Cell lines and culture conditions
Huh7, U-2 OS, HEK293T and LX-2 cells were cultured in DMEM containing 4.5 g l−1 glucose and l-glutamine (Corning, 10-017-CM), supplemented with either 10% FBS (Thermo Fisher Scientific and Gemini Bio Products) for Huh7, U-2 OS and HEK293T or 2% FBS for LX-2, penicillin and streptomycin. HepG2 and 786-O cells were cultured in RPMI 1640 medium (Gibco, 11875093) containing l-glutamine, supplemented with 10% FBS, penicillin and streptomycin. All cells were maintained at 37 °C and 5% CO2. All cell lines were tested every 6 months for mycoplasma contamination.
Generation of endogenously labelled PLIN2–GFP Huh7, HepG2, U-2 OS and 786-O reporter cells was as described8.
Plasmids and cloning
All knockout cell lines (in Huh7, HepG2, U-2 OS, LX-2 and 786-O cells) were generated using the pMCB320 plasmid, a gift from M. Bassik (Addgene 89359). Guide sequences for CLCC1, TMEM41B, VMP1, CES1 (TGH) and safe targets (sgSAFE 5784) (Supplementary Table 2) were selected from the Bassik Human CRISPR Knockout Library (Addgene plasmids 101926, 101927, 101928, 101929, 101930, 101931, 101932, 101933 and 101934). Guide sequences were cloned into pMCB320 using the restriction enzymes BstXI and BlpI.
For exogenous protein expression, CLCC1 (DNASU, HsCD00951632), 1× Flag (DYKDDDDK)-tagged CLCC1, TMEM41B (DNASU, HsCD00829148), and VMP1 (DNASU, HsCD00080545) were cloned using Gibson assembly (New England Biolabs, E2611S) and the Gateway system (Thermo Fisher, 11791020) into a pLenti-CMV-Hygro vector (Addgene 17454). A pLenti-CMV-Hygro vector containing GFP with a tandem nuclear localization signal (NLS) (PKKKRKV) and nuclear export signal (NES) (LALKLAGLDI) sequence was ordered from GenScript. For the CLCC1-Brl1 chimera, the MFH domain (G362–Y434) of Brl1 (a gift from E. Ünal) was swapped with the MFH domain (D276–H362) of CLCC1 (DNASU, HsCD00951632) using Gibson assembly and was cloned into the pLenti-CMV-Hygro vector (Addgene plasmid 17454). Wild-type and mutant CLCC1 constructs used for structure–function analyses were synthesized by Twist Bioscience and cloned into the pLenti-CMV-Hygro vector (Addgene 17454). The mutant sequences were as follows: C254S (C254S), C279S (C279S), AH1 + p22 (TKALAVTFTTF-TKALAVTFTTF inserted after V307), AH1 + p25 (KALAVTFTTFVTEPLKHIGKGTGEF inserted after L326), ∆AH2 + linker (GGSGGSGGSGS between K257 and K275), AH2 3× Asp (W260D/F268D/W272D) and AH2 3× Gln (W260Q/F268Q/W272Q). Each mutant construct included two silent mutations, one within the PAM sequence and another within the CLCC1 sgRNA target site, in addition to the mutation of interest. These silent mutations (numbered according to the CLCC1 wild-type sequence) were: PAM mutation (1011A > C) and sgRNA target site mutation (1017G > T). This design allowed stable expression of CLCC1 variants in Cas9-expressing CLCC1-KO Huh7 cells, preventing re-editing by the expressed sgRNA. Lentiviral particles were produced as described above and used to transduce CLCC1-KO Huh7 cells. Transduced cells were selected in hygromycin-containing medium (200 μg ml−1) until all uninfected control cells were eliminated.
Generation of CRISPR–Cas9 genome-edited cell lines
To generate lentiviral particles, lentiCas9-Blast plasmid (Addgene 52962) was co-transfected with third-generation lentiviral packaging plasmids (pMDLg/pRRE, pRSV-Rev, and pMD2.G) into HEK293T cells. Lentiviral medium was collected 72 h after transfection, passed through a 40-µm filter, and then used to infect Huh7 (wild type, PLIN2–GFP), HepG2 (wild type, PLIN2–GFP) and U-2 OS (wild type, PLIN2–GFP) cells. Cells were selected in medium containing blasticidin (4 μg ml−1 in Huh7 and U-2 OS; 6 μg ml−1 in HepG2) for 5 days. Active Cas9 expression was validated by flow cytometry analysis following infection with a self-cleaving mCherry plasmid (pMCB320 containing mCherry and an sgRNA targeting the mCherry gene).
Lentiviral particles with sgRNA-containing pMCB320 plasmids were generated as described above and used to infect cells stably expressing Cas9. After 72 h of growth, infected cells were selected in medium containing puromycin (2 μg ml−1 in Huh7 and HepG2 cells; 1 μg ml−1 in U-2 OS cells) until over 90% cells were mCherry positive and all uninfected control cells were dead. Huh7 CLCC1-KO and TMEM41B-KO clones (in wild type and PLIN2–GFP backgrounds) were isolated using serial dilutions. Knockout efficiencies were confirmed via immunoblotting.
