Drosophila strains and culture
Flies (D. melanogaster) were raised on standard medium (inactivated yeast 6% (w/v); corn flour 6.66% (w/v); agar 0.9% (w/v); methyl-4hydroxybenzoate 22 mM) under a 12 h–12 h light–dark cycle at 18 °C with 60% humidity (unless mentioned otherwise). All of the experiments were performed on young (aged <5 days) adult flies. For behaviour experiments, groups of mixed-sex flies were used unless indicated otherwise. For imaging experiments involving surgery, female flies were used because of their larger size. All flies obtained from libraries or received after the injection of transgenes were outcrossed for five generations to a reference strain. In general, this reference line carried the w1118 mutation in an otherwise Canton-S genetic background; as an exception, and because TRiP RNAi transgenes are labelled by a y+ marker, lines from this collection were outcrossed to a y1w67c23 strain in an otherwise Canton-S background. To restrict UAS/GAL4-mediated expression to the adult stage, we used the TARGET system involving the ubiquitous expression of the thermosensitive GAL4 inhibitor under a tubulin promoter (tub-Gal80ts)29. In general, for crosses involving binary expression control systems (GAL4/UAS and/or LexA/LexAop), female flies carrying the driver transgene(s) were crossed to male flies carrying the effector transgene(s). A list of all of the single-transgene strains used in this study is provided in Supplementary Table 9. When needed, fly lines carrying combinations of multiple transgenes were obtained from these lines through routine crossing schemes.
Classical aversive or appetitive olfactory conditioning
To induce RNAi expression using the TARGET system, adult flies (aged 0 to 2 days) were kept at 30.5 °C for 3 days before conditioning. In the case of appetitive conditioning, flies were transferred to starvation vials (containing only a mineral-water-soaked cotton disk) for the last 16 h of the induction time. For experiments that did not involve thermal induction of transgene expression, experimental flies (aged 0-3 days) were transferred to fresh bottles containing standard medium 24 h before conditioning in the case of aversive conditioning experiments. For appetitive conditioning, flies (aged 0–2 days) were transferred to fresh food vials 1 day before being transferred to starvation vials for 21 h at 25 °C.
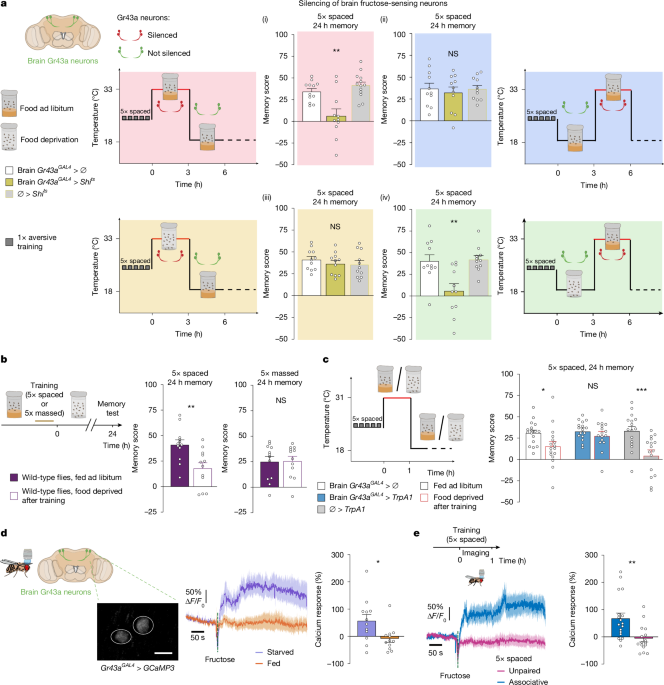
The aversive behaviour experiments, including sample sizes, were conducted similarly to other studies from our research group. Groups of 20–50 flies were subjected to one of the following olfactory conditioning protocols: a single cycle (1× training; duration of around 4 min), five consecutive associative training cycles (5× massed training; duration of around 20 min) or five associative cycles spaced by 15 min intertrial intervals (5× spaced training; duration of around 1 h 30 min). Non-associative control protocols (unpaired protocols) were also used for imaging experiments. Conditioning was performed using previously described barrel-type machines that allow parallel training of up to six groups. Throughout the conditioning protocol, each barrel was plugged into a constant air flow at 2 l min−1. For a single cycle of associative training, flies were first exposed to an odorant (the CS+) for 1 min while 12 pulses of 5-s-long 60 V electric shocks were delivered; flies were then exposed 45 s later to a second odorant without shocks (the CS−) for 1 min. The odorants 3-octanol and 4-methylcyclohexanol, diluted in paraffin oil to a final concentration of 2.79 × 10−1 g l−1, were alternately used as conditioned stimuli. In all of the behaviour experiments, two conditioning protocols were conducted sequentially on two batches of genotypically identical flies: a first set of flies was conditioned with 3-octanol as CS+ and 4-methylcyclohexanol as CS−, and a second reciprocal set with 4-methylcyclohexanol as CS+ and 3-octanol as CS−. This enabled us to balance for potential systematic choice bias during memory retrieval test (see below), as is commonly done in Drosophila memory experiments. During unpaired conditionings, the odour and shock stimuli were delivered separately in time, with the onset of electric shock delivery occurring 3 min and terminating 2 min before the first odorant.
