Animals
Animal experiments were performed following standard ethical guidelines (European Communities Guidelines on the Care and Use of Laboratory Animals, 86/609/EEC), and were approved by the Veterinary Department of the Canton of Basel-Stadt. C57BL6, PV-cre (The Jackson Laboratory, 008069) mice and PV-cre mice crossed with Ai9(RCL-tdT) mice (The Jackson Laboratory, 007909) in the C57BL6 background were used at 55–120 days old. Both females and males were used for this study. They were maintained on a normal 12 h–12 h light–dark cycle in a pathogen-free environment with ad libitum access to food and drinking water. Mice were euthanized by exposure to CO2. No formal statistical methods were used to predetermine sample sizes. Mice were allocated randomly to treatment conditions. No blinding to group allocation was performed during the experiments.
Post-mortem human retina donations
Human retina tissue was obtained from multi-organ donors through the sampling of non-transplantable eye tissue extracted during cornea collection for transplantation. Retinas from individuals with a documented history of eye disease were excluded. The collection of tissue samples adhered to the principles outlined in the Declaration of Helsinki, and all experimental protocols received the approval of the local ethics committee.
HEK293T cells
All cell cultures were maintained at 37 °C in a humidified incubator (5% CO2). HEK293T cells (ATCC, CRL-3216) were cultured in DMEM (Gibco, 10566016) supplemented with 10% FBS and 1% penicillin–streptomycin (Gibco, 15140-122). Cells were passaged at 90% confluency. Cell dissociation was performed using TrypLE Express (Thermo Fisher Scientific, 12604021).
Bone marrow mMSCs
Bone marrow mMSCs were obtained from Cyagen (MUBMX-01001). Cells were cultured in MesenCult Proliferation medium (StemCell Technologies, 05411) up to passage 11. Cells were passaged with TrypLE Express at 70–80% confluency.
Human cardiac cells
Human cardiac cells isolated from the ventricles of the adult heart (Promocell, C-12810) were maintained in myocyte growth medium (Promocell, C-22070). For passaging, cells were detached with TrypLE Express at 70% confluency and seeded at a density of 10,000–15,000 cells per cm2.
Human cardiac fibroblasts
Human cardiac fibroblasts were obtained from LifeLine Cell Technology (FC-0060) and were maintained in FibroLife S2 Fibroblast Medium (Complete Kit, LifeLine Cell Technology, LL-0011) supplemented with 1% penicillin–streptomycin (Gibco, 15140-122). Cells were grown in a dish precoated with attachment factor (Thermo Fisher Scientific, S006100) and passaged with TrypLE Express at 70–80% confluency.
HUVECs
Primary HUVECs isolated from the umbilical cord vein (Sigma-Aldrich, C-12203) were maintained in endothelial cell growth medium (Bio-techne, CCM027). Cells were passaged at 80–90% confluency and seeded at 5,000–10,000 cells per cm2 density. Cells were dissociated using TrypLE Express. For co-culturing experiments, HUVECs were cultured together with cardiac fibroblasts in 1:1 ratio (20,000 cells in total) in an eight-well slide (Ibidi, 80826-IBI) using endothelial cell growth medium.
Generation and maintenance of induced human neurons
Neuronal differentiation was performed as described previously24 with some modifications. For inductions, IMR90 (iPS(IMR90)-4; WiCell, WISCi004-B)) and patient-derived LHON (mt11778G>A, iPS(HS-0565-B-LHON-C12), this study) iPS cell lines. Freshly thawed iPS cells were cultured in mTeSR Plus medium for several passages. At 70–90% confluency, iPS cells were washed with PBS and dissociated with 0.5 mM EDTA (Invitrogen, 15575020) to form small clumps. The clumps were seeded onto six-well plates coated with Matrigel growth-factor reduced (Corning, 356230) in mTeSR Plus medium and incubated for 1 h at 37 °C (5% CO2). After incubation, mTeSR Plus was replaced with N2B27 medium (DMEM/F12 with GlutaMAX) (Gibco, 31331-028) and Neurobasal (Gibco, 21103-049) medium in 1:1 ratio, 0.5% additional GlutaMAX (Thermo Fisher Scientific, 35050038), 1% N2 (Gibco, 17502-048), 2% B27 without vitamin A (Gibco, 12587-010) and 1% penicillin–streptomycin (Gibco, 15140-122) containing 2% Matrigel growth factor reduced and incubated overnight at 37 °C (5% CO2). The next day, the medium was exchanged with fresh N2B27 supplemented with 25 μM forskolin (StemCell, 100-0249). Cells were fed every second day. After 35 days post-induction, cells were dissociated using TrypLE Express (Thermo Fisher Scientific, 12604021), diluted in N2B27 medium, filtered through a 70-μm cell strainer (pluriSelect, 43-50070-51) and centrifuged for 10 min at 300g at 4 °C. The cell pellet was resuspended in MACS buffer (80 µl for 107 cells) containing autoMACS Rinsing solution (Miltenyi Biotec, 130-091-222) and 0.5% BSA solution (Miltenyi Biotec, 130-091-376). CD90 microbeads (Miltenyi Biotec, 130-096-253) were added to the cells (20 µl for 107 cells) and incubated for 15 min on ice. CD90+ cells were sorted on a MidiMACS magnetic separator in an LS column (Miltenyi Biotec, 130-042-302, 130-042-401). The magnetically enriched CD90+ cell population was counted on the Countess system (Invitrogen). Cells were centrifuged at 4 °C for 10 min at 300g, resuspended in N2B27 and the sorted cell suspension was subsequently seeded onto a Matrigel-coated plate. Seeded cells were fed every second day with N2B27 supplemented with 10 μM forskolin and 10 ng ml−1 CNTF (Peprotech, 450-13). Experiments used sorted cells at 80 to 130 days after induction.
Induced pluripotent stem cell culture
The human iPS cell lines 01F49i-N-B733 and IMR90 (iPS(IMR90)-4, WiCell; WISCi004-B) and the IMR90-tdTomato reporter line (IMR90-4 AAVS1-tdTomato, developed according to a published protocol54) were used. Cells were cultured at 37 °C and 5% CO2 in a humidified incubator. mTesR1 and mTeSR Plus media were used (StemCell Technologies, 85850, 100-0276) on Matrigel-coated six-well plates (Corning, 356230). The culture medium was replaced daily, and cells were passaged weekly using 0.5 mM EDTA (Invitrogen, 15575020) in PBS without applied CaCl2/MgCl2, for 3–5 min. Cells were tested for mycoplasma on a regular basis using the MycoAlert PLUS Mycoplasma Detection Kit (Lonza, CABRLT07–710).