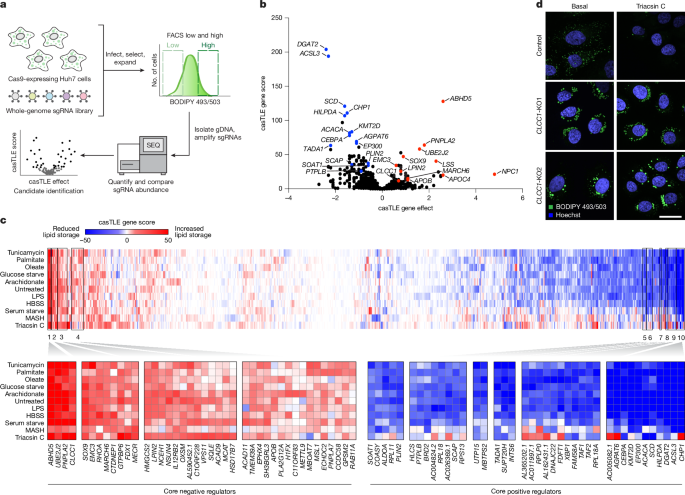
Genome-wide Huh7 CRISPR–Cas9 screens
All CRISPR–Cas9 screens reported here were performed as described previously8,39. Genome-wide CRISPR–Cas9 screens were performed using the Bassik Human CRISPR Knockout Library. The library consists of 9 sublibraries, comprising a total of 225,171 elements, including 212,821 sgRNAs targeting 20,549 genes (∼10 sgRNAs per gene) and 12,350 negative-control sgRNAs. Lentiviral particles containing each sublibrary were generated as described above. Huh7 cells stably expressing Cas9 were transduced with lentiviral packaged sublibraries (one sublibrary at a time) in 8 μg ml−1 polybrene. After 72 h of growth, infected cells were selected in medium containing 2 μg ml−1 puromycin until over 90% of cells were mCherry positive (via flow cytometry). Cells were then recovered for 3–5 days in medium lacking puromycin and frozen in liquid nitrogen.
For the screen, library infected cells were thawed (one sublibrary at a time) and maintained at 1,000× coverage (1,000 cells per library element) in 500 cm2 plates (about 2 × 107 cells per plate). Library-infected cells were passaged once before sorting. On the day of the sort, cells were dissociated using 0.25% Trypsin-EDTA (Gibco), collected by centrifugation at 300g for 3 min, stained with 1 µg µl−1 BODIPY 493/503 (Thermo Fisher Scientific, D3922) in DPBS on ice for 30 min, then washed once with DPBS. Cells were resuspended in phenol red-free medium (HyClone, 16777-406) supplemented with 3% FBS and 1% fatty acid-free BSA and kept on ice until FACS.
Cells were sorted on a BD FACS Aria Fusion equipped with 4 Lasers (488 nm, 405 nm, 561 nm and 640 nm). sgRNA-expressing, mCherry+ cells were gated into the brightest 30% and dimmest 30% by the 488 nm laser. Cells were sorted into 15 ml Falcon tubes containing DMEM with 4.5 g l−1 glucose and l-glutamine supplemented with 10% FBS. For each sort, 1,000 cells were collected (500 in each gate). Sorted cells were collected and sequenced as previously described39. Results from the genome-wide screen are available in Supplementary Table 1.
LD and metabolism library CRISPR–Cas9 screens
The custom human VLDM library contains 10,550 elements, with 8,550 sgRNAs targeting 857 genes (∼10 sgRNAs per gene) and 2,000 negative-control sgRNAs. Guide sequences were from the Bassik Human CRISPR Knockout Library, and the library construction protocol and cell line generation were previously described.
For each screen, cells were thawed and expanded at >1,000× coverage. For all screens, cells were seeded into 500 cm2 plates at 1,000-fold library coverage. For the Huh7 metabolic state-dependent screens, cells were treated the following day with: (1) no treatment; (2) 1 μg ml−1 triacsin C for 24 h; (3) 100 μM oleate–BSA complex for 24 h; (4) HBSS (Gibco, 14025092) for 24 h; (5) 0.2% FBS-containing DMEM (serum starve) for 48 h; (6) glucose-free DMEM (Gibco, 11966025) for 24 h; (7) 50 μM palmitic acid for 24 h; (8) 5 μM arachidonic acid for 24 h; (9) 5 μg ml−1 tunicamycin for 24 h; (10) 500 ng ml−1 LPS for 24 h; or (11) MASH stress mix (10 mM glucose, 5 mM fructose, 400 µM oleic acid, 200 µM palmitic acid, 100 ng ml−1 LPS and 30 ng ml−1 TNF) for 16 h. Cells were screened by FACS as described above. Results from each metabolic state screen are available in Supplementary Table 1.
CRISPR screen data analysis
Sequence reads were aligned to the sgRNA reference library using Bowtie 2 v.2.3.4.3 software. For each gene, a gene effect and score (likely maximum effect size and score) and P values were calculated using the Cas9 high-throughput maximum likelihood estimator (castle v.1.0) statistical framework as previously described.
Morpheus (https://software.broadinstitute.org/morpheus/) was used to perform unbiased gene clustering on metabolic state screens. Genes were ranked according to casTLE score and complete Euclidean linkages. Functional interactions and protein-protein interactions for high confidence candidate regulators were identified using the STRING database using STRING v.12.040.
