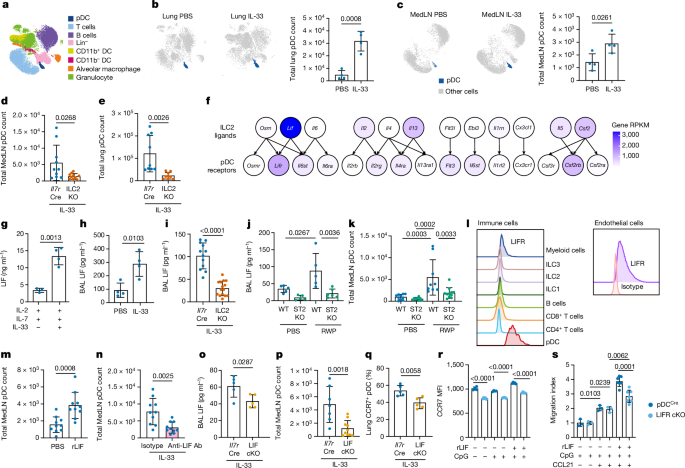
Mice
All mice were maintained in the Medical Research Council ARES animal facility under specific-pathogen-free conditions at 19â23â°C and 45â65% humidity with a 12/12âh light/dark cycle. In individual experiments, mice were matched for age, sex and background strain. All experiments undertaken in this study were performed with the approval of the LMB Animal Welfare and Ethical Review Body and the UK Home Office. C57BL/6âJOla controls were bred in house. Mouse strains Il7rCre (ref. 56), Roraflox/flox (ref. 57), Il1rl1â/â (ref. 58), Rag2â/â, Rag2â/âIl2rgcâ/â (Rag2â/âgcâ/â), Lif flox/flox, BIC33, SiglechCre (ref. 59) and Lifrâ/flox were either on the C57BL/6J Ola background or back-crossed for at least six generations.
Generation of Lif
flox/flox mice
To produce a Lif allele that could be conditionally deleted by Cre recombinase, we generated a homology-directed repair-template construct for use in combination with CRISPRâCas9 to insert LoxP sites 5â² and 3â² of the final exon of both protein-coding annotated Lif transcripts (ENSMUSE00000656154). In addition, the construct included a neomycin selection cassette flanked by Frt sites to permit Flp-mediated excision, and both LoxP sites were followed by a BglII site to facilitate screening and verification of appropriately targeted embryonic stem cell clones (Extended Data Fig. 1r). Embryonic stem cells were transfected with this repair-template construct along with expression constructs for WT Cas9 and four single-guide RNAs, two targeting sequences 5â² and two targeting 3â² of the final Lif exon. Neomycin-resistant clones were screened for correct targeting initially by PCR and digested with BglII using 5â² primer pairs P1 and P2 such that a product cleaved by BglII indicated correct targeting. Clones were further verified by Southern blot analysis using 5â² and 3â² probes that both detected a 16.4âkb fragment in the WT allele and 4.9 and 7.6âkb fragments, respectively, in the targeted allele (Extended Data Fig. 1s). Guide RNA target sequences were: G1 (F) TAATGATTCTAGTTGCCTACAGG; G2 (F) TGGAGTCCCCATGTCACAGGTGG; G3 (F) TTCCTCCATCGGTCCAGGAGGGG; G4 (R) TACCCCTCCTGGACCGATGGAGG. Screening primers were: P1 TAGGAAGCCAGAGTCTAGTGGCAGTTTTAAGAGATGG; P2 AAGGCTTCTTTGTCAGAGTGGTCGG. Primers for generation of probes were: 5â²âprobeâ1 fwd CCTGCCACCCCCTTAACCTCCATAAGTGAAAAGCAAGTGG; 5â²âprobeâ1 rev. ACTGGGCCTGCTAGGGGTTTGACAG; 3â²âprobeâ1 fwd TGATGGAGCTGTGGGATGGG; 3â²âprobeâ1 rev. ACACACTCGGGCTCCATTATGC.