Reprogramming and characterization of LHON iPS cells
The iPS cell line IOBHS-i-0565B-12 (short name, LHON) was derived from coded male donor peripheral blood mononuclear cells (PBMCs) with the m11778G>A mutation in the mitochondrial ND4 gene, 100% homoplasmic. Blood samples were received in ACD solution B (BD, 367756) then diluted 1:1 in PBS (Thermo Fisher Scientific, 70011051) and applied over lymphocyte separation medium 1077 (Promocell, C44010). The samples were centrifuged, PBMCs were isolated and resuspended in RPMI medium (Gibco, 21875). Activation of PBMCs for 1 day was completed using Stemspan II medium (StemCell Technologies, 09605) and erythrocyte supplement (StemCell Technologies, 02692). PBMCs were reprogrammed using the CytoTune-iPS Reprogramming Kit 2.0 (Thermo Fisher Scientific, A16517) according to the manufacturer’s instructions. In brief, 3.5 × 106 PBMCs were plated in one well of a 24-well ultra-low-attachment culture plate (Sigma-Aldrich, CLS347) with StemSpan medium and infected with Sendai virus at a multiplicity of infection (MOI) of 5:5:3 (KOS, MOI = 5; hc-MYC, MOI = 5; hKLF4, MOI = 3) for approximately 16 h. On day 3 after transfection cells were plated in different densities from 10,000 to 25,000 cells on six-well Matrigel-coated plates. On day 7, 50% of the medium was replaced by Essential 8 Flex medium (Thermo Fisher Scientific, A2858501). After iPS cell colonies formed, they were picked, plated on 24-well Corning Synthemax-R plates (Corning, 3979), cultured in Essential 8 FLEX medium (Thermo Fisher Scientific, A2858501) and transitioned to six-well Corning plates (Corning, 3506). iPS cells were subsequently cultured in mTeSR Plus medium (StemCell Technologies, 100-0276) up to passage 34 when cells were characterized and used for experiments.
iPS cell pluripotency was assessed 4–6 days after splitting by staining iPS cell colonies cultured in six-well plates for alkaline phosphatase using the alkaline phosphatase live stain according to the manufacturer’s instructions (Thermo Fisher Scientific, 12807552). To assess pluripotency iPS cells were seeded into Matrigel-coated (Corning, 354277) four-well chamber slides (Vitaris, 80426-IBI) and further stained for the pluripotency markers SOX2 (1:200; Millipore, AB5603), OCT4 (1:100; Abcam, Ab181557), NANOG (1:200; R&D Systems, AF1997) and TRA-1-60 (1:200; BD Pharmingen, 560193). The potential to differentiate into the three embryonic germ layers was assessed by directed differentiation into ectoderm; nestin (1:200, Sigma-Aldrich, N5413) and PAX6 (1:200, ATLAS, AMAb91372), mesoderm; Brachyury (1:200, R&D Systems, AF2085) and endoderm; FOXA2 (1:400, Abcam, ab108422) and SOX17 (1:200, R&D Systems, AF1924) using the STEMdiff Trilineage Differentiation Kit (StemCell Technologies, 05230) according to the manufacturer’s instructions on cells maintained in Matrigel-coated (Corning, 356230) four-well chamber slides (Vitaris, 80426-IBI). Secondary antibodies used were Invitrogen, A10037, A11055 and A21206 at 1:1,000. To test for chromosomal aberrations, genomic integrity testing was performed (StemGenomics) on genomic DNA extracted from iPS cells by the DNeasy Blood & Tissue Kit (Qiagen, 69504).
Generation and maintenance of retinal organoids
Retinal organoids were generated as described previously33,55. iPS cells were detached using EDTA at 0.5 mM for 3 min. EDTA was then removed and Accutase was added (Thermo Fisher Scientific, 00-4555-56) for 4 min at 37 °C to generate single-cell suspensions of which 300 to 600 cells were placed per microwell in an 81-microwell hydrogel. The hydrogels were generated using the MicroTissues 3D Petri Dish micro-mould (Sigma-Aldrich, Z764000) and 2% agarose (Thermo Fisher Scientific, R0491). Each hydrogel carrying the iPS cells was cultured in a 12-well plate (Corning, 3513) in neural induction medium DMEM/F12 (Gibco, 31331-028), 1% N2 supplement (Gibco, 17502-048), 1% NEAA solution (Sigma-Aldrich, M7145) and 2 mg ml−1 heparin (Sigma-Aldrich, H3149-50KU) for 1 week with daily medium exchanges. Embryoid bodies formed in each microwell were detached from the hydrogel and distributed across three wells of a Matrigel-coated six-well plate. Organoids in six-well plates were cultured with daily medium exchanges and began to form 2D confluent structures. For the first 16 days, they were cultured in neural induction medium. Subsequently, the medium was changed to 3:1 medium (3 parts DMEM (Gibco, 10569–010):1 part F12 medium (Gibco, 31765–027), supplemented with 2% B27 without vitamin A (Gibco, 12587010), 1% NEAA solution (Merck, M7145) and 1% penicillin–streptomycin (Gibco, 15140–122). Between days 28 and 30 of culture, checkerboard scraping was performed as previously described33. From week 6, organoids were cultured in 3:1 medium supplemented with 10% heat-inactivated FBS (Millipore, es009–b) and 100 mM taurine (Sigma-Aldrich, T0625–25 G) with medium changes three times per week. At week 10, the culture medium was supplemented with 1 mM retinoic acid (Sigma-Aldrich, R2625). From week 14, the B27 supplement in 3:1 medium was replaced by N2 supplement (Gibco, 17502–048) and retinoic acid was reduced to 0.5 mM.
Generation and maintenance of vascular organoids
Vessel organoids were induced from the IMR90 AAVS1-tdTomato human iPS cell line. The iPS cell reporter line was generated by insertion of a reporter construct into the safe harbour AAVS1 locus with TALEN technology as described previously54. The growth and cultivation of blood vessel organoids was carried out as previously described with minor modifications37. Human iPS cells were dissociated into single cells using Accutase (Invitrogen, E141421) and 150 cells per microwell were distributed in a 256-microwell hydrogel (2% agarose) in mTeSR1 containing the CEPT cocktail (Tocris, 7991). On day 1 after induction, the embryoid bodies were treated with N2B27 with 12 µm CHIR99021 (StemCell, 72054) and 30 ng ml−1 BMP4 (StemCell Technologies, 78211). On day 4, the medium was changed to N2B27 100 ng ml−1 VEGF-A (Peprotech, 100-20) and 30 ng ml−1 forskolin (Sigma-Aldrich, F3917). On day 7, the embryoid bodies were embedded in collagen I (Sigma-Aldrich, CC300) Matrigel mix and treated with StemPro34 (Thermo Fisher Scientific, 10639011) containing 15% inactivated FBS (Merck, 21C662-A), 100 ng ml−1 VEGF-A and 100 ng ml−1 FGF-2 (Miltenyi Biotec, 130-093-564). On day 11, individual blood vessel organoids were dissected out of the gel and maintained in 96-well ultra-low-attachment plates until day 16.
