Mice
Six-to-eight-week-old female and male C57BL/6 J mice (IMSR_JAX:000664), as well as other strains, were purchased from The Jackson Laboratory. OT-II (IMSR_JAX:004194) and Thy1.1 (IMSR_JAX:000406) mice were crossed to generate OT-II Thy1.1 mice. Foxp3-GFP mice (IMSR_JAX:006772) were crossed with OT-II Thy1.1 mice. 2D2 mice (IMSR_JAX:006912) were crossed with Thy1.1 mice. All animals were housed in AAALAC-accredited facilities. Sample sizes were not predetermined but are reported with each result, and randomization was performed across littermates.
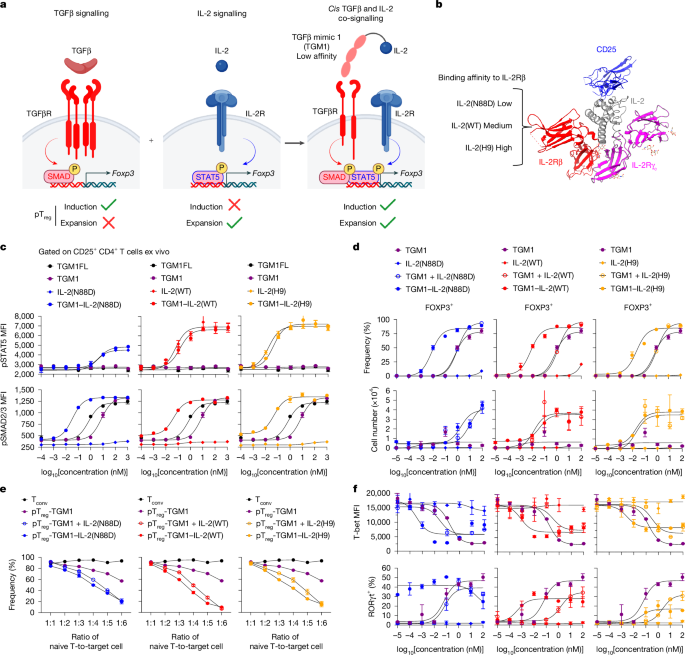
Flow cytometry
The following antibodies were purchased from BioLegend: mouse CD45.2 (109839), CD3 (100206), CD4 (100453, 100430, 100428, 100451), CD8 (100706), NK1.1 (156506), TCRvα2 (127806, 127822), TCRvα3.2 (135404), Thy1.1 (202528, 202522), CTLA4 (106310), CD62L (104453), CD25 (102012, 102022, 102047, 102038), CD44 (103026, 103032), SIGLECF (155534), CD73 (127215), ICOS (313550), CD69 (104530), CD11b (101259), CXCR3 (126514), CD39 (143806), NRP1 (145218), CD11c (117318), CD103 (110910), GITR (126316), CXCR6 (151117), CCR6 (129819), IL-17A (506928), GM-CSF (505406), IFNγ (505832, 505826), IL-10 (505034, 505026, 505034), BLIMP1 (150008), Helios (137214), Ki-67 (151212, 652406), TNFα (506346); human CD3 (317324), CD4 (980806) and FOXP3 (320126). The following antibodies were purchased from BD Biosciences: mouse BCL6 (562401), RORγt (564722, 562682, 562683), SMAD2 (pS465/pS467)/SMAD3 (pS423/pS425) (562696) and STAT5 (pY694) (612599). The following antibodies and reagents were purchased from Invitrogen: mouse PD-1 (48-9985-82), FOXP3 (12-5773-82, 17-5773-82, 404-5773-82), T-bet (25-5825-82), c-MAF (53-9855-82), GATA3 (46-9966-42) and Fixable Viability Dye (65-0865-18). PE- and Brilliant Violet 421-labelled I-Ab OVA328–337 tetramers (HAAHAEINEA) were provided by the NIH Tetramer Core Facility.
For surface marker staining, live cells were incubated with antibodies and viability dye in PBS at 4 °C for 1 h. Dead cells were excluded on the basis of viability dye staining. For I-Ab OVA328–337 tetramer staining, live cells were first incubated with tetramers in PBS at 37 °C for 1 h, followed by surface antibody and viability dye staining. For transcription factor and cytokine staining, cells were fixed and permeabilized using the FOXP3/Transcription Factor Staining Buffer Set (00-5521-00, Invitrogen), followed by intracellular staining at room temperature for 2 h. Prior to cytokine staining, cells were stimulated with Cell Stimulation Cocktail (00-4970-03, Invitrogen) and Protein Transport Inhibitor Cocktail (00-4980-03, Invitrogen) at 37 °C for 5 h. For pSMAD2/3 and pSTAT5 staining, freshly isolated T cells were stimulated ex vivo with the indicated proteins for 25 min, then were immediately fixed with Cytofix Fixation Buffer (554655, BD) and permeabilized using Phosflow Perm Buffer III (558050, BD). Cells were resuspended in PBS and analysed on CytoFLEX flow cytometer (Beckman Coulter). Data analysis was performed using FlowJo software v.10.10.0.
