DNA constructs
The α7–5HT3 and α7–GlyR sequences were the same as reported previously11. Site-directed mutagenesis was performed using QuickChange Lightning SDM (Agilent). For these constructs, the α7 nAChR LBD was based on the sequence of human CHRNA7. The 5HT3 IPD was based on the mouse sequence. The GlyR IPD was based on the human sequence. For testing in human embryonic kidney (HEK) cells, channels were expressed from pcDNA3.1, (Invitrogen/Thermo Fisher Scientific).
For Cre-dependent rAAV, constructs containing Coca-GlyR followed by IRES-mCherry were cloned into an AAV2-Synapsin(Syn)-FLEX vector57 in the reverse orientation with respect to the Syn promoter. Using these constructs, rAAV serotype 1 was produced by the Janelia Molecular Biology Core Facility.
Coca-5HT3 (α7(L141G,G175K,Y210F,Y217F)–5HT3) or coca-GlyR (α7(L141G,G175K,Y217F)–GlyR) followed by IRES-mCherry were cloned into an AAV2 vector downstream of the Syn promoter and were also packaged as AAV serotype 5 (Janelia).
For electroporation and in vitro electrophysiology in CA1 pyramidal cells, mutant channel cDNAs were cloned into a pCAG expression vector with an upstream CAG promoter and a downstream IRES-GFP sequence.
The numbering scheme used for ion channel amino acid residue positions is based on the unprocessed pre-protein sequence derived from the cDNA sequences.
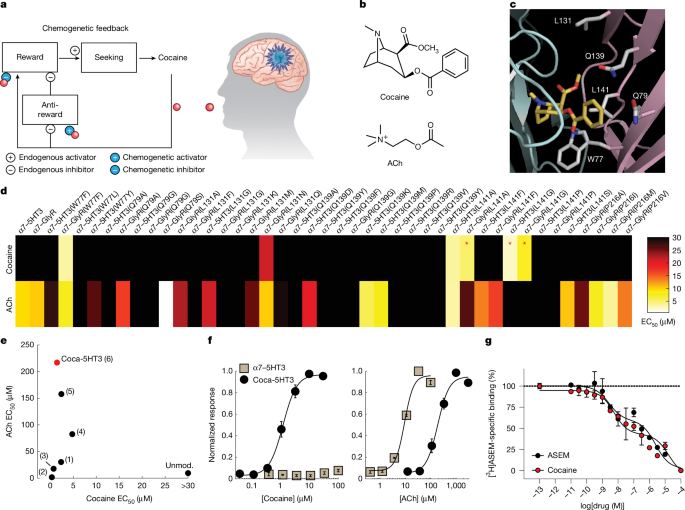
Structural model of cocaine–LBD interactions
The crystal structure of cocaine in a complex with AChBP was obtained from the Protein Data Bank (PDB: 2PGZ). Amino acid substitutions were made in PyMol for residues near cocaine in the crystal structure for the homologous residues in the human protein sequence for display purposes. The model was used to guide exploration of residues to be mutated for effects on cocaine agonism.
Ion-channel testing using a membrane potential assay
ATCC CRL-1573 cells (HEK293, passages 40–49) were plated into Corning CellBIND plates (10 cm diameter), at 3 × 106 cells per plate and grown for 18–24 h to achieve about 80% confluency. Although the HEK293 cell line is frequently misidentified, we used obtained it directly from the ATCC and used these cells because the chimeric ion channels constructs are efficiently transfected and express well in this cell line. Cell lines were tested for mycoplasma contamination. If contaminated cultures were detected, then they were not used for experiments.