Generation of LIFR conditional mice
For generation of pDC-specific LIFR knockout (SiglecHCre/Cre Lifrâ/flox) mice, pDCCre (SiglecHCre/Cre) mice were crossed with Lifrâ/flox to delete exonâ5 of Lifr (transcript variantâ1). Both Tg(Siglech-Cre,-mCherry)59 and Lifrtm1a(EUCOMM)Hmgu (ref. 25) mice were obtained from the European mouse mutant archive. To delete the lacZâneomycin-resistance cassette and generate mice with a loxP-flanked Lifr allele (EuComm Lifrtm1c is denoted as Lifrflox in this paper), chimaeras were bred to FLPe C57BL/6 mice. However, we also detected inefficient Lifrflox allele recombination and consequently analysed pDCCreâÃâLifrâ/flox mice in which two Cre-mediated recombination events occurred to produce LIFR deficiency, with pDCCreâÃâLifrâ/+ mice as controls. Despite two Cre alleles and one functional flox allele, pDCCreâÃâLifrâ/flox mice showed a reduction in LIFR expression of only 50% in pDCs (Extended Data Fig. 1t).
Mouse challenge protocols
IL-33-induced typeâ2 lung inflammation
Mice were challenged intranasally with IL-33 (Biolegend; 0.25âµg in 40âµl of PBS) on 3 consecutive days. All tissues were harvested 24âh following the final dose.
Neutralizing LIF antibody treatment
Mice were intranasally challenged with IL-33 (0.25âµg in 40âµl of PBS) and intraperitoneally injected with 100âµg of either isotype antibody (R&D systems) or anti-LIF neutralizing antibody (R&D systems) on 3 consecutive days. All tissues were harvested 24âh following the final dose.
rLIF intranasal challenge
Mice were intranasally challenged with rLIF (R&D systems; 1âµg in 40âµl of PBS) on 3 consecutive days. All tissues were harvested 24âh following the final dose. For the pDC migration kinetics experiment, mice were intranasally challenged with one rLIF dose (1âµg in 40âµl of PBS) and tissues harvested at 6 or 24âh after challenge.
RWP-induced typeâ2 lung inflammation
Mice were intranasally challenged with RWP (300âμg of protein per dose, Ambrosia artemisiifolia, short form; Greer Laboratories) on 3 consecutive days. All tissues were harvested 24âh following the final dose. For the chronic lung inflammation model, mice were intranasally challenged with RWP thrice weekly over 5âweeks. All tissues were harvested on dayâ38.
FITC-dextran-labelled cell migration
Mice were challenged intranasally with IL-33 (Biolegend, 0.25âµg) and 40âkDa FITC-dextran (Sigma-Aldrich, 40âµg in 50âµl of PBS) on 3 consecutive days. All tissues were harvested 24âh following the final dosing.
Mouse infection models
PVM infection
Mice were infected intranasally with a single dose of PVM (50âPFU in PBS). PVM strain J3666 stock was a gift from A. J. Easton. All tissues were harvested at 8âdays postinfection unless stated otherwise. For the CCL21 kinetics experiment, mice were intranasally challenged with PVM (50âPFU in PBS) and tissues harvested on 0, 4, 8 and 11âdays postinfection.
For PVM rechallenge, mice were challenged with PVM (50âPFU in PBS) on days 0 and 30 and all tissues were harvested on dayâ38.
FITC-dextran-labelled cell migration
Mice were challenged intranasally with PVM (50âPFU) and 40âkDa FITC-dextran (Sigma-Aldrich, 40âμg in 50âµl of PBS). All tissues were harvested 3âdays postinfection.
rLIF and PVM challenge
Mice were infected with a single dose of PVM (50âPFU in PBS) intranasally and treated with a daily dose of intranasal rLIF(1âμg) or PBS. All tissues were harvested on dayâ8 postinfection.
Mice were infected with a single dose of PVM (50âPFU in PBS) intranasally and, on dayâ8, postinfection intravenous CD45 labelling was performed by injection of 3âμg of anti-CD45 antibody (in 200âμl of PBS) via the tail vein. Mice were then culled 3âmin after injection and tissues harvested.
FTY720 and PVM challenge
Mice were infected with a single dose of PVM (50âPFU in PBS) intranasally and injected intraperitoneally daily with either FTY720 (ref. 34) (25âμg in 250âµl; Enzo Life Sciences) or PBS. All tissues were harvested on dayâ8 postinfection.
Tissue processing
BAL isolation
Mice were culled at the experimental endpoint, tracheae were exposed and BAL was performed by flushing the lungs three times with 0.5âml of PBS. The fluid obtained was centrifuged at 350g for 5âmin; supernatants were stored at â20â°C for cytokine detection.