Generation of mtDNA-depleted cells
mtDNA-depleted HEK293T cells and mMSCs were generated by treatment with a combination of 2′,3′-dideoxycytidine (ddC) and ethidium bromide (EtBr) according to established protocols56,57. For HEK293T cells, the base medium (DMEM + GlutaMAX containing sodium pyruvate; Thermo Fisher Scientific, 10569010) was supplemented with 10% FBS (Sigma-Aldrich, F7524), 1% penicillin–streptomycin (Thermo Fisher Scientific, 15140122), 62 μg ml−1 uridine (Thermo Fisher Scientific, A15227.14), 40 µM ddC (MedChemExpress, HY-17392) and 0.5 μg ml−1 EtBr (Thermo Fisher Scientific, 15585011). For mMSCs, they were cultured in MesenCult Proliferation Kit medium (StemCell Technologies, 05411) with 1 mM sodium pyruvate (Thermo Fisher Scientific, 11360070) and the same additives were used with a final concentration of 200 μM ddC and 1 μg ml−1 EtBr. The medium was replaced every 2 days. After mtDNA depletion, ddC and EtBr were removed from the medium and cells were maintained in culture medium containing uridine and sodium pyruvate. The depletion was monitored using PCR as previously described. Total DNA was extracted using the DNeasy Blood & Tissue Kit (Qiagen, 69506). The copy number of the mtDNA was assessed using droplet digital PCR (ddPCR). The assay was performed using the ddPCR Supermix for Probes (Bio-Rad, 1863010), as described in the product’s technical bulletin. In brief, the reaction mixes (22 μl per reaction) were prepared as follows: 500 nM primers, 250 nM probes, 1× ddPCR Supermix for probes and 1 ng of DNA. The reactions were loaded into DG8 cartridges (Bio-Rad, 1864008) with droplet generation oil for probes (Bio-Rad, 1863005). The cartridge was then fitted with a DG8 gasket (Bio-Rad, 1863009) and run on the QX200 droplet generator (Bio-Rad, 10031907). Subsequently, the droplet–oil emulsions were transferred to a ddPCR-compatible 96-well plate that was sealed with a PX1 PCR Plate Sealer (Bio-Rad, 1814000) and the PCR reactions were run on a Mastercycler X50s (Eppendorf, 6311000010) according to the PCR protocol detailed in the technical bulletin. Afterwards, data were acquired on the QX200 Droplet Reader (Bio-Rad, 1864003) using the QuantaSoft software (v.1.7.4.0917, Bio-Rad). Negative controls were processed in parallel, where only ddH2O and non-mtDNA depleted cells were used as input. Primers for ND1 (for HEK293T)58 and Nd5 (for mMSCs)57 genes were used for mtDNA amplification. For nuclear genome amplification, primers for B2M (human) and Rpp30 (mouse) genes were used.
Post-mortem human retina cultures
Post-mortem human retinas were maintained as described previously27. The tissue used in our experiments was obtained from one donor. The periphery of the donor retina was cut into pieces with fine surgical scissors and cultured individually in a Transwell with a polycarbonate membrane of 0.4 µm pore size (Corning, 7910). The medium was changed every second day. The retina pieces were cultured for up to 6 weeks. For live imaging, cultured retina pieces were transferred to a transparent Millicell cell culture insert (Merck, PICM0RG50).
Blood sample collections from healthy donors for PBMC isolation
Blood samples were obtained as buffy coats from healthy male and female donors after written informed consent (Blood Donor Center, University Hospital Basel).
Isolation and activation of human CD8+ T cells
PBMCs were isolated by density-gradient centrifugation using Lymphoprep (Serumwerk Bernburg, 1858). CD8+ T cells were enriched using CD8+ magnetic beads (Miltenyi Biotec, 130-045-201) by positive selection. CD8+ T cells were activated using soluble antibody complexes containing anti-CD3, anti-CD28 and anti-CD2 antibodies (Immunocult, StemCell Technologies, 10970) for 2 days.
Generation of constructs for cell-surface binder or antigen display
The sequence for anti-GFP nanobody was retrieved from pcDNA3_NSlmb-vhhGFP4 plasmid (Addgene, 35579), and the sequence for LaM-2 anti-mCherry nanobody was obtained from pET21-pelB-LaM-2 plasmid (Addgene, 172768). To generate the pAAV-EF1α-IgK-VHH_GFP4-OLLAS-TMD-WPRE and pAAV-EF1α-IgK-VHH_LaM2-OLLAS-TMD-WPRE constructs for cell-surface anti-GFP and anti-mCherry nanobody display, respectively, nanobody sequences codon optimized for mammalian expression were fused to the IgK signal peptide at the N terminus and an OLLAS-tag followed by PDGFRβ transmembrane domain at the C terminus. The sequences were chemically synthesized (Twist Bioscience) with overhangs and subcloned into the pAAV backbone containing the EF1a promoter via HiFi DNA assembly (NEB, E2621X), where the GFP transgene was removed using BamHI/EcoRI restriction sites (NEB). Similarly, cell-surface mCherry and enhanced GFP (eGFP) constructs (pAAV-EF1α-IgK-mCherry-OLLAS-TMD-WPRE and pAAV-EF1α-IgK-eGFP-HA-TMD-WPRE, respectively) were generated as described above. Sequences encoding either mCherry or eGFP with an N-terminal IgK and C-terminal OLLAS-tag (for mCherry) or an HA tag (for eGFP) and a PDGFRβ transmembrane domain were chemically synthesized (Twist Bioscience) and subcloned into the pAAV-EF1α-GFP backbone. To generate the pAAV-ProA5-DIO-IgK-VHH_GFP4-OLLAS-TMD-WPRE and pAAV-ProA5-DIO-IgK-LaM2_VHH-OLLAS-TMD-WPRE constructs, synthesized inserts (described above) were inverted by PCR (forward primer: 5′-TACATTATACGAAGTTATGGCGCGCCCTAACGTGGCTTCTTCTG-3′, reverse primer: 5′-ATACTTTATACGAAGTTATGCTAGCCACCATGGAGACAGACAC-3′) using Q5 DNA polymerase (NEB, M0492S) and subcloned into the pAAV-ProA5-DIO-DsRed2 backbone (Roska laboratory plasmid collection) through HiFi DNA assembly, whereby the DsRed2 transgene was removed using AscI/NheI restriction sites (NEB).