General animal care
All procedures involving mice were approved by the UC Berkeley IACUC (protocol AUP-2022-02-15079-1, approved May 2022) and conducted in accordance with the NIH Guide for the Care and Use of Laboratory Animals, PHS Policy, and the Animal Welfare Act. Mice were maintained up to 12 weeks of age on a 12 h light:12 h dark cycle at ambient temperature (23 °C) and 30–70% relative humidity in the UC Berkeley pathogen-free barrier facility with free access to water and standard laboratory chow diet (LabDiet, 5053). We used Clcc1flox/flox in the C57BL/6 J genetic background (stock no. 000664). Experimentation was performed between 8 and 12 weeks of age. In mouse experiments, all measurements were included in the analysis. Mice were randomly allocated to groups; the only criteria were sex and age as explained above. The sample size and number of replicates for this study were chosen based on previous experiments performed in our laboratory and others. No specific power analysis was used to estimate sample size. Imaging studies could not be done blinded owing to the evident intrinsic features of the datasets. In vivo studies could not be blinded owing to the viral injection protocol. Experimental and control samples were processed together using the same conditions.
Floxed Clcc1 mouse generation
Clcc1 floxed mice (generated by the Knockout Mouse Project) (C57BL/6N-Atm1Brd Clcc1tm1a(KOMP)Mbp/JMmucd), where exon 7 is floxed, were obtained from the University of California Davis Mouse Biology Program. The neomycin selection cassette and lacZ reporter were removed by breeding to CAG-Flpo (C57BL/6N-Tg(CAG-Flpo)1Afst/Mmcd). Mice were then bred for at least four generations to C57BL/6 J mice, removing the CAG-Flpo. Genotyping for the floxed allele from genomic DNA was performed with the following PCR primers: TCATGACATGAACCATATGTGAATTCC and CACCATGCCTGGCTACAAATGC.
Adeno-associated virus mediated deletion of Clcc1
To deplete CLCC1, 8-week-old homozygous Clcc1-floxed mice were injected with either AAV8-TBG-Cre (Addgene 107787, a gift from J. M. Wilson) or AAV8-TBG-GFP (Addgene 105535, a gift from J. M. Wilson), via tail vein at a titre of 1.0 × 1011 genome copies per mouse. Mice were euthanized by CO2 four weeks after injection and livers were photographed. More than four mice per group were analysed per experiment. The liver was weighed and divided into pieces, which were flash frozen in liquid nitrogen, transferred on dry ice, and stored at −80 °C.
Mouse plasma collection and analysis
Blood was collected via submandibular vein puncture and centrifuged at 2,000g in microtainer SST tubes (BD, 365967) for 15 min at 4 °C to isolate plasma. Plasma was flash frozen in liquid nitrogen and stored at −80 °C. AST, ALT, and lipoprotein levels were analysed via Clinical Analyzer (Merck & Co). TAGs were quantified by a luciferase-based assay (Promega, J3160).
FPLC separation of mouse plasma
Size-exclusion chromatography was performed using an AKTA FPLC (Amersham Pharmacia Biotech). Equivalent volumes of plasma from each group of mice were pooled, totalling 400 μl (females) or 300 μl of plasma (males). Plasma was diluted in PBS so total sample volume equalled 1,000 μl and was applied to a Superose 6 followed in tandem with a Superdex 200 column and separated into lipoprotein classes in 10 mM PBS, pH 7.4, containing 0.02% sodium azide and collected into 48× 0.5 ml fractions. Fractions were then analysed for cholesterol (C7510) and TAGs (T7532) with indicated colorimetric kits (Pointe Scientific).
Flow cytometry
Cells were washed twice in DPBS, dissociated using TrypLE Express (Gibco, 12605010), collected by centrifugation at 500g for 5 min, and stained with 1 µg µl−1 BODIPY 493/503 or 200 µM monodansylpentane (MDH, Abcepta, SM1000b) in DPBS on ice for 30 min.
For all flow cytometry assays, fluorescence was analysed using an LSR Fortessa (BD Biosciences) using BD FACS v.6.2 (BD Biosciences). The following filter sets were used: FITC (GFP, BODIPY 493/503), Pacific Blue (BFP, MDH) and Texas-Red (mCherry). FlowJo v.10 software (BD Biosciences) was used to quantify fluorescence and generate representative histograms. Example gating strategy is shown in Supplementary Fig. 14.
Immunoblotting
For intracellular proteins from tissue culture, cells were lysed in 1% SDS and sonicated at 15% amplitude for 15 s. For secreted proteins from tissue culture, cells were incubated for 24 h in FBS-free DMEM, proteins were precipitated from the medium with acetone, and the pellet was resuspended in 100 μl 1% SDS. Mouse plasma samples were diluted 1:25 in 1% SDS. Animal tissues were homogenized in NP-40 lysis buffer (1% NP-40, 50 mM Tris-HCl, 5 mM EDTA, 2 mM EGTA, 30 mM NaF, 10 mM sodium pyrophosphate and 40 mM B glycerolphosphate) with an immersion homogenizer for 15 s. Protein concentrations were determined and normalized using a BCA protein assay (Thermo Fisher Scientific, 23225). Equal amounts of protein were combined with Laemmli buffer, boiled for 10 min at 95 °C, separated on 4–20% polyacrylamide gradient gels (Bio-Rad Laboratories) and transferred onto 0.45-mm nitrocellulose membranes at 25 V for 3 min (Bio-Rad Laboratories). Membranes were incubated in 5% nonfat milk in PBS with 0.1% Tween-20 (PBST) for 30 min to reduce nonspecific antibody binding. Membranes were then incubated overnight at 4 °C in 5% BSA in PBST containing the following primary antibodies at 1:1,000 dilution: rabbit anti-CLCC1 (Thermo Fisher Scientific, HPA009087), rabbit anti-PLIN2 (Abcepta, AP5118c), rabbit anti-albumin (Proteintech, 16475-1-AP), mouse anti-MTP (Santa Cruz, sc-515742), goat anti-CES1/TGH (R&D Systems, AF4920SP), rabbit anti-BiP (Cell Signaling, C50B12), rabbit anti-TMEM41B (Proteintech, 29270-1-AP), rabbit anti-VMP1 (Cell Signaling, D1Y3E), mouse anti-lamin A/C (Cell Signaling, 4777), rabbit anti-calnexin (Cell Signaling, C5C9), mouse anti-actin (Cell Signaling, 4970), and rabbit anti-GAPDH (Cell Signaling, 2118). Membranes were incubated for at least 1 h in IRDYE secondary antibodies (LI-COR, 926-68074, 926-68070, 926-32211) at a 1:10,000 dilution in PBST containing 5% nonfat milk. Immunoblots were visualized on a LI-COR imager (LI-COR Biosciences) running Odyssey v.3.0, and Fiji/ImageJ v.1.53e (NIH) was used for quantification of protein levels.