The appetitive behaviour experiments, including the sample sizes, were conducted similarly to other studies from our research group. During appetitive conditioning, flies were first exposed to an odorant (the CS+) for 1 min paired with a dried sugar reward, followed 45 s later by a second odorant (the CS−) presented without reward for 1 min, as described previously46. The odorants were the same, and were used at the same concentration, as for the aversive conditioning experiments. Plexiglas tubes covered with dried sugar were prepared the day before conditioning: a 1.5 M sucrose solution in mineral water was spread on the tube surface and tubes were placed in front of a ventilator overnight at room temperature.
Test of memory retrieval
For aversive memory behaviour experiments, unless indicated otherwise in the figures, flies were kept on standard medium between conditioning and the memory test, at either 25 °C for flies tested 3 h after 1× training, or at 18 °C for flies tested 24 h after training. After 1× training, to assess separately the two components of memory formed after 1× training (middle-term memory, which is anaesthesia sensitive, and anaesthesia-resistant memory), a subset of the trained groups were subjected to a 4 °C cold treatment for 2 min, 2 h after training (that is, 1 h before the memory test). For appetitive memory behaviour experiments, flies were kept in starvation vials between conditioning and the memory test. The memory test was performed in a T-maze apparatus, typically 3 h after single-cycle training or 24 h after massed or spaced training. Each arm of the T-maze was connected to a bottle containing 3-octanol or 4-methylcyclohexanol, diluted in paraffin oil to a final concentration identical to the one used for conditioning. Flies were given 1 min in complete darkness to choose between either arm of the T-maze. A score was calculated as the number of flies in the CS− arm of the T-maze minus the number of flies in the CS+ arm, divided by the total number of flies in both arms. To balance for potential systematic choice bias due to a preference for either side of the maze, we combined the performance of flies conditioned with either 3-octanol or 4-methylcyclohexanol as the CS+ condition. Thus, a single performance index value reported on graphs is the average of two scores obtained from two groups of genotypically identical flies conditioned sequentially using either odorant (3-octanol or 4-methylcyclohexanol) as the CS+ condition. In particular, for cases that required counting a subset of the assayed flies after the memory test (for example, disaggregated evaluation of female and male flies, or the presence of a balancer chromosome in a parental line), scores involving fewer than six flies in total were discarded to avoid giving disproportionate statistical importance to a small number of flies. The indicated n is the number of independent performance index values for each genotype.
Innate shock avoidance assessment
Shock-response tests were performed at 25 °C by placing flies in two connected chambers identical to those used for olfactory conditioning. Electric shocks were delivered in only one of the compartments. Flies were given 1 min to move freely in these compartments, after which they were trapped, collected and counted. The compartment where the electric shocks were delivered was alternated between two consecutive groups. Shock avoidance was calculated as for the memory performance.
Innate olfactory acuity assessment
As the delivery of electric shocks can modify olfactory acuity, our olfactory avoidance tests were performed on flies that had first been presented with another odour paired with electric shocks. Innate odour avoidance was measured in a T-maze similar to those used for memory tests, in which one arm of the T-maze was connected to a bottle with odour diluted in paraffin oil at the same concentration as that used for olfactory conditioning, and the other arm was connected to a bottle with paraffin oil only. Naive flies were given the choice between the two arms during 1 min. The odour-interlaced side was alternated for successive tested groups. At these concentrations, both odorants (octanol and methylcyclohexanol) are innately repulsive.
Sugar preference assessment
Innate sugar preference assessment was measured at 25 °C in a T-maze similar to those used for memory tests, in which only one of the two arms of the T-maze was covered with dried sugar. Naive flies were given the choice between the two arms during 1 min. The tests were performed on starved flies submitted to the thermal RNAi induction protocol described above. The arm with sugar was placed alternately on the right or left between two consecutive groups. Sugar response was calculated as for the memory performance.
