 µM GDP, Pierce Universal Nuclease, and protease inhibitor cocktail, and gently stirred for 30 min at 4 °C. The lysate was then subjected to ultracentrifugation at 100,000g for 35 min, and the supernatant was discarded. Pellets were resuspended in solubilization buffer containing 20 mM HEPES pH 7.5, 100 mM NaCl, 1% sodium cholate, 5% glycerol, 1 mM MgCl2, 5 mM β-mercaptoethanol, 100 µM GDP, Pierce Universal Nuclease, and protease inhibitor cocktail, and allowed to gently stir for 1 h at 4 °C prior to ultracentrifugation at 100,000g for 35 min. Solubilized protein was supplemented with 30 mM imidazole and incubated for 1 h with Ni-NTA beads. The beads were collected by centrifugation at 300g, packed into a column, and washed with 10 column volumes of a series of buffers containing 30 mM imidazole and 50% solubilization/50% E2 buffer, 25% solubilization/75% E2 buffer, 12.5% solubilization/87.5% E2 buffer, and 100% E2 buffer (E2 buffer contained 20 mM HEPES pH 7.5, 100 mM NaCl, 5% glycerol, 1 mM MgCl2, 5 mM β-mercaptoethanol, 100 µM GDP, and 0.05% lauryl maltose neopentyl glycol (LMNG)/0.005% cholesterol hemisuccinate (CHS)). Protein was eluted with 3 column volumes of buffer containing E2 buffer and 250 mM imidazole, and was incubated with 1 mg of 3C protease per 50 mg of protein with overnight dialysis at 4 °C in dialysis tubing. The next day, the Ni-NTA column was washed with 10 column volumes of buffer containing E2 and 30 mM imidazole, and overnight cleavage was loaded over the nickel and flow through was collected. The column was washed with an additional 2 column volumes of the E2 buffer and 30 mM imidazole, and flow through was collected. Protein was concentrated subjected to size-exclusion chromatography with a Superdex 200 column and a buffer containing 20 mM HEPES pH 7.5, 100 mM NaCl, 5% glycerol, 1 mM MgCl2, 100 µM TCEP ((Tris(2-carboxyethyl)phosphine)), 20 µM GDP, and 0.01% LMNG/0.001% CHS. Fractions were pooled, concentrated, and snap frozen in liquid nitrogen for later usage. For single-molecule experiments instead of cutting the His tag and performing reverse chromatography, the purified G protein heterotrimers were then mixed with biotinylated Tris-NTA (Sigma, supplemented with equimolar NiCl2) at a molar ratio of 2:3 and incubated on ice for 1 h. The G protein heterotrimer and biotinylated Tris-Ni2+-NTA complexes were subsequently purified using size-exclusion chromatography (Superdex200, Cytiva).
Expression and purification of NTSR1
Pellets containing NTSR1 were expressed in Sf9 cells (Expression Systems, no validation or mycoplasma testing) with the baculovirus system as described12 and snap frozen in liquid nitrogen for storage. To purify NTSR1, cell pellets were thawed and lysed in hypotonic lysis buffer containing 10 mM HEPES pH 7.5, 1 mM benzamidine, Pierce Universal Nuclease, protease inhibitor cocktail, 1 mM EDTA, 1 mM MgCl2, and 100 µM TCEP, and gently stirred for 1 h at 4 °C. Subsequently, membranes were collected by ultracentrifugation at 100,000g, the supernatant was discarded, and pellets containing the membranes were resuspended in solubilization buffer containing 20 mM HEPES pH 7.5, 500 mM NaCl, 1 mM benzamidine, Pierce Universal Nuclease, 1 mM MgCl2, 100 µM TCEP, and protease inhibitor cocktail. Detergent was then added dropwise while gently stirring at 4 °C to a final concentration of 1% LMNG/0.1% CHS/0.1% sodium cholate. After 3 h of stirring, insoluble debris was removed via ultracentrifugation at 100,000g, supplemented with 20 mM imidazole, and loaded over a TALON resin column. The columns were washed with 10 column volumes of buffer containing 20 mM HEPES pH 7.5, 20 mM imidazole, 500 mM NaCl, and 0.1% LMNG/0.01% CHS. Protein was eluted with 2 column volumes of buffer containing 20 mM HEPES pH 7.5, 250 mM imidazole, 250 mM NaCl, 10% glycerol, and 0.01% LMNG/0.001% CHS. Purified receptor was concentrated and subjected to size-exclusion chromatography with a Superdex 200 column and a buffer containing 100 mM NaCl, 20 mM HEPES pH 7.5, 5% glycerol, and 0.01% LMNG/0.001% CHS. Peak fractions were pooled, concentrated, and further supplemented with 5% glycerol, then snap frozen for use for complexation.