For immunoblotting apoB, FPLC fractions were diluted with 2× Laemmli buffer and run as described above. Gels were then transferred to 0.45-μm nitrocellulose membranes at 350 mA for 1.5 h. Membranes were blocked in 5% nonfat milk in PBST overnight at 4 °C, incubated with 1:1,000 anti-apoB antibody (Abcam, ab20737) for 2 h at room temperature and subsequently incubated with 1:10,000 goat anti-rabbit secondary antibody (LI-COR, 926-68074) for 1 h at room temperature.
Nuclei isolation
Nuclei were isolated using the Nuclei EZ Prep Kit (Sigma, NUC101). Samples were diluted 1:1 with 1% SDS and were immunoblotted for purity with lamin A/C, calnexin and CLCC1 as described above.
Fluorescence microscopy
For widefield microscopy of live cells, Huh7, HepG2, U-2 OS and 786-O cells were grown in 4-well or 8-well Nunc LabTek II Chambered Coverglass (Borosilicate Glass 1.5; Thermo Fisher Scientific, 155360) coated with poly-l-lysine. LDs were stained with 1 μM BODIPY 493/503 for 30 min or 500 nM Lipi-Blue for 30 min, nuclei were stained with 5 μg ml−1 Hoechst 33342 (Thermo Fisher Scientific, 62249) for 30 min, lysosomes were stained with 75 nM Lysotracker DND-22 (Thermo Fisher Scientific, L7525) for 30 min, and mitochondria were stained with 500 nM Mitotracker Green (Thermo Fisher Scientific, M7514) for 30 min. For imaging the ER, cells were transiently transfected with BFP-KDEL (Addgene 49150) and imaged 48 h later. For imaging of nuclear blebs, cells were transiently transfected with MLF2-GFP-Flag-pCDNA3.1 (a gift from C. Shlieker) and imaged 48 h later. Prior to imaging, cells were washed twice with DPBS and imaged in fresh phenol red-free medium supplemented with 10% FBS. Live cells were imaged using a Zeiss Axio Observer 7 fitted with a 63× oil objective using DAPI, GFP, Cy-3 and Cy-5 filters. Cells were imaged at 37 °C with 5% CO2. z-stacks of 0.2-μm thickness were acquired using ZEISS ZEN v.3.2 (ZEISS Microscopy) software.
To evaluate the endogenous localization of CLCC1 and ER, Sec61β–GFP was overexpressed in control (expressing sgSAFE) and CLCC1-KO Huh7 cells. Twenty-four hours after transfection, cells were fixed with 4% paraformaldehyde for 10 min at room temperature and washed with 1× PBS three times, followed by a 20 min permeabilization using 0.2% Triton X-100 plus 3% BSA at room temperature. Cells were washed three times with 1× PBS before staining. Endogenous CLCC1 and Lamin A/C proteins were stained with rabbit anti-CLCC1 (Thermo Fisher Scientific, HPA009087) and mouse anti-Lamin A/C (Cell Signaling, 4777) antibodies overnight at +4 °C, at concentrations of 1:50 and 1:200, respectively, in 1× PBS plus 3% BSA. Cells were then washed three times with 1× PBS and stained using fluorescent secondary antibodies diluted at 1:1,000 in 1× PBS plus 3% BSA for 2 h in the dark at room temperature. Nuclear staining was performed using HCS NuclearMask Stains (Invitrogen, H10325,), 1:2,000 in 1× PBS at room temperature for 10 min. For the analysis of the effects of CLCC1 mutants on LD content, PLIN2 localization, and MLF2–GFP nuclear blebs, Huh7 cells stably expressing each CLCC1 construct were transfected with MLF2–GFP in the Flag-pCDNA3.1 vector. After 48 h, cells were fixed, processed as described above, and stained for LDs using Lipi-Blue and for PLIN2 (Proteintech, 15294-1-AP).