Serum isolation
Mice were culled at the experimental endpoint and whole blood was collected. Blood samples were allowed to clot for 2âh at room temperature. Samples were centrifuged at 2,000g for 10âmin and serum was collected and stored at â20â°C. For immunoglobulin enzyme-linked immunosorbent assay (ELISA), serum was diluted 1/50.
Viral load
Mice were culled at the experimental endpoint, and one lung lobe was snap-frozen in trizol (Invitrogen) and stored at â80â°C for RNA purification.
Tissue preparation
Lung tissue was predigested with 750âUâmlâ1 collagenaseâI (Gibco) and 0.3âmgâmlâ1 DNaseâI (Sigma-Aldrich) before obtaining a single-cell suspension at 37â°C for 30âmin; tissue was then passed through a 70âμm cell strainer. For lymphocyte enrichment, a single-cell lung suspension was centrifuged through 30% Percoll (GE Healthcare) at 800g for 15âmin. Spleen, thymus and mediastinal LN single-cell suspensions were prepared by passing tissue through a 70âμm cell strainer and lysing red blood cells. Single-bone marrow cell suspensions were prepared by flushing the femur and tibia with endotoxin-free PBS and lysing red blood cells.
Flow cytometry
Single-cell suspensions were incubated with fluorochrome- or biotin-conjugated antibodies in the presence of anti-CD16/CD32 antibody (Fc block, clone 2.4G2), followed by fluorochrome-conjugated streptavidin where necessary. All samples were costained with a cell viability dye (Fixable dye eFluor780, Invitrogen) and analysed on either a 5-5-laser LSRFortessa system (BD Biosciences, BD FACSDiva software v.6.2) or spectral cytometer ID7000 (Sony Biotechnology). Either FACSAria Fusion systems or iCyt Synergy (70âμm nozzle, Sony Biotechnology) was used for cell sorting. Precision Count Beads (BioLegend) were used to calculate cell numbers. Intracellular transcription factor staining was performed using the Foxp3 staining kit (eBioscience) according to the manufacturerâs instructions. For lymphocyte intracellular cytokine staining, cells were cultured with complete RPMI supplemented with Cell Stimulation Cocktail or protein transport inhibitors (eBioscience) for 4âh at 37â°C. Intracellular cytokine staining was performed using BD Cytofix/Cytoperm Plus reagents (BD Biosciences) following the manufacturerâs instructions. The expression of LEC CCL21 was detected by additional staining with goat anti-mouse CCL21 (R&D systems) and anti-goat-Alexaâ488 (Invitrogen), with no stimulation, and using BD Cytofix/Cytoperm Plus reagents (BD Biosciences) following the manufacturerâs instructions.
For intracellular phospho-STAT3 staining, cells were fixed with 2% paraformaldehyde (PFA) for 15âmin and overnight permeabilization with 90% methanol at â20â°C, followed by incubation with fluorochrome antibodies diluted in 2% bovine serum albumin PBS.
Flow cytometric analysis, including unsupervised dimensionality reduction and clustering, was performed using FlowJo, LLC v.10 (BD) and associated plug-ins. Unless otherwise stated, pDCs are defined as LiveCD45+CD11bâF4/80âCD317+SiglecH+. Myeloid cells include cDCs and CD11bâ cDCs as LiveCD45+CD11bâCD11chighSiglecFâMHCIIhigh; CD11b+ cDCs as LiveCD45+CD11câ/intm SiglecFâLy6GâCD19âTCRbâMHCIIhigh; monocytes as LiveCD45+CD11b+ SiglecFâLy6GâF4/80+MHCIIâ; and alveolar macrophages as CD45+CD11c+F4/80+SiglecF+. Eosinophils are defined as CD45+CD11câF4/80âCD11b+Gr1int SiglecF+; and neutrophils as CD45+CD11câF4/80âCD11b+Ly6GhighSiglecFâ. T cells are defined as LiveCD45+TCRb+ and Bâcells as LiveCD45+CD19+TCRbâ; CD4+ Tâcells as CD45+CD3+CD4+ and ILC2s as CD45+Linâ(CD3,CD4,CD8,CD19,CD11b,CD11c,FcεR1) CD127+ICOS+. Endothelial cells are defined as LiveCD45âCD31+, LECs as LiveCD45âCD31+PDPN+ and BECs as LiveCD45âCD31+ PDPNâ.
All flow cytometry data were processed and analysed using FlowJo v.10, RRID: https://scicrunch.org/resolver/SCR_008520.