To transfect cells, Opti-MEM (500 µl, Invitrogen), DNA plasmid (10 µg) and FuGene HD (30 µl, Roche) were combined, incubated for 15 min at room temperature and the mixture was then added dropwise to cells in a 10 cm2 plate. Cells were incubated at 37 °C in 8% CO2/air. After an additional 18–20 h, the medium was aspirated and cells were rinsed (1× PBS, 5 ml), treated with 0.05% trypsin-EDTA (3.0 ml, 5 min) and neutralized with DMEM + 10%FBS (7.0 ml). Cells were pooled, centrifuged (100g, 2 min) and resuspended in DMEM + 10% FBS then counted (Beckman Coulter Vi-Cell XR cell analyzer). Cells were then plated in 96 well BIOCOAT poly-d-lysine black/clear cellware with a cell density of 6 × 104 live cells in 200 µl per well. Cells were incubated another 22–26 h (37 °C in 8% CO2) then medium aspirated, and the FLIPR membrane potential assay solution was added and incubated at room temperature for approximately 20 min.
Membrane potential assay solution was prepared with a bottle of Molecular Devices membrane potential assay kit–Blue (R8034) dissolved in 200 ml of HEPES-buffered Earle’s balanced salt solution (HEBSS). Drug compound plates were prepared in Nalge Nunc 96-well conical-bottom plates in HEBSS ( < 0.5% DMSO) and tested at concentration intervals from 0.1 µM to 100 µM. (<0.1% DMSO)
Amphetamine and cocaine (Sigma-Aldrich) were dissolved in saline to a 100 mM stock before making the dilution series. Nicotine was dissolved to 100 mM stock in saline from >99% liquid (Sigma-Aldrich). All of the other controlled substances were isolated in 1 mg quantities using the SpeedVac (Thermo Fisher Scientific) from Cerilliant ACS Chemical Standard solutions in methanol (Sigma-Aldrich) and dissolved in DMSO stock to 100 mM. DMSO alone was also tested to ensure that there was no response at these dilutions. Endogenous amines for testing at coca-5HT3 (Extended Data Fig. 3) were dissolved in saline to 100 mM stocks.
To measure ion-channel activation, we used the Hamamatsu FDSS 6000 plate reader and liquid handling system. The assay plates were scanned at 1 Hz (excitation, 472/30 nm; emission, 540/40 nm). Compound solutions (50 µl) were simultaneously delivered from a 96-well compound plate after 10 baseline scans, which were followed by 170 additional scans. To calculate the MP EC50, the maximum response for each well was extracted, normalized to the maximum response for each compound and sigmoidal dose–response curves were calculated using software written in MATLAB (MathWorks Central: ec50.m, C. Evangelista, v.1.0, 14 January 2004) and the nlinfit function from the Statistics Toolbox.
HEK293 cell electrophysiology
Channel mutants and agonists were further characterized in whole-cell voltage-clamp experiments performed on HEK293 cells. At passage 44–48, cells were plated at low density (1 × 103 per cm2) on poly-d-lysine-coated glass coverslips, then transfected in 10 cm2 wells using the Fugene 6 transfection reagent (Roche) with DNA plasmids containing both an ion channel cDNA (pcDNA3.1, 2 µg per well) and cDNA for GFP (pCMV, 0.1 μg per well), followed by replacement of the medium after 3 h. Recordings were made 24–72 h after transfection.
Electrophysiological recordings were performed in whole-cell voltage-clamp mode, on an inverted fluorescence microscope (Olympus IX-51) using pClamp software and a Multiclamp 200B amplifier (Molecular Devices). Electrophysiological data was filtered at 4 kHz, digitized at 10 kHz using a Digidata 1400 interface (Molecular Devices), recorded on a PC (Dell) and analysed using custom MATLAB software (MathWorks). Solutions were delivered from a gravity fed perfusion system through a capillary tube placed approximately 100 mm from the recorded cell using an eight-way manifold pinch valve system (Nanion), which was controlled from the acquisition software. This allowed for up to seven concentrations of agonist to be tested. The duration of agonist application was based on pilot experiments to determine the time required to reach a steady-state current response for the channel. External solution (HEBSS) contained 140 mM NaCl, 10 mM HEPES, 5 mM glucose, 4 mM KCl, 2 mM CaCl2 and 1 mM MgCl2. The whole-cell pipette internal solution contained 87 mM CsGluconate, 31 mM CsCl, 5 mM NaCl, 10 mM HEPES, 10 mM EGTA and 3 mM MgATP. Osmolarities were within 285–305 mOsm l−1 and the pH was adjusted to 7.3–7.4 with NaOH.