For widefield and Lattice Structural Illumination Microscopy (SIM) of fixed cells, Huh7 cells were grown either in 12-well plates on glass coverslips coated with poly-l-lysine or in 35 mm dishes (Invitrogen, C10046). Cells were washed three times with DPBS, fixed for 15 min in 4% (w/v) PFA in DPBS and washed three times again with DPBS. Cells were permeabilized for 15 min with 1% BSA in DPBS containing 0.1% Triton X-100 when staining for ER, Golgi, or nuclear proteins or 0.01% digitonin when staining for LD proteins and then washed three times with DPBS. Cells were then incubated for 2 h in the dark at room temperature with the following antibodies diluted 1:1,000 in 1% BSA in DPBS: rabbit anti-PLIN2 (Abcepta, AP5118c), GM130 (Cell Signaling, 12480), rabbit anti-CLCC1 (Thermo Fisher Scientific, HPA009087), KDEL (Abcam, ab176333), mouse anti-lamin A/C (Cell Signaling, 4777), goat anti-ApoB (Rockland, AB742) or Mab414 (Abcam, ab24609). Cells were washed three times with DPBS before staining for LDs with 1 μM BODIPY 493/503 for 30 min or 500 nM Lipi-Blue for 30 min, nuclei with 1 µg ml−1 DAPI, and fluorescent secondary antibodies (Thermo Fisher Scientific, A21202, A-21109) diluted at 1:1,000 in 1% BSA in DPBS for 30 min in the dark. Cells were washed three times with DPBS and coverslips were mounted on 1 mm glass slides using Fluoromount-G (SouthernBiotech, 0100-01). Widefield fluorescence images were acquired using a Zeiss Axio Observer 7 as above and Lattice-SIM images were acquired on a Zeiss Elyra7 superresolution fluorescence microscope, equipped with dual sCMOS PCO Edge 4.2 cameras for simultaneous two channel acquisition, with a 63×/1.4 oil objective. For each focal plane 13 phase images were acquired. Lattice-SIM reconstruction was performed with the SIM processing Tool of the ZEN 3.0 SR Black v.16 (ZEISS Microscopy) software.
For live-cell high-throughput confocal microscopy, Huh7 cells were grown in 24-well glass bottom plates (170 μm coverglass bottom; Eppendorf, 0030741021; Cellvis, P24-1.5H-N). LDs were stained with 1 μM BODIPY 493/503 and nuclei were stained with 5 μg ml−1 Hoechst 33342 in DPBS for 30 min. Prior to imaging, cells were washed twice with DPBS and imaged in fresh phenol red-free medium supplemented with 10% FBS. Live cells were imaged using an Opera Phenix Plus High-Content Screening System (Perkin Elmer) confocal microscope equipped with a 40× water immersion objective using DAPI and GFP filters. Cells were imaged at 37 °C with 5% CO2. z-stacks of 0.3-μm slices were acquired.
Fluorescence microscopy analysis
Widefield and superresolution images were merged and brightness and contrast adjusted using Fiji/ImageJ. PLIN2+ and apoB+ LDs were quantified by hand. Nuclear pores were quantified by randomly selecting nuclei using the DAPI channel, outlining the nuclei manually, dividing the nucleus into 1 µm2 segments, and quantifying the number of foci per nucleus using the ‘analyzer particles’ function with a noise tolerance of 15. Foci were averaged per nucleus and graphed in Prism v.9 (Graphpad).
LDs and nuclear blebs (MLF2–GFP foci) were quantified from confocal images by creating custom analysis sequences using Harmony High Content Image Analysis Software v.4.9 (Perkin Elmer). For each field, maximum projection z-stacks were processed with advanced flatfield correction. Nuclei and cytoplasm were defined using the DAPI and GFP channels, respectively, and border cells were automatically excluded from analyses. LDs and nuclear blebs were defined using the ‘find spots’ building block (GFP channel), thresholding for size, intensity, and roundness. For each cell, LD number and area or number of nuclear blebs were quantified. Nucleus size and GFP intensity were also quantified using these methods. Quantification data were graphed and analysed in Prism 9 (GraphPad). The quantification of nuclear blebs (MLF2–GFP foci) and PLIN2 signal around the LDs in Huh7 cells expressing the various CLCC1 mutants (Supplementary Fig. 13) were performed following the pipeline described in Supplementary Fig. 13k, using an ImageJ macro (https://github.com/gparlakgul).
Transmission electron microscopy
For cell lines, Huh7 and U-2 OS cells were grown on 3 cm LabTek dishes and fixed in 2% paraformaldehyde and 2% glutaraldehyde in PBS. Samples were stained with 1% osmium tetroxide and 1.5% potassium ferrocyanide for 1 h and 1% uranyl acetate overnight. The next day, samples were washed and subsequently dehydrated in grades of ethanol (10 min each; 30%, 50%, 70%, 95% and twice for 10 min at 100%). Samples were embedded in increasing concentrations of eponate resin mixed with ethanol (30 min each; 1:2, 1:1, 2:1 and 100% acetone) followed by polymerization in 100% eponate overnight at 50 °C.
For liver tissues, mice were anaesthetized with 300 mg kg−1 ketamine and 30 mg kg−1 xylazine in PBS and perfused with 10 ml of DPBS followed by 10 ml of fixative buffer containing 4 parts of FP stock (2.5% PFA, 0.06% picric acid in 0.2 M sodium cacodylate buffer pH 7.4) and 1 part of 25% glutaraldehyde. After perfusion, small pieces (1–2 mm3) of liver were sliced at 300 μm thickness with a compresstome, transferred into a fresh fixative solution containing and incubated at 4 °C overnight. Samples were then washed in ice-cold 0.15 M sodium cacodylate buffer for 5 min, three times, and then incubated in 0.1 M sodium cacodylate solution containing 1% osmium tetroxide and 1.5% potassium ferrocyanide for 1 h at 4 °C. Samples were rinsed three times with water and incubated for 20 min in 1% thiocarbohydrazide and rinsed again three times for 5 min with water. Samples were incubated in 2% osmium tetroxide for 30 min and then rinsed three times for 5 min with water, followed by washing three times and incubation overnight at 4 °C in 1% uranyl acetate in maleate buffer. The next day, samples were washed and subsequently dehydrated in grades of acetone (10 min each; 50%, 70%, 90% and twice for 10 min at 100%). Samples were embedded in increasing concentrations of eponate resin mixed with acetone (30 min each; 50%, 70%, 90% and 100% acetone) followed by incubation in 100% eponate for 4 h. The samples were moved to fresh 100% eponate and polymerized at 65 °C for 24 h.
