Zebrafish care and use
Zebrafish were maintained in the Queen’s Medical Research Institute BVS Aquatics Facility at the University of Edinburgh with approval from the UK Home Office according to their regulations under the following project licences: 70/8436 and PP5258250, or the Vollum Institute zebrafish facility at Oregon Health and Science University (OHSU) in accordance with institutional ethical regulations and approved by the Institutional Animal Care and Use Committee at OHSU. Adult zebrafish were maintained by aquatics staff under standard conditions on a 14 h light, 10 h dark cycle. Zebrafish embryos were maintained at 28.5 °C in 10 mM HEPES buffered E3 embryo medium or in conditioned aquarium water with methylene blue. Larval zebrafish were analysed between 4–24 dpf, before sex is determined. Zebrafish larvae used for imaging experiments beyond 7 dpf were maintained at 28.5 °C in an incubator with a 14 h light, 10 h dark cycle from 1–8 dpf before being moved to tanks without water flow on a 14 h light, 10 h dark cycle until 24 dpf. In these longitudinal experiments, larvae were provided live rotifers at all times they were not embedded for imaging beyond 5 dpf (further details below).
Zebrafish stable line generation
The Tg(uas:myrEGFP-P2A-Lifeact-TagRFP) stable line was generated using Gateway cloning by combining pDestTol2PA2, p5E-10x-UAS, pME-myrEGFP and p3E-P2A-Lifeact-TagRFP. The pTol2PA2-UAS:myrEGFP-P2A-Lifeact-TagRFP plasmid (30 pg) was injected into single-cell zygotes with 12.5 pg of Tol2 transposase mRNA to generate F0 fishes. The p3E-P2A-Lifeact-TagRFP entry vector was generated by amplifying Lifeact-TagRFP from the sox10:Lifeact-TagRFP plasmid25 with XhoI and SpeI overlap extension PCR primers and inserted into the p3E-P2A-MCS entry vector by restriction cloning.
In vivo confocal microscopy in zebrafish
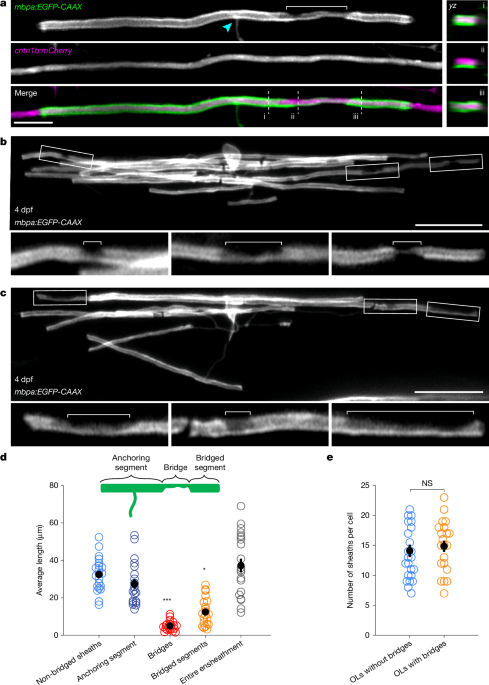
To fluorescently label the myelin sheaths of single oligodendrocytes, fertilized zebrafish eggs were injected at the single-cell stage with 1 nl containing 10 pg pTol2-mbp:EGFP-CAAX, or 35 pg each of pTol2-mbp:EGFP-CAAX and pTol2-cntn1b:mCherry plasmid DNA48,64 and 25–50 ng µl−1 Tol2 transposase mRNA. In activity-dependent experiments, botulinum neurotoxin was included in the injection solution at 100 ng µl−1 as reported previously65. Zebrafish were screened to identify isolated fluorescently labelled oligodendrocytes from 3 dpf. To screen for fluorescently labelled oligodendrocytes, larval zebrafish were first anaesthetized with MS222 before mounting them in 1.5% low melting point agarose on glass coverslips. Once zebrafish were anaesthetized and mounted, oligodendrocytes in the spinal cord were selected for imaging. z-Stacks of oligodendrocytes were acquired using an LSM 880 confocal microscope with Airyscan fast mode with a 20× objective (Zeiss W Plan-Apochromat, NA = 0.8). Or using an LSM 980 with Airyscan superresolution mode with a 20× objective (Zeiss W Plan-Apochromat, NA = 1.0). z-Stacks were acquired with an optimal z-step for each experiment.