Electrophysiology of cultured hippocampal neurons and LHb acute brain slices
Experiments and procedures complied with ethical regulations for animal testing and research and were approved by the Janelia Research Campus and UCSD animal care and use committees. Hippocampal neuron cultures were prepared by modification of a previously reported method58. Hippocampi were dissected from postnatal day 0 neonatal rat pups (Charles River) in HEPES buffered Hank’s solution containing 100 U ml−1 penicillin and 10 μg ml−1 streptomycin. Tissue was dissociated in papain (Worthington, PAP2) in dissection buffer for 30 min at 37 °C. After digestion, papain was removed and tissue was dissociated in plating medium (MEM plus 10% fetal bovine serum, 28 mM glucose, 2.4 mM NaHCO3, 100 µg ml−1 transferrin, 25 μg ml−1 insulin, 2 mM l-glutamine, 100 U ml−1 penicillin and 10 µg ml−1 streptomycin). The cell suspension was passed through a 70 µm filter and centrifuged at 90g for 7 min. The resulting cell pellet was resuspended in plating medium, counted and assayed for cell viability.
pCAG::coca-GlyR-IRES-GFP or pCAG::coca-5HT3-IRES-GFP plasmids were transfected by electroporation using the Lonza Nucleofector system (P3 Primary Cell 96-well Kit, Lonza) then cells were plated onto poly-d-lysine-coated 13 mm dia #1 glass coverslips in the wells of a 24-well plate at 50,000 cells per well. Each well received 60 µl of cell suspension plus 60 µl of NbActiv4 medium (BrainBits) and was incubated at 37 C in 5% CO2 for 4 h for initial cell attachment. NbActiv4 medium (1 ml) was then added to each well and cultures kept for the duration of the experiment. Weekly medium changes were made by replacing 0.5 ml medium from each well with fresh NbActiv4.
Whole-cell recording internal solution contained 135 mM potassium gluconate, 6.7 mM KCl, 10 mM HEPES, 1.0 mM EGTA, 4.0 mM Na2ATP and 0.3 mM Na3GTP. The osmolarity was 285–305 mOsm l−1 and the pH was within 7.3–7.4. Whole-cell recording pipettes had tip resistance between 1.5 and 4 MΩ and whole-cell series resistance was 6–29 MΩ (mean ± s.e.m., 17.2 ± 0.6 MΩ). The external solution contained: 140 mM NaCl, 10 mM HEPES, 5 mM glucose, 4 mM KCl, 2 mM CaCl2 and 1 mM MgCl2. Tight seals (>4 GΩ) were made in voltage-clamp mode and, immediately after patch disruption to establish the whole-cell recording configuration, the resting membrane potential was measured by switching briefly to the ‘I = 0’ mode on the Multiclamp Commander (Molecular Devices). After switching back to voltage-clamp mode, the other cell membrane properties were measured using the automated cell capacitance and membrane resistance compensation circuitry on the same device. Series resistance compensation was not applied. Membrane properties during current-clamp recordings were monitored by small current injections (−20 pA, 200 ms, 1 Hz). Thresholds for activation were estimated in current-clamp mode by injection of sequentially increasing depolarizing current steps (either 5 pA or 50 pA steps depending on whether a large shunt was observed due to chemogenetic receptor activation by cocaine). The threshold was measured as the amplitude of current injection that induced the first action potential.