Formation and purification of NTSR1–G protein complex
Complex formation was initiated by mixing NTS8–13-bound receptor (NTSR1 incubated with NTS8–13 for 1 h) with a ~1.25 molar excess of G protein and incubating for 1 h on ice before addition of apyrase to remove GDP and HRV 3C protease to cleave the GFP tag from NTSR1 and incubation on ice overnight. Complex was then diluted in a buffer containing 100 mM NaCl, 20 mM HEPES pH 7.5, 10 μM NTS8–13, 0.01% LMNG/0.001% CHS, 5 mM CaCl2 and loaded onto M1 Flag resin, washed with a buffer of 100 mM NaCl, 20 mM HEPES pH 7.5, 10 μM NTS8–13, 0.005% LMNG/0.0005% CHS, 5 mM CaCl2 to remove excess G protein. NTSR1–G protein complex, and then eluted with a buffer of 100 mM NaCl, 20 mM HEPES pH 7.5, 10 μM NTS8–13, 0.005% LMNG/0.0005% CHS, 200 μg ml−1 Flag peptide and 1 mM EDTA. Eluted complex was concentrated and loaded onto a Superdex 200 size-exclusion column in a buffer of 100 mM NaCl, 20 mM HEPES pH 7.5, 10 μM NTS8–13, 0.001% LMNG/0.00033% GDN/0.0001% CHS, 2 mM MgCl2. Fractions of complex were concentrated to 6–10 mg ml−1 for cryo-EM studies.
Preparation of NTSR1–G protein complex in nanodisc
NTSR1–Gi1 complex was formed overnight as described in detergent, and then incubated for 1 h with solution containing MSP1D1 belt protein (Sigma) mixed with 3:2 POPC (1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine):POPG (1-palmitoyl-2-oleoylphosphatidylglycerol) at a 1:5:400 ratio of complex:MSP1D1:lipid. Two 1 h incubations with Bio-Beads SM-2 were performed followed by a final 8 h incubation with fresh Bio-Beads SM-2. NTSR1–G protein complex was then further purified as described for the detergent complex with Flag and size-exclusion chromatography, but in the absence of detergent. Nanodisc formation was confirmed with SDS–PAGE and concentrated to 2–4 mg ml−1 for cryo-EM studies.
Cryo-EM sample preparation and data collection
UltrAufoil R1.2/1.3 300 mesh grids were glow discharged in a Pelco unit for 45 s at 10 mA before being used for grid freezing. 3 μl of sample was pipetted onto a grid in a vitrobot held at 100% humidity and 4 °C, 0.3 μl of 10 mM GTP was added for the time-resolved cryo-EM grids, and the grids were blotted and plunge frozen in liquid ethane with a preprogramed blot and wait time to achieve 6 s and 20 s time points, chosen based upon prior studies with β2AR and MOR and consistent with the lifetimes observed in single-molecule based fluorescence assays. Detergent data were collected on a G4 Titan Krios equipped with a K3 direct electron detector and a Bioquantum energy filter. Details of each data collection can be found in Extended Data Tables 1 and 2 and Supplementary Table 2, but in brief, grids were imaged with a nominal defocus range from −0.6 to −1.6 μm and a target dose of 55 e− Å−2 over 40 total frames in super-resolution mode. Nanodisc data were collected on a G3 Titan Krios equipped with a Falcion IV/Selectris energy filter, with similar parameters to the detergent datasets.
