Synthesis of buildings blocks, insulin conjugate NNC2215 and control compound NNC2215a
Detailed synthesis of NNC2215 and the control compound NNC2215a is described in the Supplementary information and in Extended Data Figs. 7â9. In brief, desB30 human insulin was conjugated at B29 to O-succinimidyl-pentyn-1-oxycarbonyl at pHâ>â10, and at B1 to O-peracetyl-d-glucosyl-β-ethylenoxy-acetic acid using Br CF3 sulfonate phenolic active ester at pHâ7.5. The acetyl groups were removed by gentle saponification. The macrocycle reagent was made from 3-(3,5-dimethylphenyl)propanoic acid by benzylic bromination, azidation, reduction of COOH to alcohol, and the azides were reduced to the amines, which were Boc-protected. The propyl alcohol was activated as the mesylate and transformed to the propyl azide. The three Boc-amino groups were transformed to the isocyanates using triflic anhydride, and the macrocycle was closed by reaction of the isocyanates with the previously described trisamino half-macrocycle reagent4. The roof azide macrocycle was triazole-coupled under Cu(i) catalysis with the given B29-alkyne insulin to give NNC2215, which was purified using high-performance LC. Chemical characterization is shown in the Supplementary information and Supplementary Data.
Glucose affinity of the free macrocycle propyl azide by calorimetry
The concentration of the free macrocycle (Extended Data Fig. 8i) in the cell was 50âμM. The concentration of glucose added in the syringe was 3âmM. The injection volume was 10âμl. The stirring speed was 310ârpm; all in 10âmM phosphate buffer, pHâ7.4 at 25â°C. The obtained Ka was 10,200âMâ1, and the Kd was 98âµM. Data were acquired on the MicroCal VP-ITC microcalorimeter and processed using the MicroCal software (MicroCal VP-ITC Analysis Add-On Software Package 7.20 for ORIGIN 7.0).
Affinity of NNC2215 towards glucose by native MS
Glucose solutions (18.75âµM, 37.5âµM, 75âµM, 150âµM, 300âµM, 625âµM, 1.25âmM, 2.5âmM, 5âmM, 10âmM, 20âmM) in presence of NNC2215 (0.5âmgâmlâ1) were prepared in 75âmM NH4Ac, pHâ7.4. The compounds were buffer-exchanged into 75âmM NH4Ac, pH7.4 by Amicon ultracentrifugal devices with 3,000âDa molecular weight cut-off filters. Direct infusion using a standard ESI probe was performed on a UPLCâESI-MS Synapt G2-S (Waters) system with a UPLC flow-through needle. No column was used, and the system was configured to allow sample flow directly from the sample manager to the electrospray ionization probe. Positive ionization mode was used and the samples were sprayed using a flow of 30âµlâminâ1 and an injection volume of 20âµl. The capillary voltage was 1.2âkV, and the source temperature was 85â°C, while the desolvation temperature was 80â°C. A Genedata workflow was used to quantify the ratio bound using the intensity (TIC) of the bound glucose complex versus the unbound form, that is, Mâ+â1 glucose/(Mâ+â(Mâ+â1 glucose)). The ratio bound (%) versus the glucose concentration (mM) was plotted using GraphPad Prism (GraphPad). The apparent glucose equilibrium Kd was determined from the binding curves by fitting the data with a one-site total-binding model.