In vivo pyruvate and glucose imaging
Pyruvate imaging experiments were performed on flies expressing the pyruvate sensor Pyronic in MB neurons through the VT30559-GAL4 driver or 13F02-LexA driver in combination with either the UAS-Pyronic or LexAop-Pyronic line, which were previously described14,16. Glucose imaging experiments were performed on flies expressing the glucose sensor FLII12Pglu-700μδ6 in MB neurons through the 13F02-LexA driver, in combination with LexAop-FLII12Pglu-700μδ6. RNAis were expressed in MB neurons using the inducible tub-GAL80ts; VT30559-GAL4 driver. Crosses for imaging experiments were raised at 23 °C. To achieve the induction of RNAi expression, adult flies were kept at 30.5 °C for 3 days before conditioning. Data were collected indiscriminately from 30 min to 1.5 h after 5× spaced training. A single fly was picked after few-second immobilization in a prechilled tube, and prepared for imaging using an established method14. The head capsule was opened, and the brain exposed by gently removing the superior tracheae. The head capsule was bathed in artificial haemolymph solution for the duration of the preparation. The composition of this solution was as follows: NaCl 130 mM (Sigma-Aldrich, S9625), KCl 5 mM (Sigma-Aldrich, P3911), MgCl2 2 mM (Sigma-Aldrich, M9272), CaCl2 2 mM (Sigma-Aldrich, C3881), D-trehalose 5 mM (Sigma-Aldrich, 9531), sucrose 30 mM (Sigma-Aldrich, S9378) and HEPES hemisodium salt 5 mM (Sigma-Aldrich, H7637). At the end of surgery, any remaining solution was wicked away and a fresh 90 μl droplet of this solution was applied on top of the brain. Two-photon imaging was performed using the Leica TCS-SP5 upright microscope equipped with a ×25/0.95 NA water-immersion objective. Two-photon excitation was achieved using a Mai Tai DeepSee laser tuned to 825 nm (for pyruvate sensor) or 820 nm (glucose sensor). Light emission was collected on external PMTs in the spectral ranges 468–500 nm and 529–556 nm for the blue and yellow fluorophores, respectively. Images were acquired using Leica LAS-AF (v.2.7.3).
Measurements of mitochondrial pyruvate consumption were performed as previously described14,16,42. This consists in pharmacological blocking of the mitochondrial respiratory chain by sodium azide, which induces an acute increase in cytosolic pyruvate, at a rate that reflects the pyruvate consumption rate before blockade. Two-photon images were acquired in both channels at a frame rate of two images per second. After 1 min of baseline acquisition, 10 µl of a 50 mM sodium azide solution (Sigma-Aldrich, 71289; prepared in the same artificial haemolymph solution) was injected into the 90 µl droplet bathing the fly’s brain, bringing the sodium azide to a final concentration of 5 mM. To analyse the pyruvate imaging experiments, regions of interest (ROIs) were delimited by hand around each visible MB vertical lobe and the average intensity of both mTFP and Venus channels over each ROI were calculated over time after background subtraction. The Pyronic sensor was designed so that FRET from mTFP to Venus decreases when pyruvate concentration increases. To obtain a signal that positively correlates with pyruvate concentration, the inverse FRET ratio was computed as the mTFP intensity divided by the Venus intensity. This ratio was normalized to the baseline value calculated over the 30 s before drug injection. The rate of azide-induced pyruvate surge was calculated as the slope between 10 and 70% of the plateau.
Cellular glucose consumption measurements were performed as previously described15. This consisted of pharmacological inhibition of the enzyme trehalase by application of validamycin A, resulting in a decrease in the cytosolic glucose concentration. The kinetics of the glucose decrease were reflective of the rate of glucose consumption. Two-photon images were acquired in both channels at a frame rate of 1 image per second. Validamycin A (Sigma-Aldrich, 32347) was directly diluted into the artificial haemolymph solution at a final concentration of 40 mM, aliquoted and stored at −20 °C. A freshly thawed aliquot was used for every fly. After 2 min of baseline acquisition, 10 μl of the solution was added to the 90 μl saline droplet on top of the brain, bringing validamycin A to a final concentration of 4 mM. The signal was then acquired for another 14 min. ROIs were delimited by hand around the labelled ROIs (somas of MB neurons). The average intensity of the YFP and CFP channels over each ROI was calculated over time after background subtraction. The FRET ratio (YFP/CFP) of the FLII12Pglu-700μδ6 glucose sensor was computed to obtain a signal that positively correlates with glucose concentration. This ratio was normalized to the baseline value calculated over the 30 s before drug injection. As the decrease in FRET signal was not linear, the area over the curve (AOC) was calculated as a metric of the decrease that was positively correlated with glucose consumption. The AOC was calculated as the integral between 200 s and 900 s of the acquisition. The indicated n value is the number of flies that were assayed in each condition.