For analysis of coca-5HT3 effects on the passive membrane properties of LHb neurons, C57BL/6J mice (male, aged 4–5 weeks) were injected with AAV5-Syn::coca-5HT3-IRES-mCherry (titre, 4 × 1012 viral genomes (vg) per ml) or with a control virus AAV5-Syn::IRES-mCherry (titre: 2 × 1012 vg per ml) in the LHb (anteroposterior (AP): −1.55 mm relative to bregma; mediolateral (ML): +0.45 mm; dorsoventral (DV): −2.65 mm, −2.75 mm, −2.85 mm, 100 nl at each depth). After 2–5 weeks, brain slices were prepared. In brief, animals were deeply anaesthetized with isoflurane, decapitated and the brains removed in ice-cold sectioning solution (described below). The brains were then mounted onto a stage using Krazy Glue, and coronal brain slices (250 µm) were cut using a cooled tissue slicer (Leica 1200S). Slices were made in an ice-cold, low-sodium, low-calcium solution containing: 135 mM N-methyl-d-glucamine, 2.2 mM KCl, 20 mM NaHCO3, 1.2 mM NaH2PO4, 10 mM glucose, 0.25 mM CaCl2, 1.75 mM MgCl2 and 113 mM HCl (HCl 25% solution, 6.85 M, 16.5 ml l−1) then kept in recording saline, with 1 mM ascorbic acid and 2 mM pyruvic acid added, until needed (30 min to 5 h). N-Methyl-d-glucamine was used as a sodium replacement. Slices were held down in a glass-bottomed chamber, perfused and viewed on the stage of an upright Olympus BX-51 fluorescence microscope using infrared gradient contrast optics. Transfected neurons were identified by their mCherry fluorescence. Electrophysiological recordings were made in whole-cell current-clamp mode, using a Multiclamp 200B amplifier (Molecular Devices) and MATLAB Ephus software for acquisition59. Data were filtered at 4 kHz, digitized at 10 kHz using the National Instruments interface hardware and recorded on a PC.
Radioligand-binding assays in HEK293 cells
HEK293 cells were transfected with 5 µg per dish of plasmids encoding coca-5HT3 or coca-GlyR (in pcDNA3.1) and collected 48 h after transfection. Cells were suspended in Tris-HCl 50 mM pH 7.4 supplemented with protease inhibitor cocktail (1:100, Sigma-Aldrich). HEK293 cells were disrupted with a 6 Polytron homogenizer (Kinematica). Homogenates were centrifuged at 48,000g (50 min, 4 °C) and washed twice in the same conditions to isolate the membrane fraction. Protein was quantified using the bicinchoninic acid method (Pierce). Membrane suspensions (50 µg of protein per ml) were incubated in 50 mM Tris-HCl (pH 7.4) containing 8 mM CaCl2, 2 nM of [3H]ASEM (26 Ci mmol−1, Novandi Chemistry) and increasing concentrations of the tested compounds during 2 h at room temperature as previously described12. Non-specific binding was determined in the presence of 100 µM of non-radiollabeled ASEM. Free and membrane-bound radioligands were separated by rapid filtration of 500 μl aliquots in a 96-well plate harvester (Brandel) and washed with 2 ml of ice-cold Tris-HCl buffer. Microscint-20 scintillation liquid (65 μl per well, PerkinElmer) was added to the filter plates, plates were incubated overnight at room temperature and radioactivity counts were determined in the MicroBeta2 plate counter (PerkinElmer) with an efficiency of 41%. Two-site competition curves were fitted using Prism 10 (GraphPad). Ki values were calculated using the Cheng–Prusoff equation.
Animals for behavioural experiments
Experiments and procedures complied with ethical regulations for animal testing and research, followed the NIH guidelines and were approved by the NIDA animal care and use committee. The experimenters were blinded to the group allocation. Rats were single housed on a reverse light cycle (lights off 07:00; lights on 19:00). Sample sizes were estimated using G*Power or based on experience from past work.
Open-field locomotion
Adult male Sprague–Dawley rats were bilaterally injected (0.5 µl per side) with AAV5–Syn::coca-5HT3-IRES-mCherry in the LHb (10° angle, AP relative to bregma: −3.8 mm; ML, ±1.7 mm; DV, −5.2 mm) (n = 6) or exposed to sham surgery (n = 6). Then, 4 weeks later, the rats were assessed for open-field locomotion. Each rat was placed in a clear Plexiglas box (40 cm × 40 cm × 30 cm) and activity was recorded by infrared beam brakes for 60 min (Opto-varimex ATM3, Columbus Instruments). The distance travelled was plotted as mean ± s.e.m. centimetres between the control and coca-5HT3 rats.