3D modelling studies
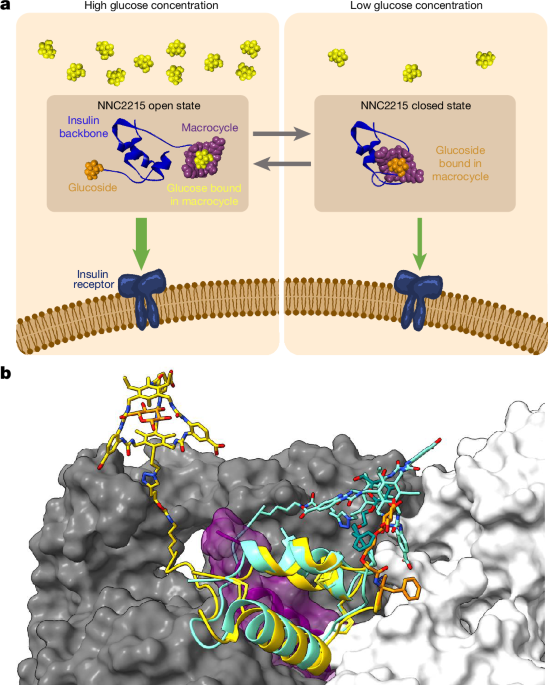
The structural models of NNC2215 were built using Maestro (Schrödinger release 2020-3)33. The macrocycle model was adapted from the models shown previously4 and connected to insulin at B2926 with the corresponding linker. The glucoside was built modifying B1. For the open configuration, the modifications were applied to the receptor-bound insulin in PDB 6PXV (chain E20), with a glucose molecule bound in the macrocycle as in the previous model4. For the closed conformation, insulin from PDB 2VJZ (ref. 21) was modified, building the glucoside bound in the macrocycle based on the same macrocycleâglucose complex model described above. Both models underwent further refinement and energy-minimization steps within Maestro. For the obtained closed state, we additionally ran a molecular dynamics simulation in explicit solvent to test the stability of the conformation. The OPLS3e forcefield was used for models of NNC2215 bound to glucose, which were solvated water molecules based on simple point charge (SPC) model in sodium-neutralized simulation boxes. A real-space cut-off of 9âà for Coulombic interactions was used throughout. Standard Desmond preparation and relaxation protocols were used33, gradually warming up the system and releasing restrained heavy atoms, consisting of initial 100âps in NVT ensemble with Brownian dynamics at 10âK with a 1âfs timestep and 12âps in Langevin dynamics with a Berendsen thermostat, followed by two subsequent equilibration runs of 12âps in NPT ensemble at 1âatm with a Berendsen barostat first at 10âK and then at 300âK, and finally 24âps at 300âK in the same NPT ensemble with full release of heavy atom restraints. The final molecular dynamics production run consisted of 100âns in NPT at 1âatm and 300âK, using the MartinaâTobiasâKlein method. The final coordinates of both open and closed models were superimposed onto the bound insulin in PDB 6PXV (chain E) to show how the closed conformation would hinder the interaction with the insulin receptor. Figure 1b was generated using ChimeraX34.
In vitro biology
hIR-A and IGF-1R affinity measurements
The following materials were used: SPA PVT antibody-binding beads, anti-mouse reagent (GE Healthcare, RPNQ0017), HSA (Sigma-Aldrich, A1887, SLCBR2530) and antibodies hIR 83.7 and IGF-1R 24-31 (produced at Novo Nordisk, licensed from K. Siddle35,36).
BHK cells overexpressing hIR-A or IGF-1R were lysed in 50âmM HEPES pHâ8.0, 150âmM NaCl, 1% Triton X-100, 2âmM EDTA and 10% glycerol. The cleared cell lysate was batch absorbed with WGA-agarose (Lectin from Triticum vulgaris-Agarose, L1394, Sigma-Aldrich) for 90âmin. The receptors were washed with 20 volumes 50âmM HEPES pHâ8.0, 150âmM NaCl and 0.1% Triton X-100, after which the receptors were eluted with 50âmM HEPES pHâ8.0, 150âmM NaCl, 0.1% Triton X-100, 0.5âM n-acetyl glucosamine and 10% glycerol37. All buffers contained cOmplete protease inhibitor mixture (Roche Diagnostic).