Generation of constructs for mitochondrial surface binders or antigen display
For mitochondrial outer membrane GFP targeting, the pCMV-GFP-OMP25 plasmid was used (Addgene, 141150). To display proteins of interest (for example, nanobodies, DARPins or SNAP-tag) on the surface of mitochondria, sequences were chemically synthesized (Twist Biosciences), fused to the C terminus of rat OMP25 and subcloned into the pcDNA3.1(+) backbone under control of the CMV by HiFi DNA assembly. Anti-GFP and anti-mCherry nanobody DNA sequences were obtained as described above. Nanobody sequences for cell-type-specific surface markers including human and mouse CD71, human CD73 and human CD142 were developed in this study and are described below. Anti-GFP (3G86.32) and anti-mCherry (2m22) DARPin sequences were retrieved from a published study36, the SNAP-tag sequence was obtained from pSNAP-tag (m) vector (Addgene, 101135).
Generation of constructs for ubiquitous intracellular expression of destabilized nanobody
To generate the destabilized nanobody construct in an AAV backbone (pAAV-EF1α-dGBP1-TagBFP-WPRE), dGBP1-TagBFP from pCAG-dGBP1-TagBFP (Addgene, 80086) was PCR amplified via Q5 DNA polymerase (forward primer: 5′-TCAGGTGTCGTGAGGTACCGGATCCGCCACCATGGCCGACGTG-3′, reverse primer: 5′-TATCGATAAGCTTGATATCGAATTCTTAATTAAGCTTGTGCCCCAGTTTGCTAG-3′; NEB, M0492) and subcloned into an AAV backbone carrying the EF1A promoter through HiFi DNA assembly.
Generation of constructs for recombinant binder production in E. coli
Genes encoding nanobodies developed in this study, and bispecific binders were synthesized by Twist Biosciences. Nanobodies were inserted into a pET28-derived vector with a modified N-terminal pelB signal peptide59 (pET28-NPelB(DN5v3)-6×His) between BamHI and XhoI sites through HiFi DNA assembly. The anti-mCherry–(G3S)2G–anti-GFP bispecific nanobody construct was inserted into a pET24 vector (pET24-NPelB-6×His). Bispecific nanobodies produced from Escherichia coli were used for mitochondrial targeting experiments in in vitro assays. For in vivo experiments, the mammalian expression system was used as described below.
Generation of CD71, CD73, CD142 and bispecific nanobody constructs for the mammalian expression system
Open reading frames of human CD71 (NCBI: NM_001128148.3), CD73 (NCBI: NM_002526.4) and CD142 (NCBI: NM_001993.5) genes were synthesized (GenScript Biotech) and subcloned into a pcDNA3.1(+)-P2A-eGFP backbone containing a CMV promoter by using HindIII/XbaI restriction sites. Bispecific nanobodies were fused through a (G3S)2G linker. The anti-mCherry–(G3S)2G–anti-GFP bispecific nanobody sequence was codon optimized and synthesized (Twist Biosciences) with an N-terminal heavy chain signal peptide (IgHC) and a C-terminal 6×His tag. The DNA fragment was subcloned into a pcDNA3.1(+) backbone containing a CMV promoter using HindIII/EcoRI restriction sites. The anti-lysozyme–(G3S)2G–anti-GFP bispecific nanobody construct was synthesized with an N-terminal mouse Igκ signal peptide and cloned into the pCSG_IBaA103 vector (IBA Life Sciences) containing a CMV promoter using BamHI/EcoRI restriction sites. The sequence for the anti-lysozyme nanobody was obtained from a published study60.
Constructs for labelling cellular organelles
Constructs to target MiniSOG (pCMV-MiniSOG-Mito-7) and DsRed2 (pCMV-DsRed2-Mito-7) into the mitochondrial matrix were obtained from Addgene (Addgene, 57773 and 55838, respectively). For TagBFP2 targeting into the matrix, the mTagBFP2 coding DNA sequence was fused to COX8 matrix-targeting signal peptide, synthesized by Twist Biosciences, and inserted into a pCMV backbone. For endoplasmic reticulum labelling, TagBFP was fused to a KDELR1 peptide sequence and inserted it into an AAV backbone containing Ef1a promoter and WPRE sequence. For mito-dsRed2 expression in iHNeurons and HUVECs, the dsRed2 sequence was inserted into the same AAV backbone. The generation of AAVs is described below.
Plasmid DNA transfection
For mitochondria labelling with outer-membrane-targeted GFP, matrix-targeted dsRed2 and TagBFP, nanobodies, DARPins or SNAP-tag, HEK293T cells were grown in a 150 mm culture dish and transfected with 20 µg of plasmid DNA (a list of the constructs is provided in Supplementary Table 4) at 70–80% confluency using the jetPrime reagent (Polyplus transfection, 101000046) according to the manufacturer’s instructions. Mitochondria were isolated 24 h after transfection. mMSCs were used for the isolation of mouse-derived mito-GFP. For this, the cells were grown in a 225 cm2 culture flask (Thermo Fisher Scientific, 159934) and transfected with 40 µg of plasmid DNA. The culture medium was exchanged after 24 h and mitochondria were isolated 48 h after transfection.
For cell-surface labelling with GFP, mCherry and nanobodies or for overexpression of CD markers (CD71, CD142 and CD73), HEK293T cells were seeded on a 96-well plate (Ibidi, 89626-90) at 30,000 cells per cm2 density and transfected at 40–50% confluency as described above.
AAV production
AAVs were produced as described previously61. In brief, adherent HEK293T cells were co-transfected with the AAV transgene plasmid, an AAV helper plasmid encoding the AAV Rep2 and Cap proteins specific to the chosen serotypes, and the pHGT1-Adeno1 helper plasmid (provided by C. Cepko) using branched polyethyleneimine (PEImax, Polysciences). AAVs were collected from cells and cell culture supernatant 72 h after transfection and isolated using a discontinuous iodixanol density gradient prepared with OptiPrep (Sigma-Aldrich, D1556) and ultracentrifugation. The AAVs underwent concentration and buffer exchange into PBS containing 0.001% Pluronic F 68 using Amicon Ultra-15 100K spin filters (Millipore, UFC9100). Genomic copies were titrated using quantitative PCR for ITR and ddPCR for WPRE, with the following primer sequences: ITR forward: 5′-GGAACCCCTAGTGATGGAGTT-3′, ITR reverse: 5′-CGGCCTCAGTGAGCGA-3′, ITR probe: 5′-(6FAM)CACTCCCTCTCTGCGCGCTCG(MGB)-3′; WPRE forward: 5′-GGCTGTTGGGCACTGACAA-3′, WPRE reverse: 5′-CCAAGGAAAGGACGATGATTTC-3′, WPRE probe: 5′(6FAM)TCCGTGGTGTTGTCG(MGB)-3′. The WPRE ddPCR value was used for AAV titre matching of vector preparations for in vivo experiments. The validation of identity and purity of input plasmids was confirmed by complete plasmid next-generation sequencing (NGS) at MGH CCIB DNA Core.