Data processing
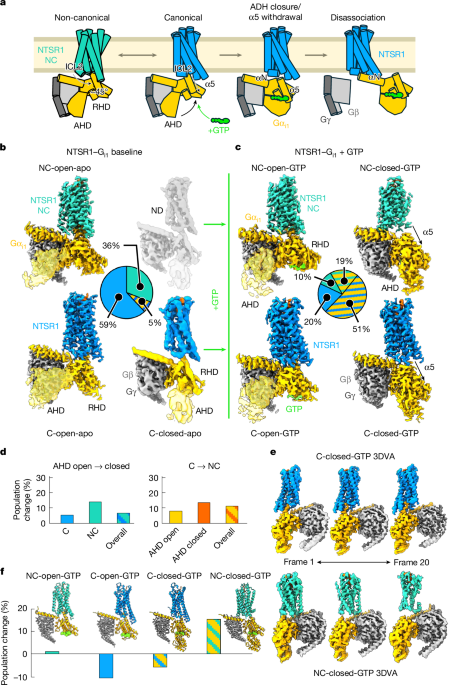
All data were processed in cryoSPARC45. Raw videos were aligned with patch motion correction and contrast transfer function (CTF) estimation was performed with patch CTF estimation. Particle picking was performed with template-based picking using templates for GPCR–G protein complexes. Extracted particles were subjected to two-dimensional classification to remove noise particles. Further cleaning was performed with iterative rounds of ab initio model generation with multiple classes and heterogeneous refinement. Once particle stacks were largely free of junk particles, non-uniform refinement was applied to obtain reconstructions. 3DVA was run on these reconstructions to probe for further heterogeneity, and the first principal component analysed. Final reconstructions came from non-uniform refinement, in cases of particularly high-resolution maps CTF refinement and reference-based motion correction were performed. In the case of the Gi1 NC-open-apo and Gi NC-open-GTP states, manual sharpening and preferred orientation correction, respectively, were applied to improve the map quality. Particle counts for comparing results were based upon the pre-orientation filtering numbers. A graphic example of the processing for the major experiments performed in this work is provided in Extended Data Fig. 1. Consistent with prior time-resolved cryo-EM studies of GPCR–G protein complexes, data from individual time points was merged to boost the resolution of reconstructions, and assessment of the time point-related changes in populations were based on whether particles contributing to final reconstructions came form 6 s or 20 s micrographs, although analysis of the individual time point data separately provided similar results (Extended Data Fig. 1d).
Model building
The prior cryo-EM structures of NTSR1–Gi1 were docked for all initial complex structures (PDB: 6OSA and 6OS9)12, while the Gi AHD closed state was docked from the prior MOR–Gi1 time-resolved work (PDB: 9ODL). Initial structures for G11 were obtained from crystal structures of the G11 heterotrimer (PDB: 8QEH)46. Manual model building was performed in Coot47 with refinement in Phenix48. Details of cryo-EM map and model refinement are found in Extended Data Tables 1 and 2. Euler angle distribution plots, FSC curves, local resolution plots, and selected map–model agreement panels are provided in Extended Data Figs. 7 and 8 and Supplementary Figs. 1 and 2.
Molecular dynamics simulations and analysis
GPCR–G protein complexes were prepared by extending the N-terminus of Gα, the C-terminus of the receptor, and the C-terminus of Gγ to add enough residues to include their sites of lipidation (if not already modelled) by using residual low-resolution map features for that respective state. The PPM webserver47 was used to orient systems which were then solvated in a box of POPC/CHS at a concentration of ~5% CHS (chosen to provide the presence of this important signalling molecule without significantly perturbing the bulk bilayer properties), TIP3P water49, 100 mM NaCl, and 15 mM MgCl2 using the CHARMM-GUI50. Simulations were performed in NAMD51 with the CHARMM36 forcefield52,53,54 using a Langevin thermostat and Nose-Hoover Langevin piston barostat at 1 atm with a period of 150 fs and decay of 75 fs. Periodic boundary conditions were used with nonbonded interaction smoothing at 10 Å to 12 Å with long-range interactions handled with particle mesh Ewald. A 2 fs timestep was employed with SHAKE and SETTLE algorithms used. All non-hydrogen, non-water/ion atoms were restrained with harmonic restraints of 1 kcal mol−1 Å−2 and the systems were minimized for 1,500 steps before gradual heating from 0 to 303.15 K in 20 K increments with 0.4 ns of simulation per increment, with an additional 10 ns of equilibration at 303.15 K. A further 10 ns of equilibration with harmonic restraints only applied to non-hydrogen protein atoms were performed, followed by another 10 ns of equilibration with harmonic restraints only applied to Cα atoms. The first 30 ns of unrestrained simulation was discarded from averaging as equilibration, with the following simulation considered production. All simulations were run with 5 replicates with different initial seeds for random assignment of velocities. VMD55 and Python scripting were employed for analysis. Distances for ICL2–β2–β3 were calculated from the centre of mass of residue F174 for NTSR1 and V175 for MOR and K192 of Gi1, while TM6-α4 distances were calculated based upon residue 6.26 in Ballesteros–Weinstein notation and residue D309 of Gi1. AHD–RHD distances were calculated based on the centre of mass of the two domains.