Binding studies were performed with dilution series of ligands in 100âmM HEPES, 100âmM NaCl, 10âmM MgSO4 and 0.025% (v/v) Tween-20, pHâ7.4, with or without 1.5% HSA in the presence of 0, 3, 5, 10 or 20âmM d-glucose in triplicates for each dilution in 96-well Isoplates (PerkinElmer, 6005049), 5,000âCPM Tyr A14-125I-insulin, 25âµl SPA beads, 25âng IR 83-7 antibody, 0.006âµl hIR-A per well. After 22âh incubation at 22â°C, the bound radioactivity was quantified by counting in a Microbeta2 2450 Microplate counter (Perkin Elmer), essentially as described previously37.
IGF-1R-binding assays were conducted essentially as for the IR binding assays, except that solubilized IGF-1Rs, [125I-Tyr31]-human IGF-1 and the IGF-1R-specific antibody 24-31 were used as previously described38.
The IC50 and relative affinities to human insulin were calculated. Each point in the competition curve was a measure of triplicates with the mean and s.d. The IC50 of the one-site binding model was fitted with a nonlinear regression algorithm using GraphPad Prism. The top, bottom and slope were set to be equal for all of the compounds in each experiment. As the logarithm cannot be calculated for zero, the concentration without unlabelled ligand was set to 1âÃâ10â14âM in the calculations. On each plate, human insulin was included for calculation of the relative affinities of NNC2215 and insulin degludec for each set of plates. The average and s.d. for three independent experiments were calculated using Excel. The fold changes in the relative affinity to hIR-A were calculated from 0 or 3âmM glucose to 20âmM glucose in each experiment and the average and s.d. were calculated.
hIR phosphorylation and AKT and ERK assays
In three independent experiments, CHO-hIR cells39 were stimulated with increasing concentrations of human insulin and NNC2215 (0-1âµM) for 10âmin. After stimulation, cells were homogenized in lysis buffer. Insulin receptor phosphorylation was measured in technical duplicate with the InsR(pY1158) Thermo Fisher Scientific ELISA kit according to the manufacturerâs instructions. Phosphorylation of AKT and ERK were measured in technical duplicate using AlphaScreen, SureFire AKT1/2/3 (p-Ser473) and SureFire ERK1/2 p-T202/Y204 Assay Kits from PerkinElmer according to the manufacturerâs instructions and a previous study39.
Lipogenesis in primary mouse adipocytes
In primary rat mouse adipocytes isolated from epididymal fat pads, the effect of NNC2215 and insulin degludec on lipogenesis was determined by measuring the incorporation of [3H]-labelled glucose into fat as described previously27 with a slight modification: l-glucose was added to a final concentration of 3âmM or 20âmM.
In vivo studies
Animal experiments were performed under a license granted by the Danish national Animal Experiments Inspectorate and complied with relevant ethical regulations on animal research.
l-Glucose rat model
Non-fasted healthy male SpragueâDawley rats (aged 11 weeks; 380â430âg) were dosed i.v. with NNC2215 or insulin degludec (4.5 nmolâkgâ1) at timepoint zero. Then, 30âmin later, the rats received an additional i.v. dosing of vehicle or 0.5, 1 or 2âg kgâ1 l-glucose (nâ=â7 per group). Blood was drawn from the sublingual vein into EDTA Eppendorf tubes at timepoints 0, 25, 35, 45, 60, 90 and 120âmin and immediately centrifuged for quantification of plasma d-glucose concentrations by the glucose oxidase technique (Biosen, EKF Diagnostics) and NNC2215 or insulin degludec plasma concentrations. The rats had no access to food during the experiment.
Hypoglycaemia study in pigs
Female LYD pigs aged approximately 18 weeks and weighing between 74.5 and 86.5âkg on the day of the first dose were used. Body weight increased to 99â110âkg on the day of the last dose. Before the experiments, all of the animals were instrumented with two venous catheters, one for infusion and one for sampling. Fasting glucose was measured before each experimental day and ranged from 4.43 to 6.23âmM. Animals were subjected to constant i.v. infusions of somatostatin (1âµgâkgâ1âminâ1)âto suppress endogenous insulin and glucagon secretionâand glucagon (0.45âpmolâkgâ1âminâ1) to replace the suppressed glucagon secretion. Suppression of insulin secretion was verified by measuring C-peptide levels, which documented that endogenous insulin secretion was below basal levels throughout the experiment despite plasma glucose being elevated (Extended Data Fig. 3c). Primed constant infusions of NNC2215 or insulin degludec and infusion of glucose were given as indicated below. Suitable priming doses of each insulin analogue were estimated based on their pharmacokinetics after i.v. administration.