AAV transduction of iHNeurons, primary endothelial cells, post-mortem human retinas and mouse retinas
For iHNeurons and primary endothelial cells cultured in an eight-well slide (Ibidi, 80826-IBI), AAVs were diluted to a titre of 1 × 1011 genome copies per ml. The existing medium in the well was replaced with 50 µl of culture medium containing diluted AAVs and incubated for 1 h. An additional 150 µl of culture medium was then added on top of the well. The medium was exchanged after 48 h and cells were fed every second day. For iHNeurons in a 35-mm MatTek gridded coverslips dish, AAVs were diluted to a titre of 2.5 × 1010 genome copies per ml. The existing medium in the well was replaced with 200 µl of culture medium containing diluted AAVs to cover the gridded coverslip area. After 1 h, an additional 1,800 µl of medium was applied on top. The medium was exchanged after 48 h and cells were fed every second day. The cells were cultured for a further 3 weeks before the experiments were conducted.
For post-mortem human retinas, AAVs were diluted to a titre of 3.8 × 1012 genome copies per ml. For each explant, 30 µl of culture medium containing diluted AAVs was applied topically covering the whole explant. The explant tissues were then kept at room temperature for 10 min before transfer to an incubator. The medium was exchanged after 48 h and the explants fed every second day. The infected explants were cultured for at least 5 weeks before the experiments were conducted.
For in vivo transduction in mouse retinas, 1 × 1011–1012 genome copies were delivered bilaterally by intravitreal injection using a nanoinjector (Nanoliter 2020, WPI) and 1 × 1011 genome copies per gram were injected retro-orbitally. Treated mice were given carprofen (50 mg ml−1, Rimadyl) 24 h before the surgery for up to 48 h. The injected eye was post-treated with Novesin 0.4% (OmniVision). Mice were used for the experiments after 3 weeks.
Western blotting
Mitochondria were freshly isolated and 5 µg of mitochondria was mixed with loading buffer (Thermo Fisher Scientific, NP0007) and reducing agent (Thermo Fisher Scientific, NP0004) and heated for 10 min at 90 °C. A total of 2.5 µg of each sample was loaded onto SDS–PAGE gel (4–12% acrylamide gel, Thermo Fisher Scientific, NP0322) and run in MOPS running buffer (Thermo Fisher Scientific, NP0001). The gel was moved onto a PVDF membrane (Millipore, IPVH00010) in Tris-glycine transfer buffer with 20% methanol (Thermo Fisher Scientific, LC3675). The transfer was done at 20 V for 2 h at 4 °C. The membrane was then blocked in 5% milk (Roth, T145.3) in TBS-T buffer (TBS + 0.1% Tween-20) at room temperature for 1 h and incubated overnight at 4 °C in primary antibodies in TBS-T containing 2.5% milk. The membrane was washed three times with TBS-T and incubated with secondary antibodies for 1 h at room temperature in 2.5% milk in TBS-T. After washing with TBS-T, the membrane was developed using Pierce ECL western blotting substrate (Thermo Fisher Scientific, 32106). Primary antibodies were as follows: anti-SNAP-tag (1:1,000, NEB, P9310S), anti-TOMM20 (1:2,500, Sigma-Aldrich, HPA011562), MCU (1:1,000, AMAB91189), GRP78 (1:2,000, G8918), PDIA3 (1:500, AMAB90988), KDELR1 (1:1,000, NBP2-12873) and membrane-integrity antibody cocktail (1:232, Abcam, ab110414). Secondary antibodies were as follows: anti-rabbit IgG-HRP antibodies (1:1,000, Jackson, 711-036-152), anti-mouse IgG-HRP antibodies (Thermo Fisher Scientific, A16017). All original membrane images are shown in Supplementary Fig. 1.
Expression and purification of TOMM20 cytoplasmic domain
The gene encoding the cytoplasmic domain of TOMM20 (UniProt: Q15388, residues 59–145, IDT) was cloned into a pET28-derived vector behind an N-terminal 6×His tag, SUMO domain and 3C-protease cleavage site and transformed to Shuffle T7 express cells (NEB, C3029J). A single, sequence verified clone was grown in LB with 50 μg ml−1 kanamycin overnight. The next day, 100 ml TB auto induction medium (Formedium, AIMTB0210) with 50 μg ml−1 kanamycin was inoculated with 1 ml of the overnight culture, grown at 37 °C for 2.5 h, and overnight at 26 °C. Cells were pelleted (15 min, 8,000g), lysed with 1× BugBuster extraction buffer (Millipore, 70584-3), protease inhibitor tablets (Thermo Fisher Scientific, A32955) in 50 mM Tris pH 8 and 100 mM NaCl for 30 min at room temperature. Cell debris was removed by centrifugation for 15 min at 15,000g. The supernatant was loaded onto a Streptactin XT column (IBA Lifesciences, 2-5028-001), washed with 5 column volumes (CV) of wash buffer (IBA Lifesciences, 2-1003-100) and eluted with 10 CV elution buffer (IBA Lifesciences, 2-1042-025). The elution fractions were pooled and concentrated to 2 ml and incubated overnight at 4 °C with homemade produced 3C protease (Addgene, 162795) added at 2% of total TOMM20 weight. The mixture was diluted to 25 ml with 20 mM Tris HCl pH 8.0 and incubated with 1 ml PureCube Ni-INDIGO MagBeads (CUBE Biotech, 75225) for 2 h at 4 °C to remove cleaved SUMO domain. The supernatant was loaded onto a Capto HiRes Q 5/50 column and eluted with a gradient to 35% 20 mM Tris HCl pH 8.0, 500 mM NaCl. Fractions containing pure TOMM20 were identified by SDS–PAGE, pooled and frozen at –80 °C until further use.
Expression and purification of TOMM70 cytoplasmic domain
A gene fragment of the cytoplasmic domain of human TOMM70 (amino acids 111–608) was synthesized (Twist Bioscience) and subcloned into pET28 vector containing an N-terminal 7×His-tag followed by a TEV protease cleavage site. The construct was transformed into the Rosetta 2 T7 express E. coli strain for protein expression and purification. The protein was produced as previously described62, with some modifications. The genes were expressed at 17 °C in E. coli T7 Express cells (NEB) co-transformed with the pRARE2 plasmid. The purification protocol comprised mechanical cell lysis by sonication (SONOPULS HD 2200, Bandelin), affinity chromatography on the 5 ml HisTrap FF crude column (Cytiva), 7×His-tag cleavage with 1:30 (w/w) tobacco etch virus protease while dialysing into 20 mM HEPES-NaOH pH 7.5, 0.25 M NaCl, 5% glycerol and 1 mM DTT, a 5 ml HiTrap Heparin HP column (Cytiva) equilibrated with 20 mM HEPES-NaOH pH 7.5 and 0.15 M NaCl, and size-exclusion chromatography on the 26/600 Superdex 200 prep grade gel-filtration column (Cytiva) equilibrated with PBS buffer pH 7.4. The purified protein was concentrated to 4 mg ml−1, sterile filtered, flash-frozen in small aliquots with liquid nitrogen and stored at −70 °C until further use. The intact molecular mass was confirmed by liquid chromatography coupled with time-of-flight mass spectrometry (LC–TOF-MS).