Kinetic BRET assays
HEK 293 cells (ATCC, revalidated by STR and mycoplasma negative) were transfected in 6-well plates with plasmids encoding a GPCR (NTSR1, MOR or variants), Gα subunit (Gαi1 or Gαq), Venus1-155Gγ2, Venus155-239Gβ1 and memGRKct-Nluc at a ratio of 1:0.8:0.4:0.4:0.1 μg per well using linear PEI. For activation of Gq heterotrimers PTX-S1 (0.2 μg) was also transfected to prevent activation of endogenous Gi/o heterotrimers. For activation of Gi/o heterotrimers 1 μM YM-254890 was added to prevent activation of endogenous Gq heterotrimers. Kinetic BRET assays were performed 24 h after transfection using a BMG Lumistar plate reader equipped for simultaneous measurement of donor and acceptor emission. Cells were resuspended in DPBS containing 5 μM furimazine, and NTS8–13 (1 μM final concentration) or DAMGO (10 μM) were injected after collecting a 10 s baseline. NTSR1 assays were performed in triplicate with technical quadruplicates while MOR assays were performed in quadruplicate with technical quadruplicates. ICL2 swaps exchanged the nine residues from 34.51 to 4.39 while ICL3 swaps exchanged the residues from 5.68 to 6.25.
Fluorophore labelling of wild-type NTSR1
Purified wild-type NTSR1 was mixed with iodoacetamide-derivatized LD555 (custom synthesis by Lumidyne technologies) at a molar ratio of 1:0.3 and incubated at room temperature for 1 h. The labelling reaction was quenched by L-cysteine (10 mM). The excess dye was removed by purifying the labelled NTSR1 on a G50 desalting column (2 ml).
Single-molecule fluorescence experiments
Single-molecule imaging experiments were performed using a custom-built prism-based total internal reflection fluorescence microscope, as described56. The single-imaging platform used is based upon a Nikon Ti2 inverted microscope equipped with sCMOS cameras (Kinetix; Teledyne Vision Solutions), a super-resolution water-immersion objective lens (Nikon CFI SR Plan Apo IR 60× 1.27 NA WI), a continuous wave 6 W 532 nm excitation laser (Laser Quantum Opus) coupled to the observation chamber via a custom-fabricated quartz prism (Eksma).
Nucleotide-free NTSR1–G protein complexes were prepared by incubating dye-labelled receptors with purified complexes of G protein and biotinylated Tris-Ni2+-NTA at a molar ratio of 5:4, in the presence of saturating NTS8–13, for 5 min at room temperature to promote GDP release. The reaction mix was then incubated with Apyrase (NEB; 5 U ml−1) for an additional 10 min at room temperature to remove the free GDP. The nucleotide-free complexes were then surface immobilized within (1,3) trans-divalent streptavidin57-coated microfluidic devices prepared by passivating quartz slides and coverslips with polyethylene glycol (PEG) and biotin–PEG followed by capping treatment with sulfo NHS acetate58.
All experiments were performed in imaging buffer (20 mM HEPES, pH 7.5, 100 mM NaCl, 2 mM CaCl2, 5 mM MgCl2, 0.01%LMNG/0.001% CHS), supplemented with an enzymatic oxygen scavenging system consisting of 2 mM protocatechuic acid, 50 nM protocatechuate-3,4-dioxygenase. For NTSR1 dissociation measurements, imaging buffer with GTP (1 mM) was rapidly introduced 1 s after the start of the data acquisition, using a custom-built computer-controlled microfluidic system58. All single-molecule fluorescence data were recorded in stroboscopic mode under low 532 nm laser illumination intensity (6.5 W cm−2), at a frame rate of 5 s−1, with an interval of 2 s using custom microscope control software written in LabVIEW (National Instruments) and synchronizing the microfluidic system (controlled by Cetoni Elements software) with the imaging system via TTL triggering58. Fluorescent particles were counted using SPARTAN56.
Control experiments were performed by injecting buffer without GTP to measure the intrinsic rate of fluorophore photobleaching and spontaneous dissociation. These data were fit to a single exponential decay function. The rates of GTP-induced dissociation of NTS8–13-bound, labelled NTSR1 from G proteins were estimated by plotting the loss of fluorescent particles over time and fitting the data to a triple exponential decay function, where one component corresponds to the intrinsic rate of photobleaching and dissociation. The aggregated rate of dissociation was calculated by the weighted average contributions of fast (A1/t1) and intermediate (A2/t2) decay phases.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.