Sucrose preference
Rats from the open-field locomotor experiments were presented with two bottles of water in their home cage to acclimatize them to the procedure for 1 day. The next day, one bottle was replaced with a 1% sucrose solution dissolved in water. After 24 h, the consumed volume was noted in ml and the position of the bottles was switched to control for potential side preference. On day 3 the consumed volume was calculated for both water and sucrose 24 h after position switch. This volume was then plotted as percentage mean ± s.e.m. consumption between control and coca-5HT3 rats.
Sucrose pellet operant responding
Adult male Sprague–Dawley rats were bilaterally injected (0.5 µl per side) with AAV5–Syn::coca-5HT3-IRES-mCherry in the LHb (10° angle; AP relative to bregma, −3.8 mm; ML, ±1.7 mm; DV, −5.2 mm) (n = 8) or exposed to sham surgery (n = 5). Then, 4 weeks later, the rats were restricted to 18–20 g of chow per day and trained to press a lever for 20 mg sucrose pellets on an FR1 schedule (1 h per day for 7 days). Only the active lever was extended during the session. Presses on this lever activated the food magazine to dispense one pellet (Dustless Precision Pellets, F07595, 20 mg, Sucrose; Bio-Serv) accompanied by a 20 s cue light and timeout. Responses during the timeout period were recorded but did not result in pellets being dispensed.
Dopamine levels
Rats from the sucrose preference experiment were bilaterally injected (0.5 µl per side) with AAV5-Syn::coca-5HT3-IRES-mCherry in the LHb (10° angle; AP relative to bregma, −3.8 mm; ML, ±1.7 mm; DV, −5.2 mm) (n = 6) or exposed to sham surgery (n = 6). Then, 6 weeks later, the rats were euthanized, and brain tissue was extracted and flash-frozen in isopentane. The tissue was then chunked, removing the olfactory bulbs and coronally sectioning at the optic chiasm. The tissue that contained the bilateral striatum was then homogenized and analysed for dopamine levels using an enzyme-linked immunosorbent assay (ELISA; Abcam, ab285238). Dopamine levels were plotted as the mean ± s.e.m. pg per mg between control and coca-5HT3 rats.
Cocaine IVSA
Male Long Evans rats were anaesthetized with isoflurane (1.5–2%). The rats were then placed onto a stereotaxic frame (Kopf) and injected with the AAV vector AAV5-Syn::coca-5HT3-IRES-mCherry (0.5 µl, 4.95 × 1012 vg per ml) into the LHb (AP relative to bregma, −3.8 mm; ML, ±0.7 mm; DV, −5.25 mm, at a 10° angle) (n = 11) or underwent sham surgery (n = 15).
Approximately 1 week later, the rats were trained to self-administer food in daily operant sessions. For food training, rats were restricted to four large chow pellets per day and the body weight was monitored daily. Rats were tested for 10 days, and the sessions lasted for 3 h. For the first 5 days, the rats were tested using a fixed ratio 1 (FR1) schedule of reinforcement and were then switched to an FR5 schedule for the 5 subsequent days. Each lever press led to delivery of a 45 mg food pellet (maximum of 200 pellets per session) and activation of a cue light above the lever for 1 s. Each reinforced lever press was followed by a 20 s timeout during which lever responses were recorded but had no programmed consequences.
After these operant sessions, the rats underwent surgery for jugular vein catheterization. The rats were anaesthetized with isoflurane (1.5–2%). Catheters made from Silastic tubing attached to a modified 22-gauge cannula (Plastics One, C313G-5up) and cemented to polypropylene mesh (Elko Filtering, 05–1000/45) were inserted into the jugular vein, and the mesh was fixed to the mid-scapular region of the rat. Rats were injected with carprofen (2.5 mg per kg, subcutaneous, Norbrook) after surgery and on the next day. Rats recovered for 6 days before self-administration procedures. Catheters were flushed daily with gentamicin (4.25 mg ml−1, Fresenius Kabi, 1002) dissolved in sterile saline. If catheter failure was suspected, catheter patency was tested with a short-acting barbiturate anaesthetic Brevital (methohexital sodium, 10 mg ml−1 in buffered saline, 0.1–0.2 ml injection volume, i.v.).