Expression and purification of fluorescent proteins
Gene fragments were obtained from IDT or Twist Biosciences and subcloned by Gibson assembly (NEB HiFi DNA assembly master mix). Fluorescent proteins were subcloned into modified pET28 vectors with either N-terminal 6×His tag, HRV 3C protease cleavage site and avi-tag or C-terminal MYC tag and 6×His tag. The plasmids were transformed into BL21 (DE3) cells, and the avi-tagged constructs were co-transfected with the pBirAcm plasmid (Avidity).
Starter cultures were grown in LB medium and 25 μg ml−1 kanamycin. Expression was performed in autoinduction TB medium with 25 μg ml−1 kanamycin (Formedium, AIMTB0260) by inoculation with 1/100 volume of starter culture incubation at 37 °C for 2–3 h followed by 25 °C overnight. Cells were collected by centrifugation at 5,000g for 15 min, resuspended in PBS and lysed by sonication. Cell debris was removed by centrifugation. Avi-tagged fluorescent proteins was removed with NT*-HRV3CP (Addgene, 162795) by incubation with 2 wt% of protease for 2 h at room temperature. Uncleaved proteins and protease were removed using PureCube Ni-INDIGO MagBeads.
TOMM70 biotinylation
TOMM70 (4 mg ml−1) in PBS was incubated with a fivefold molar excess of EZ-Link NHS-PEG4-Biotin (Thermo Fisher Scientific, A39259) for about 1 h at room temperature. Unreacted reagent was blocked by adding 100 mM Tris/HCl pH 7.4 and removed with PBS equilibrated Zeba Spin Desalting columns (Thermo Fisher Scientific, 89882). The degree of biotinylation was determined using the QuantTag Biotin Quantitation Kit (Vector Labs, BDK-2000) according to the manufacturer’s instructions, and was determined to be 4.4 biotins per TOMM70 molecule.
Camelid-derived nanobody generation and selection
The general workflow for nanobody generation, production and selection was based on published protocol63. Nanobodies for human CD73 and mouse CD71 were generated at VIB Nanobody Core (VUB). Commercially available recombinant human CD73 (Sino Biological, 10904-H08H) and mouse CD71 (Sino Biological, 50741-M07H) proteins were used for llama immunizations. Llamas were immunized up to six times once a week with 120 µg of recombinant protein per each immunization. VHH DNA libraries were constructed from the animal’s peripheral lymphocytes cDNA, subcloned into the pMECS-GG phagemid vector64 and transformed into E. coli TG1 cells for subsequent nanobody selection through phage-display. Phages from the constructed VHH DNA libraries were panned on solid phase coated with target protein (100 µg ml−1 in 0.1 M NaHCO3 pH 8.2) for three rounds. Periplasmic extracts from the enriched clones after the second and third selection rounds were analysed by enzyme-linked immunosorbent assay (ELISA). Validated clones were sequenced by Sanger sequencing using the MP057 primer (5′-TTATGCTTCCGGCTCGTATG-3′).
Nanobodies for human CD71 were produced at the Nanobody Service Facility (University of Zurich). Commercially available recombinant human CD71 (Sino Biological, 11020-H07H) was used for alpaca immunization. The llama was immunized four times every second week with 200 μg of recombinant protein per injection. Similarly, cDNA was synthesized from RNA isolated from peripheral lymphocytes. VHH DNA libraries were constructed by PCR and subcloned into the pDX phagemid vector34. The library was transformed into E. coli TG1 cells for subsequent nanobody selection through phage-display. Phages from the constructed VHH DNA libraries were panned on solid phase coated with target protein (10 µg ml−1 in 0.1 M NaHCO3 pH 8.4) for three rounds. Periplasmic extracts from the enriched clones after the second and third selection rounds were analysed using ELISA. Validated clones were sequenced by Sanger sequencing using SQ-NbREV primer (5′-GTAGCATTCCACAGA CAGCCCTCATAGT-3′).
Synthetic nanobody library design
Nanobody libraries were designed with two different CDR3 lengths (long and short CDR3 libraries). The framework regions were partially humanized using human germline V3-23*J04 as a template, hallmark camelid residues in framework 2 were kept (long CDR3 library) or partially kept (short CDR3 library) similar to the framework designs reported previously65,66. The design of the CDR3 regions and the randomization strategy of all complementarity-determining regions (CDRs) were inspired by the strategy used previously67. Further mutations beneficial for nanobody stability were incorporated into the library68. The final sequences of the libraries are shown in Extended Data Fig. 10. Libraries flanked by regulatory regions necessary for ribosome display were ordered from Twist Biosciences.
Nanobody selections against TOMM70 and TOMM20 using mRNA ribosome display
The nanobody selections followed the published protocol69, with a few modifications: chemically biotinylated TOMM70 and TOMM20 with a TwinStrep tag were used as target proteins. The panning procedures were automated on a KingFisher DUO instrument (Thermo Fisher Scientific, 5400110) using Streptavidin-coated magnetic beads for TOMM70 (Thermo Fisher Scientific, 65001 and 65601) or Streptactin-XT-coated beads for TOMM20 (IBA Lifesciences, 2-5090-002). All nanobody sequences generated in this study are shown in Supplementary Data 1 and their antigen binding kinetics are described in Supplementary Table 3.
Prokaryotic expression and purification of camelid-derived nanobodies
Newly generated and affinity matured nanobodies were produced in E. coli Shuffle T7 express strain (NEB, C3029J). Starter cultures were grown in LB medium with 1% glucose and 25 μg ml−1 kanamycin. Expression was performed in autoinduction TB medium with 25 μg ml−1 kanamycin (Formedium, AIMTB0260) by inoculation with 1/100 volume of starter culture incubation at 37 °C for 2–3 h followed by 25 °C overnight incubation.
Prokaryotic expression of nanobodies from synthetic libraries and bispecific nanobodies
Gene fragments were obtained from IDT, Twist Biosciences and Genscript, or were generated by PCR (Q5 High-Fidelity 2× Master Mix, NEB, M0492L). Nanobodies from synthetic libraries were cloned by Gibson assembly (NEB HiFi DNA assembly master mix) into a pET28-derived vector without a PelB signal sequence and with a C-terminal 6×His tag. Expression was carried out from a single, sequence-verified colony of Shuffle T7 express cells (NEB, C3029J). TB Autoinduction medium (Formedium, AIMTB0210) was inoculated with overnight culture grown in LB (100:1 ratio), grown for 4 h at 30 °C, and overnight at 25 °C. Pelleted cells were lysed in 5 ml detergent extraction buffer per 50 ml expression culture consisting of 50 mM Tris HCl pH 8.0, 500 mM NaCl, 1% CHAPS, 1% NP40, 10 mM imidazole and protease inhibitors (Thermo Fisher Scientific, A32955) and incubated for 30 min. Cell debris was removed by centrifugation. Proteins were purified from the supernatant with PureCube Ni-INDIGO MagBeads (CUBE Biotech, 75225) according to the manufacturer’s instructions. Elution fractions were pooled and polished by size-exclusion chromatography with PBS as the running buffer (Sepax, SRT-10C SEC300 column). Monomeric fractions were collected, pooled and concentrated if necessary.