Protein production
Recombinant proteins were cloned into the pD649 mammalian expression vector (ATUM), which includes a haemagglutinin (HA) secretion signal peptide, an N-terminal MSA fusion, and a C-terminal 6×His tag. Expression constructs were transfected into Expi293F cells using the Expi293 Expression System (Gibco). After 3–4 days of culture, proteins were purified from supernatants by Ni-NTA Agarose (Qiagen), followed by size-exclusion chromatography using a Superdex 200 column in ÄKTA chromatography system (Cytiva). Endotoxin was removed using the Proteus NoEndo HC Spin Column Kit (VivaProducts), and levels were confirmed to be acceptable using the Pierce Chromogenic Endotoxin Quant Kit (Thermo Fisher Scientific). Final protein preparations were formulated and concentrated in sterile PBS, flash-frozen in liquid nitrogen, and stored at −80 °C until use.
CD4+ T cell isolation and in vitro differentiation
Mouse lymph nodes and spleens were collected and mechanically dissociated to obtain single-cell suspensions. Red blood cells were lysed using ACK lysis buffer (A10492-01, Gibco), followed by magnetic isolation of CD4+ T cells using the EasySep Mouse CD4+ T Cell Isolation Kit (19852, STEMCELL). Naive CD4+ T cells (CD4+CD44−CD25−Foxp3-GFP−) and activated CD4+ Tconv cells (CD4+CD44+CD25−Foxp3-GFP−) were subsequently sorted using a Sony SH800S Cell Sorter. The purity of the sorted populations was consistently greater than 99%. Human CD4+ T cells were isolated from frozen PBMCs using the EasySep Human CD4+ T Cell Isolation Kit (17952, STEMCELL). Complete medium was prepared using RPMI 1640 with GlutaMAX supplement (Gibco, 61870036) and supplemented with 10% fetal bovine serum (Gibco, A5256701), 10 mM HEPES (Gibco, 15630080), 1% sodium pyruvate (Gibco, 11360070), 1% penicillin–streptomycin (Gibco, 15140122) and 0.1% 2-mercaptoethanol (Gibco, 21985023).
For the in vitro mouse CD4+ T cell differentiation assay, naive CD4+ T cells were plated at 0.75 × 106 cells per ml in flat-bottom 96-well plates pre-coated overnight with 5 μg ml−1 InVivoMAb anti-mouse CD3 (Bio X Cell, BE0002) and 5 μg ml−1 InVivoMAb anti-mouse CD28 (Bio X Cell, BE0015-1). For OT-II cell differentiation, mouse splenocytes were plated at 1.5 × 106 cells per ml in flat-bottom 96-well plates and stimulated with 0.05 μg ml−1 OVA323–339 peptide (GenScript, RP10610). For human CD4+ T cell differentiation, CD4+ T cells were plated at 0.75 × 106 cells per ml in flat-bottom 96-well plates pre-coated overnight with InVivoMAb anti-human CD3 (Bio X Cell, BE0001-2) and 5 μg ml−1 InVivoMAb anti-human CD28 (Bio X Cell, BE0248). Recombinant proteins were added at the indicated concentrations at the start of the culture, and cells were incubated at 37 °C for ~4 days.
In vitro suppression assay
Thy1.1+ mouse naive CD4+ T cells were labelled with CellTrace Violet Cell Proliferation Dye (Invitrogen, C34571) and plated at 0.1 × 106 cells per well in flat-bottom 96-well plates pre-coated with 5 μg ml−1 anti-CD3 and 5 μg ml−1 anti-CD28 antibodies. Thy1.1− mouse Tconv cells or in vitro–differentiated pTreg cells generated with 1 nM proteins were added at the indicated ratios. Thy1.1+ cells were analysed after 48 h of co-culture.
Naive OT-II cell transfer and OVA administration
One million sorted Thy1.1+ naive OT-II cells were adoptively transferred into Thy1.1− C57BL/6 recipient mice via retro-orbital intravenous injection. Starting one day post-transfer, mice were administered 100 μg OVA protein (A5503, Sigma-Aldrich) along with 50 pmol of the indicated proteins via intraperitoneal injection every other day, for a total of six injections. On day 11, mLN, ILN, and spleens were collected for analysis. For FTY720 treatment, 20 µg FTY720·HCl (ENZO Life Sciences) was dissolved in 100 µl of 5% DMSO in PBS and administered via intraperitoneal injection daily from day 0 to day 10, for a total of 11 doses.