After approximately 1 week, the rats began daily cocaine IVSA operant training sessions. Rats were tested using an FR1 schedule of reinforcement and were then switched to an FR5 schedule. In these sessions, a response on the lever previously paired with food had no programmed consequences (that is, inactive lever). A response on the cocaine-paired lever (that is, active lever) led to the infusion of a 0.5 mg per kg dose of cocaine (maximum of 60 infusions per session) and coincided with activation of a cue light located above the active lever for 20 s (that is, timeout period). During this time, lever responses were recorded but had no programmed consequences. After cocaine training, the rats were tested for IVSA using different cocaine unit doses (0.0625, 0.125, 0.25, 0.5 and 1 mg per kg per infusion). The procedures were identical to the initial sessions. The order of unit dose testing was randomized across rats. Rats could commit a maximum of 60 infusions when tested at the 0.5 and 1 mg per kg unit doses but no infusion limit was present at other unit doses. Statistical analyses were performed using Prism 10 (GraphPad).
PET analysis with [18F]fallypride
Male Sprague–Dawley rats were anaesthetized with isoflurane (1.5-2%) and then placed onto a stereotaxic frame (Kopf) and injected with AAV5-Syn::coca-5HT3-IRES-mCherry (0.5 µl, 4.95 × 1012 vg per ml) into the LHb (AP relative to bregma, −3.8 mm; ML, ±0.7 mm; DV, −5.25 mm, at a 10° angle) (n = 5) or underwent sham surgery (n = 9). Male Th–cre rats were anaesthetized with isoflurane (1.5–2%) and then placed onto a stereotaxic frame (Kopf) and injected with AAV1-Syn::FLEX-coca-GlyR-IRES-mCherry (0.5 µl, 6 × 1012 vg per ml) into the VTA (AP, −6.0 mm relative to bregma; ML, ±1.9 mm; DV, −8.3 mm at a 10° angle) (n = 9) or underwent sham surgery (n = 6). Approximately 3 weeks later, the rats were anaesthetized, placed inside the PET scanner (Mediso NanoScan PET/CT), injected with [18F]fallypride (~13 MBq) and then scanned using PET. [18F]fallypride reaches steady-state by around 45 min after its injection and then remains stable for at least 150 min60. We therefore defined the baseline [18F]fallypride binding from 60 to 90 min after its injection and then, to elicit an increase in extracellular dopamine, at 90 min after [18F]fallypride injection, we injected rats with cocaine (3 mg per kg, i.v.) and continued to scan for another 30 min (90–120 min). The PET data were reconstructed and corrected for dead-time and radioactive decay and then co-registered to an magnetic resonance imaging template as previously described46,60,61. Co-registered images were analysed using one-way ANOVA and the resulting parametric images were filtered for statistically significant (P < 0.01) clusters larger than 100 contiguous voxels. All statistical parametric mapping (SPM) analyses were performed using MATLAB R2016 (MathWorks) and SPM12 (University College London)62. Next, additional quantitative assessments of PET images were performed using the PMOD software environment (PMOD Technologies). We generated time–activity curves using the co-registered dynamic PET images and the significant voxel clusters defined by the voxel-wise SPM analysis as the volume of interest. The standardized uptake value (SUV) was calculated using the equation SUV = C/(dose/BW) where C is the tissue concentration of [18F]fallypride (kBq cm−3), dose is the administered dose (MBq) and BW (kg) is the animal’s body weight. Statistical analyses were performed using Prism 10 (GraphPad).