Bispecific nanobodies were subcloned into pET28-derived vectors with or without a modified PelB signal peptide (DN5v3.1) and transformed into E. coli Shuffle T7 express strain (NEB, C3029J) as described above. 2YT medium with 0.5% glucose and 25 μg ml−1 kanamycin was inoculated with 1/100 of starter culture grown in LB with 1% glucose and 25 μg ml−1 kanamycin. Cultures were grown at 37 °C until they reached an optical density at 600 nm of 0.7, induced with 1 mM IPTG and grown overnight at 25 °C. Constructs without PelB signal peptide were purified as described above. Cells carrying constructs with PelB signal peptides were resuspended in periplasmic extraction buffer (3–5 ml per 50 ml expression culture, 20% sucrose 50 mM Tris/HCl pH 8.0, 0.5 mM EDTA and 0.5 μg ml−1 lysozyme) and incubated for 30 min on ice. Four times the volume of 20 mM Tris/HCl pH 7.5, 150 mM NaCl and 2 mM MgCl2 was added. Cells were separated by centrifugation (5,000g, 15 min) and the supernatant was decanted. Purification was continued as described above.
Mammalian expression and purification of bispecific nanobodies
For in vivo experiments to reduce endotoxin contamination, the bispecific nanobodies were produced in ExpiCHO-S cells (Thermo Fisher Scientific, A29127). For the anti-mCherry–(G3S)2G–anti-GFP bispecific nanobody production (produced at the EPFL Protein Production Platform), the cells were cultured in ProCHO5 medium (Lonza, BELN12-766Q) supplemented with 2% DMSO. The plasmid DNA encoding the anti-mCherry–(G3S)2G–anti-GFP bispecific nanobody was transfected with PEI-MAX (Polysciences, POL24765-1). After 6 days, the supernatant was filtered and incubated with Fastback Ni Advance Resin (Protein Ark, Fastback-Ni-Adv) overnight at 4 °C. The resin was loaded into a disposable column and washed with 500 mM NaCl, 50 mM HEPES pH 7.5 and the nanobody was eluted with 500 mM NaCl, 50 mM HEPES pH 7.5, 500 mM imidazole. Eluted protein was dialysed overnight in PBS at 4 °C and concentrated using an Amicon 30 kDa cut-off column (Merck, UFC9030). For the anti-lysozyme–(G3S)2G–anti-GFP bispecific nanobody production (produced in-house), the cells were cultured in ExpiCHO Expression Medium (Thermo Fisher Scientific, A29100). The plasmid was transfected with Expifectamine CHO reagent (Thermo Fisher Scientific, A29129). The protein from the supernatant was purified using Ni-IMAC (column: HisTrap excel; Cytiva, 17371206). The column was washed with 500 mM NaCl, 50 mM Tris pH 8.0 and 20 mM imidazole, and the nanobody was eluted with 500 mM NaCl, 50 mM Tris pH 8.0 and 500 mM imidazole. Pure protein fractions were subsequently purified by anion-exchange chromatography and then size-exclusion chromatography for monomeric protein fractions. The pure protein fractions were pooled and concentrated in PBS.
Nanobody affinity maturations
Site saturation libraries targeting all three CDRs and adjacent sequences of initial VHH hits were synthesized by Twist Biosciences. Libraries were subcloned into the pDX plasmid and electroporated into TG1 DUOs electrocompetent cells (Biosearch Technologies, 60502-2). Transformed cells were superinfected with M13K07 helper phage (NEB, N0315S) and phagemids were purified from culture supernatant. Phagemids were cycled through 3−4 rounds of phage display with increasing stringency. Mutations that were enriched over sequential rounds were identified by NGS (Illumina MiSeq) of PCR amplified VHH inserts from phages. Data analysis was performed using the PipeBio platform. Genes of clones with single or combined enriched mutations were synthesized (eblocks, IDT) and expressed as described above.
Nanobody affinity measurement using surface plasmon resonance
All measurements were conducted on a Biacore T200 instrument using PBS with 0.05% Tween-20 as running buffer. CM5 chip (Cytiva, BR100399) surfaces were functionalized using an amine coupling kit (Cytiva, BR100050) according to the manufacturer’s instructions with a goat anti-human IgG Fc antibody (Southern Biotech, 2048-01) or a goat anti-mouse IgG2a Fc antibody (Southern Biotech, 1081-01) to immobilize Fc-tagged target proteins. Recombinant human CD71 (110202-H01H) and human CD142 (13133-H02H) proteins were obtained from Sino biologics. Recombinant mouse CD71 (TFR-M5263) and human CD73 (CD3-H5252) proteins were obtained from Acro Biosystems. To immobilize biotinylated proteins, we used the RGD200M sensor chip (Xantec) in combination with oligonucleotide-modified Streptavidin reagent (Xantec, C RG-SA-50I) according to the manufacturer’s instructions. To immobilize TwinStrep-tagged proteins, we modified a CM5 chip surface with Streptactin XT using the Twin-Strep-tag Capture Kit (IBA Lifesciences, 2-4370-000) according to the manufacturer’s instructions. Increasing concentrations of VHHs were injected with a single cycle protocol. Regeneration of measurement surfaces was achieved with 200 mM H3PO4 for anti-Fc surfaces or regeneration solutions according to the manufacturer’s instructions for other capture reagents. Data were double referenced and fitted to a 1:1 binding model in the instrument software.
Thermostability measurement of selected nanobodies
Nanobodies were diluted to 0.3–0.5 mg ml−1 in PBS. Data were collected from 20–95 °C on the Prometheus nanoDSF instrument (Nanotemper) in a standard capillary. Fitting was performed in the instrument software.
BG conjugation to monoclonal antibodies
Anti-CD31 monoclonal antibodies (130-108-038) and mouse IgG1 isotype control (130-106-545) were obtained from Miltenyi Biotec. BG conjugation to antibodies was as described previously20. In brief, BG-GLA-NHS (NEB, S9151S) were incubated with the antibodies (1 mg ml−1) at a 60-fold molar excess for 30 min at room temperature. The antibodies were column purified using Zeba Spin Columns (7 K MWCO; Thermo Fisher Scientific, 89882) and eluted in 1× PBS, pH 7.4.