The resin-embedded sample blocks were trimmed, and 70 nm ultrathin sections were cut using a Leica UC6 ultramicrotome (Leica Microsystems) and collected onto formvar-coated slot grids. Sections were imaged to find target regions using a Tecnai 12 120 kV TEM (FEI) and data recorded using an Gatan Rio16 CMOS camera and GMS3 software (Gatan).
FIB-SEM
Mouse livers were fixed and prepared as described above. The trimmed sample blocks were glued with silver paint (Ted Pella) onto Al stubs, and sputter coated (Pd/Au) with a Tousimis sputter coater on top of a Bio-Rad E5400 controller. FIB-SEM imaging was performed using a Zeiss Crossbeam 550 (Carl Zeiss Microsystems). The sample was tilted at 54° in order to be perpendicular to ion beam. The FIB milling and SEM imaging of the target area were set up using Atlas 5 3D tomography (Carl Zeiss Microsystems). Slices with a thickness of 10 nm were milled from the target area using the 30 kV 300pA ion beam. Energy-selective Backscattered (ESB) images were collected at 1.5 kV, 1 nA, with a dwell time of 18 ns, image pixel size of 10 nm, and tilt correction angle of 36°. The collected images were aligned with slice registration in Dragonfly 2022.2 (Comet Technologies).
FIB-SEM data segmentation, quantification and visualization
Ground truth labels were generated by manually annotating each class (ER, mitochondria, nucleus and LDs) in five consecutive full-size images using Napari v.0.4.18. Tunable 2D-U-Net networks (DLSIA) were used to obtain rough predictions for each class41. These rough predictions were manually proofread and corrected in Napari. A block consisting of at least 250 × 250 × 250 voxels was used to train and fine-tune 3D-U-Net network models with Incasem42. Additional proofreading and manual corrections were performed in Napari. Objects, images, videos and quantifications from each class were generated using Arivis Vision 4D v.3.6.0 (ZEISS Microscopy).
Nuclear pores were manually labelled and annotated in Napari, image by image (1,142 images for wild type and 897 images for the knockout), in both datasets. After manual annotation, nuclear pore objects were reconstructed in Arivis Pro software to visualize the three-dimensional continuity and organization of the nuclear pores. To quantify the density distribution, the nuclear membrane surface was divided into ~100 patches using a Python 3.0 script (https://github.com/gparlakgul/nuclear_pore), with each patch assigned a unique pixel intensity as an identifier (Supplementary Fig. 15). These patches were imported into Arivis Pro. False nuclear surfaces (flat surfaces adjacent to the dataset borders) were excluded. Nuclear pores were assigned to the corresponding nuclear surface patch, and the number of nuclear pores per patch was calculated. The quantifications were normalized by the surface area of each nuclear membrane patch. Nuclear blebs were separated from the nuclear membrane, and the number of blebs was quantified.
Liver histology
Liver preparation was performed as described above with 4% PFA. Liver pieces were embedded in OCT, frozen, and cryosectioned into 5-µm-thick sections. Liver sections were fixed in 4% paraformaldehyde for 20 min and stained with either haematoxylin and eosin, oil red O, Masson’s trichrome, or picrosirius red by HistoWiz. Images were analysed using PathologyMap 2.0 software.
Primary hepatocyte isolation
Mice were anaesthetized using 300 mg kg−1 ketamine and 30 mg kg−1 xylazine in PBS. The livers were perfused with 50 ml of buffer I (11 mM glucose, 200 μM EGTA, 1.17 mM MgSO4 heptahydrated, 1.19 mM KH2PO4, 118 mM NaCl, 4.7 mM KCl and 25 mM NaHCO3, pH 7.32) through the portal vein with an osmotic pump set to the speed of ~4 ml min−1 until the liver turned pale. The speed was gradually increased until ~7 ml min−1 afterwards. When the entire buffer I had been infused, it was substituted for 50 ml of buffer II (11 mM glucose, 2.55 mM CaCl2, 1.17 mM MgSO4 heptahydrated, 1.19 mM KH2PO4, 118 mM NaCl, 4.7 mM KCl, 25 mM NaHCO3, BSA (fatty acid-free) 7.2 mg ml−1 and 0.18 mg ml−1 type IV collagenase (Worthington Biochemical, LS004188)), BSA and collagenase were added immediately before use. The buffers were kept at ~37 °C during the entire procedure. After perfusion, the primary hepatocytes were carefully released and sedimented at 500 rpm for 2 min, washed twice and suspended with Williams E medium supplemented with 5% CCS and 1 mM glutamine (Invitrogen, A1217601). To separate live from dead cells, the solution of hepatocytes was layered on a 30% Percoll gradient and centrifuged ~1,500 rpm for 15 min. The healthy cells were recovered at the bottom of the tube and plated in 35 mm imaging dishes for experimentation.