The duration of the experiment was 540âmin. At time 0, infusions of hormones and glucose were started. Glucose was infused at 6âmgâkgâ1âminâ1 from 0 to 360âmin after which it was turned off for 90âmin and restarted at 6âmgâkgâ1âminâ1 for the last 90âmin of the experiment. To investigate a range of plasma glucose concentrations before turning off the glucose infusion, different insulin infusion rates were used (NNC2215: 1.30, 1.44, 1.58, 1.72, 1.86, 2.00 and 2.14âpmolâkgâ1âminâ1; insulin degludec: 0.7, 0.9 and 1.1âpmolâkgâ1âminâ1). The insulin infusion rates were chosen with the aim of having comparable glucose concentrations for NNC2215 and insulin degludec before turning off the glucose infusion. Owing to the limited experience with the glucodynamic potency of NNC2215, a broader dose range was tested for NNC2215 than for insulin degludec. The lower infusion rates of NNC2215 were tested at the beginning of the study and the higher rates were tested at the end of the study. Samples for glucose determination were drawn into EDTA Eppendorf tubes. The samples were then immediately centrifuged, and the plasma analysed using the glucose oxidase technique (YSI 2900, YSI).
s.c. and i.v. pharmacokinetic study of NNC2215 in pigs
Seven female LYD pigs aged approximately 22 weeks and weighing between 93.0 and 97.5âkg were used. Before the experiment, the animals were instrumented with a venous catheter for i.v. injection of NNC2215 and for blood sampling. Four animals were injected s.c. in the neck area with 2ânmolâkgâ1 NNC2215 (600ânmolâmlâ1) and three animals received i.v. bolus administration of 0.3ânmolâkgâ1 NNC2215. Blood samples were drawn into EDTA Eppendorf tubes and immediately centrifuged for quantification of plasma glucose concentrations by the glucose oxidase technique (Biosen; EKF Diagnostics) and NNC2215 plasma concentrations.
Pharmacokinetic/pharmacodynamic models
Methodology on the pharmacokinetic/pharmacodynamic modelling used to quantify the change in insulin activity of NNC2215 with changing plasma glucose based on data from the hypoglycaemia study in pigs is provided in the Supplementary information.
Glucose challenge study in diabetic rats
Healthy male SpragueâDawley rats (aged approximately 9â10 weeks; 350â400âg) were surgically instrumented with permanent arterial (blood sampling) and venous (insulin and glucose infusions) catheters under isoflurane anaesthesia and recovered from surgery with analgesia (carprofen, 5âmg per rat per day, s.c.) for 7â8âdays before being made acutely diabetic with streptozotocin (65âmgâkgâ1, s.c.). Then, 3â4 days after streptozotocin treatment, the rats were fasted overnight and subjected to one of three different constant i.v. infusion rates of either NNC2215 (42, 84, 100âpmolâkgâ1âminâ1; primed, see below) or human insulin (20, 25, 30âpmolâkgâ1âminâ1) to lower plasma glucose from â¥15âmM to a predefined target of 5.7âmM. Appropriate priming doses of NNC2215 to obtain steady state in plasma exposure within the duration of the study was calculated using its pharmacokinetics after i.v. administration in rats. When the glucose target was reached, it was clamped by adjusting the i.v. GIR. When the GIR had been at steady state for at least 30âmin, a GTT was carried out by infusing an additional 25âmgâkgâ1âminâ1 of glucose for 60âmin on top of the individual steady-state GIR without clamping the plasma glucose. Blood for measurement of plasma glucose concentrations was drawn from the arterial catheter into heparinized Eppendorf tubes and immediately centrifuged for 30âs. Plasma glucose was measured every 10âmin throughout the study by the glucose oxidase technique (YSI 2900). To mimic the effect of a glucose-sensitive insulin and to quantify its extra effect during the GTT, another group of streptozotocin diabetic rats was given 50% additional human insulin on top of the constant human insulin infusion (20â+â10âpmolâkgâ1âminâ1; human insulin +50%) during the GTT. The maximal plasma glucose concentrations during the GTT were compared using one-way analysis of variance and pairwise comparisons by Tukeyâs post hoc test.