Fibre photometry of dopamine release after cocaine administration
Male Sprague–Dawley rats were anaesthetized with isoflurane (1.5–2%) and then placed onto a stereotaxic frame (Kopf) and injected with AAV5-Syn::coca-5HT3-IRES-mCherry (0.5 µl, 4.95 × 1012 vg per ml) into the LHb (AP, −3.8 mm relative to bregma; ML, ±0.7 mm; DV, −5.25 mm, at a 10° angle) (n = 5) or underwent sham surgery (n = 6). Rats were also implanted with an optic fibre (400 µm core, 0.50 NA) attached to a 2.5 mm diameter ferrule (Thorlabs) directed to the medial shell of the nucleus accumbens (AP, +1.7 mm relative to bregma; ML, ±0.9 mm; DV, −7.0 mm). To ensure that AAV9-Syn::GRABDA1m was transduced at the direct vicinity of the fibre tip, we coated the fibre with the AAV (0.4 µl, 5 × 1012 vg per ml) using a silk fibroin film before its implantation as previously described63.
For fibre photometry, dopamine (GRABDA) dynamics were measured using a Tucker-Davis Technologies (TDT) system in anaesthetized rats. The dopamine and isosbestic signals were excited by 465 (~40–50 µW) and 405 nm (~20 µW) LEDs, respectively. Data were acquired at 1 kHz. On the testing day, rats were anaesthetized (isoflurane: induction, 5%; maintenance, 3%) and attached to a photometry patch cord (400 μm core, 0.50 NA). GRABDA signals were then collected for 5 min, followed by a saline i.p. injection, and additional GRABDA signal measurement for 20 min. Cocaine was then injected i.p. at 20, 10 or 5 mg per kg, and the GRABDA signal was measured for an additional 20 min. Each rat was tested three times with cocaine doses administered in randomized order. Fibre photometry analyses were performed using scripts in MATLAB R2023b (MathWorks) based on codes from the Barker (https://github.com/djamesbarker)64 and Lerner (https://github.com/talialerner/)65 laboratories. The dopamine signal was fitted with the isosbestic signal [ΔF/F] and transformed to a z score for comparison of the dopamine signal between rats64. The GRABDA signal was normalized to values at 1 min before saline or cocaine injection and shown as the average z-scored activity per min. The AUC of the percentage ΔF/F of GRABDA fluorescence was calculated in a 20 min bin after the saline or cocaine injection. Fibre photometry data are represented as mean ± s.e.m. Statistical analyses were performed using Prism 10 (GraphPad).
Immunohistochemistry
Rats were anaesthetized with isoflurane and transcardially perfused with PBS, followed by 4% PFA. Brains were stored overnight in 4% PFA at 4 °C and then transferred to PBS with 30% sucrose for 3 to 4 days at 4 °C. Brains were frozen and sectioned on a cryostat (30 μm to 50 μm; Leica) and slices were collected in PBS. On the first day, the sections were blocked with 0.1% saponin and 1% BSA in physiological saline for 30 min at room temperature and incubated with specific antibodies: rat monoclonal anti-mCherry (1:1,000 diluted in 0.01% saponin and 0.1% BSA in physiological saline, Invitrogen, M11217) or chicken polyclonal anti-GFP antibody (1:2,000 diluted in 0.01% saponin and 1% BSA in physiological saline, Abcam, ab13970) at 4 °C overnight. The slices were soaked in physiological saline (three times for 5 min at room temperature), then incubated with a secondary antibody in 0.01% saponin and 0.1% BSA dissolved in physiological saline: goat-anti rat immunoglobulin G (IgG) (H+L) cross-absorbed, Alexa Fluor 594 (1:500, Invitrogen, A11007), or goat-anti chicken immunoglobulin Y (IgY) H&L (Alexa Fluor 488) (1:1,000, Abcam, ab150173) for 2 h at room temperature (protected from light). After secondary antibody incubation, the slices were washed with physiological saline (three 5 min washes) and then mounted onto SuperFrost Plus Slides, counterstained with Fluoromount-G medium (Southern Biotech) and coverslipped. Images were taken using a Leica LMD6 microscope equipped with the Leica DFC7000T camera and processed using LasX software.
Statistics
Statistical analysis was performed using SigmaStat or GraphPad Prism. We used mixed, two-way or three-way ANOVA taking repeated measures into account when appropriate or independent t-tests or paired-sample t-tests, as defined in the text. Statistical tests were two-sided. Post hoc tests were performed using Holm–Šidák multiple-comparison correction. Data are shown as mean ± s.e.m.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.