For longitudinal imaging of individual zebrafish from 3–24 dpf, Tg(sox10:KalTA4) adults were crossed to F0 or F1 Tg(uas:myrEGFP-P2A-Lifeact-TagRFP) adults to sparsely label oligodendrocyte lineage cells. Larvae anaesthetized with Tricaine at 3 dpf were mounted in 0.8% low melting agarose on a Petri dish and maintained in embryo medium with a low Tricaine concentration (<153 µM) and moved to an LSM 980. OPCs or newly generated oligodendrocytes (not present or existed as OPC in previous day) were imaged every 2 h until 7–8 dpf. Embryo medium was exchanged with fresh medium (lacking Tricaine) for at least 1–2 h each day before replacing Tricaine. Every other day, each larva was fully extracted from agarose and allowed to swim freely in a well of a 12-well plate with embryo medium and live rotifers for at least 2 h before being re-embedded for imaging to continue. Additional static images of the same larvae were acquired at 16 dpf and 24 dpf.
Zebrafish image analysis
For single time point analysis at 4 dpf, the number and lengths of myelin sheaths and paranodal bridges were quantified with the segmented line tracing tool in Fiji. Oligodendrocytes were analysed throughout the depth of each z-stack per cell. No cells were excluded from analyses unless there was too much myelin overlapping from neighbouring cells to reliably quantify myelin sheaths and paranodal bridges. One oligodendrocyte was analysed per zebrafish unless otherwise specified in figure legends. For long-term imaging experiments, both the green channel containing myr-eGFP signal and red channel containing Lifeact-TagRFP signal were first brightness adjusted using the Fiji Bleach Correction tool using the simple ratio function across the entire time course. Tracing of sheaths and Lifeact signal was done using the SNT plugin. Relative distance (difference in y coordinate between traces) of Lifeact signal to either the bottom or top edge of the sheath was calculated in MATLAB. The hypothetical triangular function was created with an increasing and decreasing linear array between 0.1 and 1. Cumulative sum of Lifeact movement up to 25 h post peak sheath was normalized in y from 0 to 1 with the rescale function, and in x to 101 points with imresize. For ease of viewing in plots, values for unsampled time points were filled linearly with the fillmissing function, though mixed effects models used only existing values to determine statistical significance. Linear mixed effects models used the following formula: cumulative_movement ~ time*group + (1|cell_ID) + (1|cell_ID:sheath_ID), where group consisted of anchoring segment, bridge, or bridged segment and random effects of cell ID or sheath ID were accounted for in the nesting structure. Software used for all image quantification and analysis are listed in Supplementary Table 4.
Paranodal bridge identification and quantification
In the zebrafish transgenics used, membrane-targeted eGFP denotes the outsides of sheaths, forming a tube of labelled membrane around the axon. These ensheathed regions are clearly distinguishable from processes and cell bodies, especially with sparse-labelling approaches, allowing successful reconstruction of full morphologies48. In zebrafish experiments, bridged ensheathments were thus defined as complete tubes (eGFP signal fully around an unlabelled axon—that is, a circle in cross-section) that were connected via thinner eGFP signal (partial circle in cross-section) to a complete tube with a direct connection to an oligodendrocyte process.
In mice, paranodal bridges were identified within in vivo images during reconstruction of oligodendrocyte morphology as performed previously18. Traces were initiated on primary processes at the soma, with additional traces continuing on subsequent branches. With cytoplasmic labelling in the Mobp-EGFP mouse line, cytoplasmic process intersection with myelin sheaths can be determined by both the sudden change in angle between the process and sheath as well as an increase in diameter. Sheaths are also morphologically distinct, as compacted myelin leaves parallel cytoplasmic channels within the sheath (inner and outer tongues) and bright accumulation of fluorescence at the cytoplasmic-rich paranodal loops. These identification methods have been validated previously with SCoRe imaging as well as MBP immunolabeling to isolate sheaths from the rest of oligodendrocyte morphology4. Thus, bridged sheaths were defined during tracing as a sheath (internode flanked by bright punctae at paranodes) with: (1) no cytoplasmic process connected; and (2) thin cytoplasm across a NOR connected to at least one adjacent sheath. Oligodendrocyte morphology in human myelinoids was quantified in the same way, as CNPase immunohistochemistry is confined to cytosolic channels and shows similar organization as observed in Mobp-EGFP mice.
All data are represented with mean ± standard error unless otherwise noted.