In vivo calcium imaging
Calcium imaging experiments were performed by expressing the genetically encoded calcium reporters GCaMP3 or GCaMP6s in Gr43a neurons, and GCaMP6f in dFB neurons. GCaMP3 was initially chosen to image Gr43a neurons as its higher baseline fluorescence enabled localization of the neurons, which we could not achieve with the GCaMP6f sensor. Using GCaMP6s, another GCaMP6 variant with higher baseline fluorescence, we could verify that a more-sensitive sensor and a lower fructose concentration yielded similar results to GCaMP3. Fly preparation was performed as described above for in vivo pyruvate and glucose imaging. Two-photon imaging was performed on the Leica SP8 DIVE microscope equipped with spectrally tunable hybrid detectors and a ×25/1.0 NA water-immersion objective, coupled to an Insight X3 dual-emission IR laser (Spectra Physics). Two-photon excitation was tuned at 920 nm. Emitted light was collected in the 492–570 nm spectral range. Images were acquired with Leica LAS-X software v.3.5.7. Images were acquired at a rate of one frame every 385 ms for a total of 7 min. For experiments performed on starved flies, flies were starved for 24 h at 25 °C before imaging. The composition of the physiological solution for starved flies (Fig. 1d) was the same as used for glucose imaging, except that it contained 36 mM ribose (Sigma-Aldrich, W379301) instead of sucrose and trehalose, as in ref. 37. For fructose-response experiments, fructose was added at a final concentration of 75 mM (or 20 mM when using GCaMP6s) after 120 s. This fructose concentration was previously shown to elicit Gr43a responses in the fly brain23. Image alignment was performed using the Template Matching plugin in Fiji to correct for motion across frames and ensure accurate measurement of calcium responses. After alignment, ROIs for data analysis were delimited by hand and the signal was calculated over time after background subtraction and normalized to the baseline value calculated over the 30 s before fructose injection. Calcium response was calculated as the average from the injection time to the end of the recording. For experiments measuring the spontaneous calcium activity in dFB neurons, signals were analysed as reported previously26.
Sucrose preference assay using FlyPad
The food-choice assay was done using FlyPAD44 according to the manufacturer’s instructions. One channel of the arena was loaded with 3 μl of 1% agar mixed with sucrose 0.6 M (Sigma-Aldrich), while the other channel was loaded with 3 μl of 1% agar. Single flies were captured by mouth aspiration and gently blown into each arena. Data were acquired for 60 min. Interaction with either channel (number of sips) was extracted for each arena. Flies showing zero interaction with both electrodes were not taken into account for further analysis. A sucrose preference index (PI) for each fly was calculated as: (interactions with sucrose − interactions with agar)/(interactions with sucrose + interactions with agar).
Lgr1-HA line generation
The Lgr1-HA line was generated using CRISPR–Cas9 (outsourced to Rainbow Transgenic Flies). The 3×HA sequence (TACCCATACGATGTTCCTGACTATGCGGGCTATCCCTATGACGTCCCGGACTATGCAGGATCCTATCCATATGACGTTCCAGATTACGCT) was preceded by a GS linker (GGTGGCGGCGGAAGCGGAGGTGGAGGCTCG) and inserted at the end of the Lgr1 coding sequence using a guide RNA (5′-ATTATGTTTAAACCGAACCCGGG-3′). A loxP-flanked sequence of the mini-white gene was added after the Lgr1 coding sequence to provide a phenotypic marker of the presence of the HA-tagged construct.