BODIPY 558/568 C12 incorporation assay
Huh7 safe-targeting control and CLCC1-KO cells were seeded in 60-mm plates at 350 cells per plate. To determine the rate of LD biogenesis, cells were incubated in BODIPY C12-BSA complex (complete medium with 1% BSA and 1 μM BODIPY 558/568 C12 (Invitrogen, D3835)) for 0, 1, 3 or 6 h. Cells were collected by washing them twice, collecting in cold DPBS, and transferring to Eppendorf tubes (Eppendorf, 022363352). Cells were centrifuged at 500g for 5 min, washed in DPBS, and centrifuged again. Cell pellets were stored at −80 °C until the lipid extraction step. For the lipolysis assay (measuring loss of esterified C12), cells were incubated in BODIPY C12-BSA complex for 16 h. Cells were then washed three times with medium and incubated in fresh medium for 1 h. Cells were then treated with 1 µg ml−1 triacsin C for 0, 6 or 24 h. Cells were collected, and pellets stored at −80 °C as described above.
TAG measurements by TLC
Cell pellets were thawed at room temperature and resuspended in 50 μl DPBS. Liver tissues (approximately 30 mg, 3 per mouse) were homogenized in 1 ml methanol using an immersion homogenizer for 5 min at 4 °C. Lipids were extracted by adding tert-butyl methyl ether (1.25 ml) and methanol (375 μl). The mixture was incubated on an orbital mixer for 1 h at room temperature. To induce phase separation, water (315 μl) was added, and the mixture was incubated on an orbital mixer for 10 min at room temperature. Samples were centrifuged at 1,000g for 10 min at room temperature. The upper organic phase was collected and subsequently dried in vacuo.
Dried lipid extracts were reconstituted in 30 μl (cells) or 200 μl (liver) chloroform/methanol (2:1, v/v). Lipids were then separated using HPTLC Silica gel 60 F254 plates (Sigma, 1137270001). Ten microlitres of the cell samples and 2 μl of the liver samples were spotted onto TLC plates and developed in CHCl3/ethanol/triethylamine/H2O (5:5:5:1, v/v). Plates were imaged on a ChemiDoc MP Imaging System (Bio-Rad Laboratories). Band densitometry analysis was performed using Image Lab v.6.0.1 (Bio-Rad Laboratories). The reported mean ± standard deviation was determined from three biological replicates.
Proteomic analysis of LD proteins
Safe-targeting and CLCC1-KO cell lines were grown until confluent in 500 cm2 plates of cells were collected by scraping in DPBS, centrifuged for 10 min at 500g, and stored at −80 °C. Buoyant fractions containing 1% SDS were acidified to a final concentration of 15% trifluoroacetate. Samples were then cooled on ice for 30 min and centrifuged at 20,000g for 30 min at 4 °C. The protein pellet was washed three times with 500 μl of ice-cold acetone and centrifuged for 10 min between each wash. The protein pellet was then dried in a vacuum evaporator for 10 min. Dried, precipitated proteins were resuspended in 0.1% RapiGest with 6 μl of sequencing-grade trypsin (Promega, 0.5 μg μl−1) added to each sample and digested overnight at 37 °C. Trypsinized samples were quenched with a final concentration of 5% TFA. Samples were desalted using the Waters Sep-pak 1cc (50 mg) C18 cartridge.
Peptides were resuspended in 1% formic acid and 0.5 μg of peptides were separated on an Easy nLC 100 UHPLC equipped with a 15 cm nano-liquid chromatography column. Using a flow rate of 300 nl min−1, the linear gradient was 5% to 35% over B for 90 min, 35% to 95% over B for 5 min and 95% hold over B for 15 min (solvent A 0.1% formic acid in water, solvent B 0.1% formic acid in acetonitrile). Peptide identified and relative abundances were determined using Proteome Discoverer v.2.4 (Thermo Fisher Scientific). Results are represented as mean ± s.d. of duplicates.
RNA sequencing
Triplicates of safe-targeting and CLCC1-KO cells were seeded in 6 cm plates. RNA was isolated using the Monarch Spin RNA Isolate Kit (New England Biolabs, T2110S). Sequencing results were analysed using Partek Flow (Illumina) running DESeq2 v.1.5.
ApoB ELISA assay
Safe-targeting and CLCC1-KO cells were seeded in 6 cm plates and treated with 1 µg ml−1 DMSO or 50 nM MTPi for 72 h. Twenty-four hours before collection, cells were changed into FBS- and phenol red-free medium. Medium was collected and apoB ELISA Assay (Sigma Aldrich, RAB069) was performed according to protocol. ApoB levels were normalized to cell protein levels and results are represented as mean ± s.d. of two biological duplicates.
ASGR luciferase assay
The ASGR reporter plasmid was generated by the laboratory of G. S. Hotamisligil as previously described15. Safe-targeting and CLCC1-KO cells were infected with lentivirus containing the ASGR construct. For the experiment, cells were changed to a fresh phenol red-free medium and incubated for 24 h with or without increasing doses of thapsigargin. 10 µl of medium was transferred to 96-well white plates (Corning, 3917) for luciferase assays following the manufacturer’s protocol. In brief, 50 µl of luciferase substrate (1 µM Cypridina (CLUC) or 10 mM coelenterazine (GLUC) in 100 mM tris buffer, pH 7.5) was added to the 10 µl medium and incubated in the dark for 5 min. The luminescence was read on Infinite 200 PRO plate reader using i-control software (Tecan).
Structure predictions
Monomeric and multimeric sequences were submitted to AlphaFold2 using MMseqs2 using either the Google Colabatory43 or COSMIC244 or were submitted to DMFold, MultiFOLD, or trRosetta45. The pLDDT of core homology regions as monomers and ring oligomers predicted by AlphaFold2: CLCC1 residues 209-353: monomer 79.2%, 16-mer: best 80.9%, average 80.2%; Brr6 residues 44-185: monomer 80.5%, 16-mer: best 78.1%, average 77.0%46,47.