Mouse care and use
All experiments involving mice were conducted in strict accordance with protocols approved the Animal Care and Use Committee at Johns Hopkins University, in compliance with federal regulations. Female and male adult Mobp-EGFP mice were used for experiments and randomly assigned to experimental groups. All mice were healthy and did not display any overt behavioural phenotypes. Mice were maintained on a 12 h:12 h light:dark cycle, food and water were provided ad libitum, and housed in groups no larger than 5. Mice were housed with at least one other cage mate when possible. Three Mobp-EGFP mice were aged to 1.5 years before being implanted with cranial windows for our ageing experiment. Cuprizone experiments were performed as previously described18. Powdered standard mouse diet was mixed with 0.2% bis(cyclohexanone) oxaldihydrazone (cuprizone, Sigma C9012) and provided to mice in custom feeders in their home cages for three weeks, refreshed at least twice per week. Control mice received powdered chow without cuprizone. After three weeks, both cuprizone-treated and control mice were returned to normal pellet diet.
Mouse cranial windows
Cranial window surgeries were performed as described previously18. In brief, Mobp-EGFP mice were deeply anaesthetized with isoflurane (5% at 1 l min−1 O2 induction, 1.5–2% at 0.5 l min−1 O2 maintenance) and their scalps shaved and cleaned. A portion of the scalp was removed, and the underlying skull was cleaned and dried before cementing (Metabond) on a custom aluminium headplate. A 3-mm circle of skull was removed with a high-speed dental drill and replaced with a coverslip that was secured in place with VetBond and Krazy Glue. Mice recovered in their home cage on a heating pad and were monitored for at least 1 h. Mice were imaged two to three weeks following window surgery.
In vivo two-photon microscopy in mice
In vivo imaging was performed as described previously18. In brief, Mobp-EGFP mice were deeply anaesthetized under isoflurane (5% at 1 l min−1 O2) and then transferred to a custom stage on a Zeiss 710 microscope and clamped in place by their headplate where they remained under isoflurane (1.5–2% at 0.5 l min−1 O2 maintenance) for the remainder of the imaging session. Two-photon images were collected on a Zeiss LSM 710 or Zeiss LSM 880 microscope with a GaAsP detector and mode-locked Ti:sapphire laser (Coherent Ultra) tuned to 920 nm (Mobp-EGFP mice) or 1,000 nm (Mobp-EGFP; PV-cre; Ai9 mice) with average power at the sample <30 mW. A Zeiss coverslip-corrected 20× water-immersion objective (NA 1.0) was used to acquire 2,048 × 2,048 pixel (425 µm × 425 µm) stacks from the pia to depths of 110 µm or 230 µm (1-µm z-step).
To quantify bridge frequency on PV axons, axons in layer II/III were fully traced using SNT in ImageJ from in vivo images of Mobp-EGFP; PV-cre; Ai9 mice. Myelin sheaths surrounding these axon traces were traced, including any paranodal bridges and cytoplasmic processes leading to oligodendrocyte cell bodies. Sheaths were labelled as either being anchoring, bridged, or undefined and bridges were labelled as either spanning a branch or not.
Mouse cortical flatmount preparation
Cortical flatmount preparation was performed as described previously2. In brief, deeply anaesthetized mice (100 mg kg−1 w/w sodium pentobarbital) were transcardially perfused with 20–25 ml warm (30–35 °C) PBS followed by 20–25 ml ice-cold 4% paraformaldehyde. Cortical mantles were dissected from the underlying brain structures, unrolled, placed between two glass slides separated by 1 mm, and post-fixed in 4% paraformaldehyde at 4 °C for 6–12 h. Flattened cortices were removed from the clamped slides and stored in 30% sucrose in PBS for at least 24 h until sectioned on a cryostat (Thermo Scientific Microm HM 550) at –20 °C at thicknesses of 35–50 µm. Cryostat chucks were pre-frozen with TissueTek mounting medium and sectioned until flat. Flatmounts were removed from sucrose solution, covered with mounting medium dorsal-side down on a silanized glass slide, and frozen onto the prepared chuck. Care was taken to ensure complete horizontal sections were acquired by aligning the blade angle to the surface of the tissue.
Mice used for nodal component immunostaining were perfused only with 20–25 ml warm (30–35 °C) PBS. Brains were dissected and lightly fixed for 30–60 min in 4% paraformaldehyde (PFA). Flatmounts were lightly post-fixed in 4% PFA for 60 min in the clamped slide configuration. Flatmounts continued to be maintained in the clamped slide configuration during 30% sucrose incubation for at least 24 h before sectioning.