In vitro cultured cells
Lung immune cell sorting
Mouse ILC2s were purified from IL-33-treated lungs (see Methods for IL-33-induced typeâ2 lung inflammation) as LiveCD45+LineageâIL-7Rα+ST2+KLRG1+; and pDCs were purified from IL-33-treated lungs (see Methods for IL-33-induced typeâ2 lung inflammation) as LiveCD45+F4/80âCD11bâCD317+SiglecH. Cells were snap-frozen in trizol for RNA purification, and conditioned medium was collected and stored at â20â°C.
Mouse lung ILC2s and Tâcells for qPCR analysis were purified from PVM-challenged mice using the same gating strategy as for ILC2s: LiveCD45+LineageâIL-7Rα+ST2+KLRG1+; CD4+ Tâcells as LiveCD45+TCRb+CD4+; CD8+ Tâcells as LiveCD45+TCRb+CD8+; BECs as LiveCD45âCD31+PDPNâ; and LECs as LiveCD45âCD31+PDPN+. Purified cells were snap-frozen in trizol for RNA purification.
ILC2 in vitro stimulation
Purified ILC2s were cultured for 24âh with IL-7 (10ângâmlâ1) and IL-2 (50ângâmlâ1) with or without IL-33 (10ângâmlâ1). Cells for RNA purification and conditioned media were collected and stored at â20â°C.
pDC culture, purification and activation
Bone marrow cells were obtained by flushing femurs and tibias with RPMI, followed by incubation with red blood cell lysis buffer for 5âmin. Following washing, cells were cultured with RPMI containing 10% fetal calf serum, 1% penicillin/streptomycin, sodium pyruvate, non-essential amino acids, l-glutamine, β-mercaptoethanol and Flt3L (10ângâmlâ1) for 7â10âdays. Medium was refreshed on days 3 and 6. pDCs were sorted from bone marrow-derived cultures by fluorescent activated cell sorting as LiveCD45+CD11cintSiglecH+CD317+ cells. Purified pDCs were activated with CpG (6âμgâmlâ1, Invivogen) and treated with or without rLIF (500ângâmlâ1) for 24âh.
Chemotaxis assay
Migration assays were performed using Millicell cell culture inserts (Merck Millipore) with 2â3âÃâ105 cells per well. Purified pDCs were activated with CpG (6âμgâmlâ1) and treated with or without rLIF (10ângâmlâ1) for 24âh. Activated pDCs were placed in inserts with 5âμm pores for 3âh in the presence or absence of cytokine rLIF (500 or 1000 ng mlâ1) or chemokine rCCL21 (R&D systems) at 150ângâmlâ1. The number of migrating cells was then evaluated using a flow cytometer. The results are expressed as migration index (number of migrating cells in chemokine/number of migrating cells in medium).
Lung LEC purification and culture
The preparation of single-cell suspensions from lung tissues is described in âTissue preparationâ. CD31+ lung cells were isolated from lung cell suspension using magnetic beads (CD31 biotin, Streptavidin dynabeads)60. Isolated CD31+ lung cells were seeded onto 0.2% gelatin-coated, six-well plates and cultured on complete growth medium. consisting of ECGS (Corning), 20% fetal bovine serum, 1% penicillin/streptomycin, sodium pyruvate, non-essential amino acids and 25 mM HEPES, in a humidified incubator with a gas mixture of 21%âO2 and 5%âCO2 at 37â°C until 70â80% confluence was achieved (usually reached in 4â7âdays). Endothelial cells were detached with Accutase (Stemcell Technologies) and purified using magnetic beads (podoplanin biotin, Streptavidin dynabeads). Purified LECs were seeded onto 0.2% gelatin-coated, six-well plates, cultured in complete growth medium and used for experiments.
In vitro LEC treatment
Isolated LECs were treated with or without rLIF for 6 or 24âh, detached using Accutase and stained for flow cytometry analysis.
ELISA and MAGPIX Luminex Array
Culture supernatant was collected and stored at â20â°C until analysis. Serum IgE was measured by ELISA (Invitrogen). LIF, IL-5, type I IFN, CCL19, CCL21, CCL25, CXCL9 and CXCL10 were measured using ProcartaPlex kits (Invitrogen).