Immunohistochemistry
Before dissection, 2- to 4-day-old female flies from appropriate crosses were fixed in 4% paraformaldehyde in PBST (PBS containing 1% Triton X-100) at 4 °C overnight. Brains were dissected on ice in PBS solution and rinsed three times for 20 min in PBST, then blocked with 2% BSA in PBST for 2 h. Next, brains were incubated with primary antibodies. For primary antibodies, this study used 1:400 rabbit anti-GFP (Invitrogen, A11122), 1:200 rat anti-Gpb541 (provided by P. Herrero), 1:200 rat anti-HA (Roche, 11867423001) and 1:100 mouse anti-nc82 (DSHB, nc82). Primary antibodies were incubated in the blocking solution (2% BSA in PBST) at 4 °C overnight. The next day, brains were rinsed three times for 20 min with PBST and then incubated for 3 h at room temperature with secondary antibodies diluted in blocking solution. For secondary antibodies, we used 1:400 anti-rabbit conjugated to Alexa Fluor 488 (Invitrogen, A11034), 1:400 anti-rat conjugated to Alexa Fluor 594 (Invitrogen, A11007), 1:400 anti-mouse conjugated to Alexa Fluor 594 (Invitrogen, A11005) and 1:400 anti-rat conjugated to Alexa Fluor 488 (Invitrogen, A11006). Brains were then rinsed once in PBST for 20 min, and twice in PBS for 20 min. After rinsing, brains were mounted in Prolong Mounting Medium. Images were acquired with a Nikon A1R confocal microscope using Nikon NIS-Element v.4.40 software or with an Olympus BX61-FluoView FV1000 confocal microscope using FV10-ASW v.4.2 software, as z-stack slices in 1 µm steps. For expression patterns of genetic driver lines, maximum projections are shown, obtained using Fiji (Image J1.52p).
RT–qPCR analyses
To validate the efficiency of the knockdowns used in this study, the mRNA of the target gene was measured using RT–qPCR. Female flies carrying the elav-GAL4 pan-neuronal driver were crossed with either male flies carrying the specified UAS-RNAi or with CS male flies, and the resulting crosses were reared at 25 °C. Adult fly progeny (aged 0–1 days) were transferred to fresh food for 1 day before RNA extraction. RNA extraction and cDNA synthesis were performed as described previously16 using the same reagents: the RNeasy Plant Mini Kit (Qiagen), the RNA MinElute Cleanup kit (Qiagen), 614 oligo(dT)20 primers and the SuperScript III First-Strand kit (Life Technologies). The cDNA level for each gene of interest was compared against the level of the αTub84B (CG1913) reference cDNA. Amplifications were performed using a LightCycler 480 (Roche) and the SYBR Green I Master mix (Roche). Reactions were carried out in triplicate. The specificity and size of amplification products were assessed by melting curve analyses. Expression relative to the reference was expressed as a ratio (2−ΔCp, where Cp is the crossing point). RT–qPCR results are presented in Supplementary Table 5. The primers used in this study are described in Supplementary Table 6.
Quantification and statistical analysis
Sample sizes in this study were similar to previous studies from our or other groups for aversive memory assays14, appetitive memory assays37, metabolic imaging15,16 and calcium imaging9, rather than being predetermined by statistical power estimation. As group assignment was based on the fly genotype, random assignment of the experimental animals could not be performed, and experimenters were not blinded to the group assignment during experiments. For memory assays, a single datapoint is the mean of two scores from two groups of flies of the relevant genotype conditioned with octanol or methycyclohexanol as the odorant paired with electric shocks, as described above, which represents an experimental replicate. For in vivo imaging experiments and immunohistochemistry, one replicate corresponds to one fly brain. For RT–qPCR experiments, one replicate corresponds to the average value of three separate measurements (triplicate) done in parallel on the same biological sample. For FlyPad experiments, one replicate corresponds to one fly. All behavioural experiments were run at least twice and in vivo imaging experiments were run at least three times, on different days and with different batches of flies. All replicates are included in figures and extended data figures. Memory analyses of Gr43a neurons and dFB neurons were replicated in independent series of experiments, using different genetic drivers targeting Gr43a neurons or dFB neurons. Memory analyses involving gene knockdown in Gr43a neurons were replicated in independent series of experiments with two different RNAi constructs, except Gpa2 for which only one RNAi line was publicly available. All replicates were successful. In general, comparisons of the data series between two conditions were achieved by a two-tailed unpaired t-test. Comparisons between more than two distinct groups were made using a one-way ANOVA test, followed by Tukey pairwise comparisons between the experimental group and its controls, or using a two-way ANOVA followed by Sidak pairwise comparison (Fig. 1c). ANOVA results are presented as the value of the Fisher distribution F(x,y) obtained from the data, where x is the number of degrees of freedom between groups and y is the residual number of degrees of freedom for the distribution. For the analysis of sucrose preference indices obtained with FlyPAD, data series were intrinsically non-normally distributed, therefore nonparametric statistical tests were applied, that is, Mann–Whitney test for pairwise comparisons between two conditions and Kruskal–Wallis test for comparisons across multiple groups, followed by Dunn’s multiple-comparison test between the experimental group and its controls. Statistical tests were performed using the GraphPad Prism v.9.0. On the figures, asterisks illustrate the significance level of the t-test or Mann–Whitney U-test, or of the least significant pairwise comparison after an ANOVA or –Kruskal–Wallis test.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.