Mouse immunohistochemistry
Immunohistochemistry on mouse brain was performed on free-floating tissue sections preincubated in blocking solution (5% normal donkey serum, 0.3% Triton X-100 in PBS, pH 7.4) for up to 2 h at room temperature, then incubated for 24–48 h at 4 °C or room temperature in primary antibodies. Sections were subsequently washed in PBS before being incubated in secondary antibodies at room temperature for 2–6 h or overnight at 4 °C. Sections were mounted on slides with Aqua Polymount (Polysciences). Specific antibodies used are listed in Supplementary Tables 2 and 3.
Brainbow tamoxifen recombination, tissue processing and imaging
Tamoxifen (Sigma) was dissolved in sunflower oil (Sigma) to achieve a concentration of 10 mg ml−1. At 6 weeks of age, Mobp-creER; Brainbow(+/-) (https://www.jax.org/strain/017492) mice were injected intraperitoneally with a single dose of tamoxifen at 100 mg kg−1. Animals were perfused transcardially 2.5 weeks post-injection with 4% PFA, flatmounts prepared as described above, and fixed overnight before being transferred to 30% sucrose solution. Endogenous signal was weak and made accurate bridge quantification challenging. Fluorophores were thus enhanced by immunostaining. Nuclear GFP, cytoplasmic YFP, and membrane-tethered mCerulean were amplified with a chicken anti-GFP primary antibody (Aves), and cytoplasmic RFP (tdimer2(12)) was amplified with either an anti-RFP nanobody conjugated to Alexa 546 or a rat anti-mCherry primary antibody. Although by this method GFP, YFP, and mCerulean were excited by a 488 nm laser, their identities could be ascertained by their distinct cellular localization as well as excitation of weak endogenous mCerulean (445 excitation). Additionally, most cells observed only had a single fluorophore, as expected from heterozygous Brainbow mice. Images were acquired on a Zeiss LSM 980 using a water-immersion 40× objective (NA 1.2) with Airyscan SR. SCoRe imaging and AnkG staining additionally verified that identified paranodal bridges passed nodes and linked compacted sheaths.
Mouse image processing and analysis
Image registration, processing and tracing of oligodendrocyte morphologies was performed as described previously18. High-resolution imaging of individual bridged nodes was performed on a Zeiss 800 or 880 in Airyscan mode. Regions were ~45 µm × 45 µm in xy with a resolution of ~1,800 × 1,800 pixels. z-Stacks ranged in depth but had z-steps of 0.18 µm. For analysis of degenerating sheaths in aged mice, individual regions acquired with a 20× objective were subdivided into quadrants of 212 µm × 212 µm × 100 µm volumes prior to beginning analysis. Quadrants that had overlying blood vessels, bone or thickened meninges during the course of imaging were excluded from analysis. Loss of individual sheaths was detected in syGlass volumetrically by observing 10–20-µm-thick slices at a time and continuously rotating through time points. Lost sheaths were verified in ImageJ and their identities (bridged or non-bridged) were then determined. The density of myelination and abundance of lipofuscin at these ages were substantial, preventing accurate tracing of full morphologies of individual oligodendrocytes. However, we were able to distinguish bridged sheaths by their lack of intersecting cytoplasmic process between paranodes. Neighbouring sheaths that degenerated simultaneously were considered the anchoring sheath of the bridged pair.
Electron microscopy analysis
For all mouse electron microscopy quantification, we used the publicly available 250 µm × 140 µm × 90 µm electron microscopy volumetric dataset of a P36 mouse visual cortex layer II/III (Pinky)30, acquired at a resolution of 3.58 nm × 3.58 nm × 40 nm. Additional example reconstructions were taken from the 1.4 mm × 0.87 mm × 0.84 mm dataset of P87 visual cortex (Minnie). These datasets, as part of the Machine Intelligence from Cortical Networks (MICrONS) programme (https://microns-explorer.org/), underwent automated cytosolic segmentation. All segmentation fragments of putative paranodal bridges or nascent sheaths identified in the datasets were manually validated to confirm cytoplasmic continuity and meshes were corrected in VAST or Fiji. Not all myelin sheaths were fully contained within the bounds of the volume. When possible, lack of a direct cytoplasmic process of one of the sheaths connected by paranodal bridge was confirmed by following the entirety of the outer tongue between paranodes of each sheath. Paranodal bridges identified in Pinky may be found at the coordinates listed in Supplementary Table 1. We also used a volumetric electron microscopy dataset of roughly 4 myotome segments (74 µm × 74 µm × 207 µm) of a 6 dpf zebrafish larval spinal cord at 9 nm × 9 nm × 21 nm resolution29 and a dataset acquired from a sample of human temporal cortex at 4 nm × 4 nm × 33 nm resolution47. Data were visualized with KNOSSOS and nascent ensheathments were separately reconstructed with VAST. NORs in a human serial electron microscopy data of temporal cortex47 were manually identified within layers II–IV by paranodal loops flanking bare axon segments <5 µm in length. To quantify the proportion of these nodes that were bridged, paranodal loops were visually traced to determine whether there was cytoplasmic continuity between sheaths flanking the node.