General molecular dynamics simulation details
Simulations were performed with Gromacs (v.2023.340) using the molecular dynamics integrator (unless stated otherwise), and the Martini 3 force field (v.3.0.048) at a 20 fs time step. A 1.1 nm cut-off was used for reaction-field electrostatics and Van der Waals potentials49. Temperatures were held constant at 310 or 400 K (Supplementary Table 4) by the velocity rescaling50 thermostat (τT = 1 ps). Parrinello–Rahman51 semi-isotropic pressure coupling was applied to maintain a 1 bar reference pressure (τP = 12 ps, compressibility = 3 × 10−4). All images were rendered using PyMOL (v.2.5.0)52.
Simulations system setup
Coarse-grained representations of AlphaFold2 protein models were generated using martinize253, with secondary structure assignment by the DSSP (Dictionary of Secondary Structure in Proteins) algorithm54. Intermolecular and intramolecular orientations between backbone atoms within a 5–10 Å distance were constrained by an elastic network with a 500 kJ mol−1 nm−2 force constant, except for the flexible linkers (residue 164–172) in the full-length 16-mer. If specified in Supplementary Table 4, position restraints in x, y and z were applied to protein atoms with a 1,000 kJ mol−1 nm−2 force constant. Self-assembly runs (Supplementary Table 4a) were initiated from simulation boxes with a centred protein model and randomly placed DOPC molecules. DOPC membranes (Supplementary Table 4b,c) were built around the protein model using insane55. To avoid clashes between protein and lipid beads, DOPC molecules within 6 nm or 4 nm radii from the box centre (in xy) were removed from the system for the lower and upper bilayers, respectively (Supplementary Table 4b,c; see ‘initial setup’ in Extended Data Fig. 9c). All systems were solvated using Gromacs, including 150 mM NaCl and additional Na+ ions for charge neutralization. All simulations were preceded by energy minimization and a 5 ns number pressure temperature (NPT) equilibration.
Steps II–IV of simulating the CLCC1-induced fusion process (Supplementary Table. 4c and Extended Data Fig. 9c) required several additional settings:
-
Step II: To break the periodicity of the bilayers, the simulation box was expanded by 90 Å in the x and y dimensions, followed by recentring and resolvation with water and ions as described. Flat-bottom potentials (k = 500 kJ mol−1 nm−2) were applied to retain all DOPC lipid molecules in a box-centred cylinder with a 240 Å radius and 200 Å height to prevent them from crossing the periodic boundaries.
-
Step III: Van der Waals and electrostatic interactions between protein beads and the rest of the system (protein, lipid, water and ions) were switched off linearly using Gromacs’ lambda code (λ = 0 → λ = 1, with Δλ = 10−5 ns−1). The stochastic dynamics integrator was used with the flat-bottom potentials from step II. After completion, the now uncoupled protein atoms were deleted from the system.
-
Step IV: Membrane periodicity was restored by removing all lipid and solvent molecules outside of a 300 × 300 × 230 Å3 system-centred box. Simulation was continued without flat-bottom potentials.
CLCC1–lipid contact simulation analysis
For every frame (1 ns−1) in the three self-assembly simulations (Supplementary Table 4a), interactions (<7 Å) between the outermost protein sidechain beads (or backbone, for Gly) and any DOPC tail bead were counted. Contact frequency (%) was calculated as the fraction of simulation frames where a contact occurred, averaged over the eight dimers. The final contact map (Fig. 5f) was constructed by mapping the per-residue contact frequencies to the full-length CLCC1 AlphaFold model using the B-factor field in the PDB file (Supplementary Data 1, CLCC1_lipid_contact_map.pdb).
Statistics and reproducibility
In vitro assays were run as biological triplicates—three independently seeded and processed cultures on separate days—to demonstrate reproducibility across preparations and reduce batch effects; this level of replication is standard for mechanistic cell-biology readouts. Figure 3k and Extended Data Fig. 6j contain sample sizes of n = 2 due to technical limitations, and therefore statistics are not derived. Fluorescence and transmission electron microscopy images are representative of at least n = 10 imaged cells, except for Extended Data Fig. 6k which is n = 5 imaged cells. Batch retest CRISPR screens in Fig. 1 were performed in biological duplicates because of the large sample sizes and results are shown as ‘combination’ scores derived from casTLE analysis. Proteomics in Fig. 3c were performed as technical duplicates which is standard protocol for such a large dataset.
For in vivo work, we used n > 4 mice per group, balancing statistical precision with the 3Rs (replacement, reduction, refinement). Based on prior or pilot variability for our endpoints, this sample size was expected to detect large effect sizes (≥(1.5–2.0) × s.d.); smaller effects were outside the scope of this study and would motivate a larger, confirmatory cohort. Mice were divided between males and females and sexes are specified in figure legends and methods. All mouse experiments were produced twice with similar results, except for Fig. 2g,h and Extended Data Fig. 2j, k, which could only be performed as one replicate but were further supported by Fig. 2i,j and Extended Data Fig. 2l.
All FIB-SEM data in Fig. 5, Extended Data Fig. 7, and Supplementary Fig. 15 have a sample size of n = 1 cell because of the high density of data per cell.
All statistical analyses were performed using Prism 9 (GraphPad). For each panel, the number of biological replicates (n), P values, and statistical tests employed are reported in figure legends and methods.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.