Chemists have synthesized a carbon-based molecule with an unprecedented ‘half-Möbius’ twist.Credit: IBM Research and the University of Manchester
The Möbius strip, a twisted loop with one continuous surface, is inspiring chemists to create novel molecular structures. A team has synthesized a type of carbon-based molecule with an unprecedented type of twist, which they call ‘half-Möbius’.
To make a regular Möbius strip, one end of a ribbon is flipped by 180º compared to the other and the two are joined together. This object is named after the eighteenth-century German mathematician August Ferdinand Möbius who first described its properties.
What’s peculiar about the half-Möbius molecule is that a chain of atoms is twisted by only 90º to make the loop, instead of a full 180º. The results are described in a Science1 paper published on 5 March.
“90º is fun,” says study co-author Igor Rončevic, a theoretical chemist at the University of Manchester, UK, because it can twist either left or right. The two versions differ in what chemists call chirality — meaning that, like left and right-handed gloves, they are distinct from their mirror images. “The system knows whether you are twisting clockwise or anticlockwise,” Rončevic says.
“This is a tremendous achievement,” says Gemma Solomon, a theoretical chemist at the University of Copenhagen.
“To my knowledge, this is the first molecule of this kind,” says Rainer Herges, a chemist at Christian-Albrecht University in Kiel, Germany, who led the synthesis2 of the first Möbius-strip molecule in 2003.
Do the twist
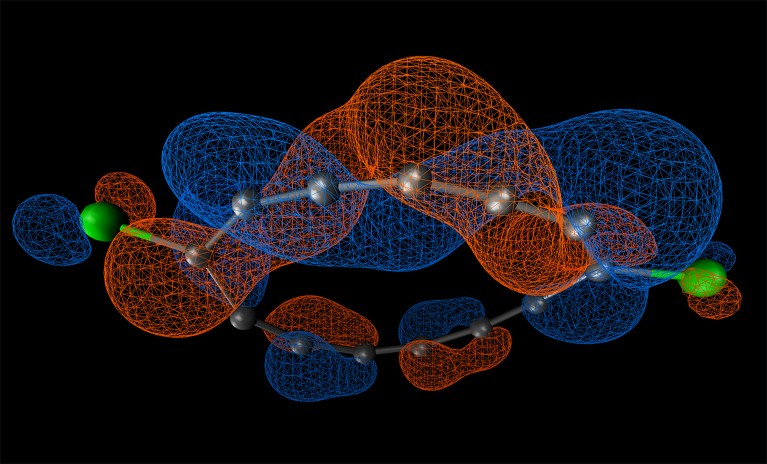
In the latest paper, the team created a loop of 13 carbon atoms, two of which, at opposite sides of the circle, are bound to a chlorine atom. This leaves 11 carbon atoms that link directly to their neighbouring carbons. They also each have two electrons whose orbitals are barbell-shaped and stick out, perpendicular to the loop.
These barbells form additional bonds with their neighbours, creating what chemists call a ‘conjugated’ structure, in which the electrons become shared among many atoms in the chain. (Various types of conjugated structures are responsible for some of the unusual properties of aromatic molecules such as benzene and of two-dimensional materials such as graphene.)