OVA-induced airway inflammation model
One million sorted Thy1.1+ naive OT-II cells were adoptively transferred into Thy1.1− C57BL/6 recipient mice, followed by five intraperitoneal injections of 100 μg OVA protein combined with 50 pmol of the indicated proteins. One week after the final injection, mice received three weekly intraperitoneal injections of 100 μg OVA protein formulated in 150 μl Alhydrogel adjuvant (2%; InvivoGen, vac-alu-50). One week after the last adjuvant injection, mice were administered three intranasal doses of 100 μg OVA protein every other day. Tissues were collected and analysed one day after the final intranasal dose.
OVA-induced food allergy model
One million sorted Thy1.1+ naive OT-II cells were adoptively transferred into Thy1.1− C57BL/6 recipient mice, followed by five intraperitoneal injections of 100 μg OVA protein combined with 50 pmol of the indicated proteins. One week after the final injection, mice received three weekly oral gavages of 5 mg OVA protein together with 10 µg cholera toxin (C8052, Sigma-Aldrich). Donor OT-II cells in the gut were analysed one day after the last gavage. One week later, mice were challenged with 200 µg OVA protein via intraperitoneal injection. Rectal temperature was recorded immediately thereafter every 5–10 min for 75 min using a Type J/K/T thermocouple thermometer (Kent Scientific), and serum was collected one day later for analysis.
Naive 2D2 cell transfer and MOG35–55 administration
Three million sorted Thy1.1+ naive CD44−CD25− 2D2 cells were adoptively transferred into female Thy1.1− C57BL/6 recipient mice via retro-orbital intravenous injection. Beginning one day after transfer, mice received 40 µg MOG35–55 peptide (Genemed Synthesis) together with 50 pmol of the indicated proteins by intraperitoneal injection every other day, for a total of six doses. On day 11, mLN, ILN and spleens were collected for analysis.
MOG35–55-induced EAE model
Female mice were administered 40 µg MOG35–55 peptide together with 50 pmol of the indicated proteins every other day for a total of five doses. One week later, mice were immunized subcutaneously with 100 µg MOG35–55 peptide per mouse in incomplete Freund’s adjuvant (BD Biosciences) containing 200 µg Mycobacterium tuberculosis per mouse (BD Biosciences), injected at the axilla of both sides. Concurrently, 400 ng pertussis toxin per mouse (PTX, List Labs) was administered intraperitoneally, with a second dose given 48 h later. Mouse body weight and clinical signs of disease were recorded daily and scored according to the following scale: 1, tail paralysed; 1.5, mild hind limb weakness; 2, moderate/typical hind limb weakness; 2.5, severe hind limb weakness without paralysis; 3, one or both hind limbs paralysed, front limbs fully functional; 3.5, both hind limbs paralysed, front limbs/paws weak but not paralysed; 4, front limb paralysis; 5, moribund or deceased. Tissues were collected and analysed on day 25.
DSS-induced colitis model
Two million sorted Thy1.1+ naive OT-II cells were adoptively transferred into Thy1.1− C57BL/6 recipient mice via retro-orbital intravenous injection. Beginning one day after transfer, mice received 100 µg OVA protein together with 50 pmol of the indicated proteins by intraperitoneal injection every other day, for a total of five doses. Two days later, the mice were given drinking water containing 2.5% DSS (colitis grade, 36,000–50,000; MP Biomedicals) supplemented with 2.5 mg ml−1 OVA protein. Simultaneously, mice were administered 5 mg OVA protein via oral gavage every other day for a total of six doses. After six days, the DSS- and OVA-supplemented water was replaced with regular drinking water. Mouse body weight was recorded daily throughout the experiment, and tissues were collected and analysed at the endpoint.
Isolation of immune cells from tissues
Mice were euthanized after completion of the respective treatments. BALF was collected by flushing the lungs three times with 0.75 ml of PBS via a catheter inserted into the trachea. For lymphocyte isolation from the lung, tissues were mechanically dissociated using the plunger of a 1 ml syringe and filtered through 70-μm cell strainers to obtain single-cell suspensions. For lymphocyte isolation from the lamina propria, Peyer’s patches in the small and large intestines were first removed. The intestines were then opened longitudinally, cut into ~2-cm pieces, and incubated in 5 mM EDTA (15575020, Invitrogen) with 1 mM DTT (R0861, Thermo Fisher Scientific) at 37 °C for 30 min to remove epithelial cells. Tissues were then minced and digested in DNase I (40 µg ml−1; Roche) and collagenase D (0.5 mg ml−1; Roche) at 37 °C for 30 min with shaking to generate single-cell suspensions, which were filtered through 70-μm cell strainers. For lymphocyte isolation from the spinal cord, mice were first perfused, and the collected spinal cords were mechanically dissociated using the plunger of a 1 ml syringe and passed through 70-μm cell strainers to obtain single-cell suspensions. The resulting cells were subjected to density gradient centrifugation using a 40%/70% Percoll (Cytiva) gradient. Immune cells located at the interface between the two Percoll layers were collected and processed for flow cytometry analysis.