Human organoid generation and processing
The human pluripotent stem cell-lines used in this study were obtained with full Ethical/Institutional Review Board approval by the University of Edinburgh and validated using standard methods including chromosomal analysis, pluripotency and absence of plasmid integration. The iPSC lines CS02iCTR-NTn1 (male) and CS25iCTRL-18n2 (male) were obtained from Cedars-Sinai and the embryonic stem cell line SHEF4 (male) was obtained from the UK Stem Cell Bank. The maintenance of human pluripotent stem cells and generation of myelinoid cultures has been described recently40. In brief, cells were maintained in Essential 8 medium before being lifted into suspension and patterned towards the pMN domain of the developing spinal cord. Spheroids containing ventral, caudal neuroepithelial cells were then patterned towards a glial cell fate using PDGF-AA before being transferred onto PTFE-coated Millicell Cell Culture Inserts (Merck) and maintained until cultures were 19 weeks old (corresponding to MI-12 in ref. 40).
Myelinoids were fixed in 4% PFA, washed, then permeabilized in 0.25% Triton X-100 in PBS for 40 min and blocked in 10% normal goat serum (Vector Laboratories) + 0.25% Triton X-100 for 2 h at room temperature. For CNP immunostaining, myelinoids were incubated in citrate buffer (pH 6) at 95 °C for 20 min followed by a further 1 h in blocking solution. Primary antibodies rat anti-MBP, mouse anti-CNP and rabbit anti-CASPR were incubated overnight at 4 °C in blocking solution. After washing in PBS (3 × 20 min), secondary antibodies (goat anti-rat, goat anti-mIgG2b and goat anti-rabbit) were incubated for 2 h at room temperature in blocking solution. Myelinoids were stained with DAPI, washed in PBS and whole-mounted onto microscope slides (Thermo Scientific) with FluorSave (Calbiochem) and No. 1.5 coverslips (Thermo Scientific). Images were captured using a Zeiss 710 confocal microscope and analysed in Fiji using the Cell Counter and SNT plugins for counting cells and tracing myelin sheath lengths, respectively.
Human postmortem brain tissue
Post-mortem brain tissue (motor cortices) from people without neurological defects were provided by a UK prospective donor scheme with full ethical approval from the UK Multiple Sclerosis Society Tissue Bank (MREC/02/2/39) and from the MRC-Edinburgh Brain Bank (16/ES/0084). The clinical history was provided by R. Nicholas and C. Smith. Supplementary Table 5 provides the details of the samples that were used in the study. The mean age of the human tissue donors was 68.5 years. Tissue blocks of 2 cm × 2 cm × 1 cm were collected, fixed, dehydrated and embedded in paraffin blocks. Four-micrometre sequential sections were cut and stored at room temperature.
Immunohistochemistry on human tissue
Paraffin sections were rehydrated, washed in PBS and microwaved at high power for 15 min in Vector Unmasking Solution for antigen retrieval (H-3300, Vector Laboratories). The sections were then incubated with Autofluorescent Eliminator Reagent (2160, MERCK-Millipore) for 1 min and briefly washed in 70% ethanol for 5 min. Image-iT FX Signal Enhancer (I36933, Thermo Fisher Scientific) was subsequently applied for 30 min at room temperature, and then the sections were washed and blocked for 1 h with 10% normal horse serum, 0.3% Triton X-100 in PBS. Primary antibodies were diluted in antibody diluent solution (003118, Thermo Fisher Scientific) and sections were incubated overnight at 4 °C in a humidified chamber. The next day the sections were incubated with Alexa Fluor secondary antibodies for 90 min at room temperature, counterstained with Hoechst 33342 (62249, Thermo Fisher Scientific) for the visualization of the nuclei and mounted using Mowiol mounting medium (475904, MERCK- Millipore). z-Stack images were acquired from layers 2 and 3 of the human primary motor cortex with Leica TCS SP8 confocal microscope using a 63× objective. From each sample up to 14 different regions of ~62 µm × 62 µm in xy with a resolution of ~2,048 × 2,048 pixels and a system’s optimized z-step were acquired and the average percentage of bridged NORs were quantified.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.