Virus neutralization assay
Sera from PVM-rechallenged mice were diluted in a 1:10 ratio and heat-inactivated at 55â°C for 30âmin. An equal volume of PVM at 500âPFU per well concentration (1:20 final serum dilution) was incubated with serum for 1âh at 37â°C and 5% CO2. BHK-21 cell monolayers (1âÃâ105 per well) were infected with the virus mixture and incubated for 72âh at 37â°C and 5% CO2. Before harvesting the cells were washed three times with PBS, snap-frozen in Trizol (Invitrogen) and stored at â80â°C for RNA purification.
qPCR with reverse transcription
RNA was purified using Direct-zol RNA Purification Kits. For assessment of viral load, frozen tissue samples were homogenized before RNA purification. Complementary DNA synthesis was performed using SuperScriptâIV Reverse Transcriptase and oligo d(T)20 (Invitrogen). PVM viral load was tested with forward primer 5â²-GCCTGCATCAACACAGTGTGT and reverse primer 5â²-GCCTGATGTGGCAGTGCTT38 in a SYBR green qPCR assay. For lung samples, the mouse HPRT gene was used as an internal control. For the PVM neutralization assay, the dCT for viral amplification was measured with respect to the hamster GAPDH gene. For other qPCR analyses, commercially available Taqman gene expression assays (Applied Biosystems; Extended Data Table 1) were used. Samples were run on the ViiA7 real-time PCR system (Applied Biosystems).
RNA-seq
Cells were sorted by flow cytometry into PBS and 50% fetal calf serum. and RNA was extracted using the RNeasy Plus Micro kit (Qiagen). Following assessment using a Bioanalyser (Agilent), RNA was processed for RNA-seq using Ovation RNA-seq System v.2 (Nugen), fragmented by a Covaris M220 ultrasonicator and bar-coded using Ovation Ultralow Library Systems (Nugen). Samples were sequenced using an Illumina HiSeqâ4000 by running a single-read 50-base-pair protocol (Cancer Research UK, Cambridge Institute). Sequence data were trimmed to remove adaptors and sequences with a quality score below 30 using Trim Galore (v.0.50, Babraham Bioinformatics) and then aligned to the mouse genome (GRCm38) using STAR (v.2.6.0a); differential expression was calculated using DESeq2 (v.1.18.1).
Bioinformatic identification of candidate ligands and receptor pairs
Gene lists for cytokines and cytokine receptors were obtained by downloading Gene Ontology gene lists for âCytokine Activityâ and âCytokine Receptor Activityâ from the Mouse Genome Informatics website. The curated mouse CellTalkDB database of ligandâreceptor pairs was used to identify interacting gene pairs between these gene lists61. Using Râprogramming language, the dplyr package was utilized to filter the CellTalkDB database by the cytokine and cytokine receptor gene lists to remove non-cytokine-related ligandâreceptor pairs. This filtered list of ligandâreceptor pairs was then used to interrogate bulk RNA-seq data of pDCs and ILC2s isolated from mouse lung. Expression of cytokine ligandâreceptor pairs in which expression of the receptor by pDCs was greater than 10âRPMK and expression of the ligand by ILC2s was greater than 10âRPKM was then extracted. Ligandâreceptor pairs involving Cd44 or Cd74 were excluded from the analysis due to the high expression levels of these transcripts.
Histology
Tissue was fixed in 10% formalin overnight and paraffin embedded; sections were stained with haematoxylin and eosin. Lung histology sections were assessed by a researcher blinded to groupings and given a score between 0 and 5 based on the presence or absence of large cellular aggregates.
Microscopy
Mice were euthanized and received intracardiac perfusion with PBS, followed by 4% PFA (Invitrogen). Lung and LN were collected and fixed with 4% PFA overnight. Fixed tissues were washed with PBS and placed in 30% sucrose for 24âh. Subsequently these were embedded in Optimum Cutting Temperature compound (VWR, catalogue no. 25608-930), frozen in Isopentane and sectioned on a Leica CM1860 cryostat. Sections were incubated in a blocking solution (2% goat serum and 0.5% Triton X-100 in PBS) for 1âh at room temperature. Tissue sections were then incubated overnight at 4â°C with primary antibodies against CD3e, B220 and VEGFR3, then with secondary antibodies for 1âh at room temperature. Images were acquired with an Olympus VS200 slide scanner and processed and analysed using ImageJ2 v.2.14.0/1.5âf.
Lung histology sections were assessed for TLS by a researcher blinded to groupings and given a score between 0 and 5 based on the pathology.
Data and statistical analyses
Statistical analysis was performed using GraphPad Prism v.10.0b software.
Bulk RNA-seq data generated in this study have been deposited at the Gene Expression Omnibus (GEO) under accession number GSE243691.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.