Quantification of insulin and C-peptide concentrations in plasma samples
The plasma samples were analysed for levels of NNC2215, insulin degludec and human insulin using luminescence oxygen channelling immunoassay (LOCI)/Alpha-LISA, which is a homogeneous immunoassay method without washing steps. In the LOCI/Alpha-LISA assay, streptavidin-coated donor beads are used (Alpha-LISA donor beads, Perkin Elmer) in addition to acceptor beads (Alpha-LISA acceptor beads, Perkin Elmer), which were conjugated to a monoclonal antibody specific for the insulin analogues of interest (NNC2215, insulin degludec or human insulin). A second monoclonal antibody recognizing another part of the insulin analogues of interest was biotinylated and used in the LOCI/Alpha-LISA assay. The antibody pairs used for determination of the three analytes were as follows: NNC2215 (monoclonal antibody HUI-018 conjugated to acceptor beads and biotinylated polyclonal antibody GP a-HI 4080-E; when measuring NNC2215 in pig plasma, unmarked monoclonal antibody OXI-005 was added to prevent measurement of endogenous pig insulin; when measuring in rat plasma, unmarked monoclonal antibodies anti-rat insulin 1 and anti-rat insulin 2 were added to prevent measurement of endogenous rat insulin); insulin degludec (monoclonal antibody NN454-1F31 conjugated to acceptor beads and biotinylated monoclonal antibody S1); human insulin in rat plasma (monoclonal antibody HUI-018 conjugated to acceptor beads and biotinylated monoclonal antibody OXI-005). All of the indicated antibodies were generated in-house at Novo Nordisk. A dilution row of NNC2215, insulin degludec or human insulin was prepared as calibrators in species-specific plasma pools in appropriate concentration ranges. The calibrator curves for each of the insulin analogues were used for the quantification of the specific analytes in unknown samples. Plasma samples containing the analytes of interest were incubated with antibody-coated acceptor beads in addition to the described biotinylated monoclonal antibody (Bio-mAb) in 384-well plates. After 1âh (insulin degludec and human insulin) or 24âh (NNC2215) of incubation, the streptavidin-coated donor beads were added to the wells. The acceptor beads, the analyte of interest, the Bio-mAb and the donor beads all form a complex within the solution. Illumination of the complex releases singlet oxygen atoms from the donor beads. These are channelled into the acceptor beads and trigger a chemiluminescence response, which is measured in an Envision plate reader (Perkin Elmer). The amount of light is proportional to the concentration of the analyte. The lower limit of quantification was determined to be 42 pM in LYD pig and 27 pM in rat for NNC2215, 15 pM in LYD pig for insulin degludec and 2.6 pM in rat for human insulin.
Plasma samples were analysed for pig C-peptide content using LOCI/AlphaLISA technology as described above for quantification of insulin concentrations in plasma samples. During the assay, a concentration-dependent beadâanalyteâimmune complex is created, resulting in light output, which is measured on the Perkin Elmer Envision reader. In the assay, anti-pig C-peptide monoclonal antibody M-grC-pe-1F34A1-conjugated acceptor beads and biotinylated monoclonal antibody 4F16A6 (also raised against pig C-peptide) were used together with generic streptavidin-coated donor beads. Both antibodies were made in house. The lower limit of quantification was 45 pM.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.