ELISA
Blood samples were centrifuged at 3,000g, and serum was collected from the supernatant. Immunoglobulin levels were measured using ELISA kits: total IgE (ELISA MAX Standard Set Mouse IgE; BioLegend, 432401), OVA-specific IgE (LEGEND MAX Mouse OVA-Specific IgE ELISA Kit; BioLegend, 439807) and OVA-specific IgG1 (Mouse Anti-OVA IgG1 Antibody Assay Kit; Chondrex, 3013).
Histology
Lung tissues from perfused mice and colon tissues were fixed in 10% neutral buffered formalin (Sigma-Aldrich, HT501128) and submitted to S. Avolicino for paraffin embedding, sectioning, and haematoxylin and eosin staining. Slides were imaged on a Leica DM2000 microscope, and histopathology was evaluated in a blinded manner. Lung inflammation was scored on the basis of the extent of peribronchial and perivascular cellular infiltration: 0, no infiltrates; 1, a few inflammatory cells; 2, a one-cell-thick ring of inflammatory cells; 3, a 2–3-cell-thick ring; 4, a 4–5-cell-thick ring; 5, a ring >5 cells thick. Colon inflammation was scored as follows: 0, no evidence of inflammation; 1, low-level inflammation with scattered infiltrating mononuclear cells (1–2 foci); 2, moderate inflammation with multiple foci; 3, high-level inflammation with increased vascular density and marked wall thickening; 4, severe inflammation with transmural leukocyte infiltration and loss of goblet cells.
scRNA-seq
Donor OT-II cells and endogenous Foxp3-GFP+ CD4+ T cells were sorted from the mLNs of the respective treatment groups on day 11 and submitted to MedGenome for library preparation and RNA sequencing. The FASTQ files were processed using Cell Ranger v.9.0.0. The gene expression matrix was processed and analysed using Seurat (v.5.1.0)46. For quality control, we excluded cells that contained fewer than 500 read counts for genes or fewer than 200 genes detected (minimal cutoff), or more than 50,000 read counts for genes or more than 6,500 genes detected (maximum cutoff). We also excluded cells in which more than 20% of transcripts were derived from mitochondrial RNA. These QC filters left 190,728 cells. Graph-based unsupervised clustering was employed to identify clusters representing minor contaminant cells other than T cells, such as neurons (expressing Cntn1, Dscam and Pde7b) and B cells (expressing Igkc, Ms4a1 and Cd79a). These minor clusters were excluded from subsequent analyses, leaving 180,038 cells. UMAP embedding was computed with 10 principal components, with n.neighbors being 20 and min.dist being 0.1. Differential expression analysis between groups was performed using Wilcoxon’s rank sum test implemented in Seurat’s FindMarkers function. Gene set enrichment analysis47 was performed using the log2FC ranking of differentially expressed genes using fgsea. Human hallmark gene sets were retrieved from MSigDB. The signature scores were calculated using Seurat’s AddModuleScore function. To calculate cell cycle score, we used the CellCycleScoring function in Seurat. Built-in human gene sets for the S and G2M phases in Seurat were converted into mouse homologues and used to calculate S and G2M scores. Transcription factor activity inference was conducted using pySCENIC with default parameter settings48. To identify differentially expressed genes from public bulk RNA-seq datasets for computing gene set signature scores, bulk RNA-seq FASTQ files were aligned to the GENCODE VM25 (mm10) reference genome using Rsubread49, and gene expression was quantified with featureCounts50. Differential expression analysis was performed using DESeq2 (ref. 51). Pathway analysis was performed using Metascape52.
Statistical analysis
Statistical analyses were performed using GraphPad Prism 10. Differences were considered statistically significant at P < 0.05, with significance denoted as follows: *P < 0.05, **P < 0.01, ***P < 0.001 and ****P < 0.0001. Detailed statistical information, including sample sizes, numbers of independent experiments, and the statistical tests used, is provided in the corresponding figure legends.
Ethics statement
All experimental mouse procedures were approved by the Stanford University Institutional Animal Care and Use Committee (IACUC; protocol IDs 32279 and 34708) and conducted in accordance with institutional guidelines. Blood for PBMC isolation from healthy donors was provided by the Stanford Blood Center, which also obtained ethical approval for the donors.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.