BG-conjugated antibody linking to mitochondria through the SNAP-tag
BG-conjugated antibodies at different concentrations (0–500 nM) were incubated with 2.5 µg of SNAP-tag-displaying freshly isolated mitochondria at room temperature for 20 min. The mitochondria were then transferred to ice and incubated for an additional 10 min before transplantation.
AlphaFold2 and AlphaFold3 predictions
Structure predictions were done with AlphaFold2 implemented in a google colab notebook70 available online (https://colab.research.google.com/github/sokrypton/ColabFold/blob/main/AlphaFold2.ipynb.Predictions); predictions using AlphaFold371 were done at https://alphafoldserver.com/.
Mitochondria isolation
Mitochondria were freshly isolated from donor cells using the Qproteome Mitochondria Isolation Kit (Qiagen, 37612) according to the manufacturer’s instructions. The mitochondria sources were HEK293T cells for targeting human cells (for example, HEK293T, iHNeurons, human retina) and mMSCs for targeting mouse cells (mouse retina). The amounts of freshly isolated mitochondria used for the transplantation experiments corresponded to the total protein amount in the isolated mitochondrial fractions, measured using the Qubit Protein Assay Kit (Thermo Fisher Scientific, Q33212).
Mitochondrial transplantation
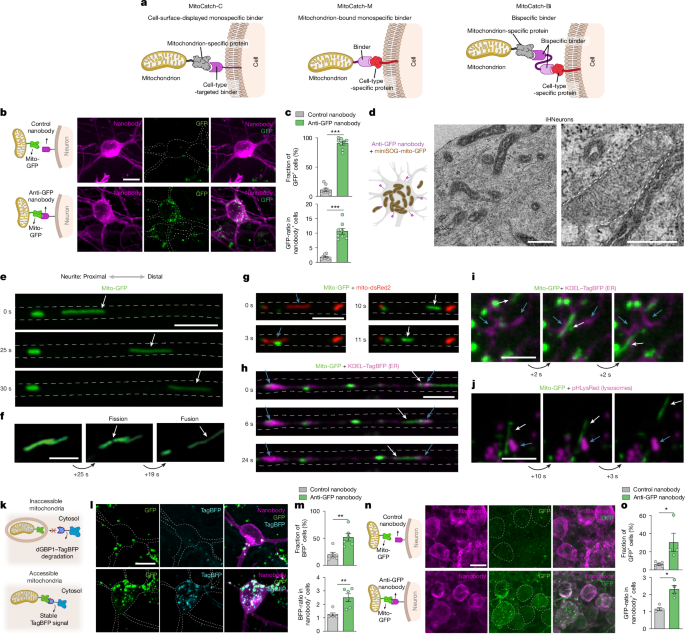
Freshly isolated mitochondria were transplanted within 1 h of isolation in the storage buffer provided in the kit. For proliferating cells in 2D culture, we used a 2 h incubation for quantification of mitochondrial delivery efficiency, avoiding any possible dilution of mitochondria due to cell division. For cytosolic accessibility, we incubated cells for 1 day to allow enrichment of BFP at the GFP-labelled mitochondria. For post-mitotic cells such as neurons, organoids and retinal ganglion cells, we waited for 1 day to allow the distribution of donor mitochondria along the processes of the cells. The amounts of mitochondria used were as follows: for transplantation targeting experiments in cultured cells, 0.5–10 µg; for mitochondrion-surface display experiments in HEK293T cells with affinity matured nanobodies, 0.5–2.5 µg; for transplantation in LHON iHNeurons respiration assays, 2.5–10 µg; for transplantation in glycolysis inhibition experiments involving LHON iHNeurons, 5 µg; and, for retinal and blood vessel organoids, 10 µg. For isolated suspensions of CD4 and CD8+ T cells, the amount of mitochondria was normalized to 2.5 µg per 100,000 cells. For ex vivo cultured post-mortem human retinal explants, 3 µg of mitochondria was injected per piece. For in vivo targeted mitochondrion-delivery experiments, 2.5 µg of mitochondria was injected, while for cell rescue experiments the amount injected intravitreally was 0.5–5 µg. For MitoCatch experiments involving anti-mCherry–(G3S)2G–anti-GFP bispecific binders, 2.5 µg of isolated mitochondria was co-incubated with bispecific binders (4 µM) for 25 min on ice before they were added to cells or injected in vivo. MitoCatch experiments involving anti-TOMM20–(G3S)2G–anti-CD4 bispecific binders, 0.5–5 µg of isolated mitochondria was co-incubated with bispecific binders (0.5 µM) for 1 h on ice before the mitochondria were added to cells. For MitoCatch experiments involving a SNAP-tag, 2.5 µg of isolated mitochondria was incubated with either 0.5 µM anti-CD31 or 0.5 µM IgG1 and transplanted into endothelial cells; 10 µg of mitochondria was incubated with 0.5 µM anti-CD31 or 0.5 µM IgG1 and transplanted into individual blood vessel organoids. For unstimulated CD8+ T cells, 5 µg of mitochondria was incubated with either 1 µM CD8 or 1 µM IgG1 and transplanted into CD8+ T cells (2.5 µg per 100,000 cells).
For in vitro transplantations, HEK293T cells, cardiac cells or endothelial cells were cultured in 96-well plates (Ibidi, 89626). iHNeurons, were cultured in MatTek gridded coverslips dishes (MatTek, P35G-1.5-14-C-GRD) or in 8-well slides (Ibidi, 80826-IBI). For retinal and vascular organoids or T cells, 96-well cell culture plates with a conical bottom (Thermo Fisher Scientific, 277143) were used.
For ex vivo transplantation into post-mortem human retinas, mitochondria were injected with a nanoinjector (Nanoliter 2020, WPI); five injections per explant were performed in a volume of 150 nl (0.6 μg). For in vivo transplantation into eyes, mitochondria were injected intravitreally with a Hamilton syringe (5 µl, 32 gauge, Hamilton; 87931) in a volume of 1.5 µl. Treated mice were given carprofen (50 mg ml−1, Rimadyl) 24 h before the surgery for up to 48 h, and the injected eye post-treated with Novesin 0.4% (OmniVision).
In targeted mitochondrial delivery experiments in HEK293T, cardiac cells and endothelial cells, the cells were washed 2 h after transplantation three times with PBS and fixed with 4% PFA for further processing. T cells were washed and stained for flow cytometry after 4 h, vascular organoids were fixed after 6 h, and iHNeurons, retinal organoids, mouse retina and post-mortem human retinal explants were washed with 1× PBS and fixed the day after mitochondrial transplantation.
Flow cytometry
Mitochondrial targeting efficiency in T cells was assessed by flow cytometry. After incubation with donor mitochondria, T cells were transferred to a U-bottom 96-well plate (Thermo Fisher Scientific, 163320) for antibody staining; at each step, cells were maintained on ice and stained with antibodies for 30 min. Cells were washed before and after staining with staining buffer (1% FBS in PBS). For mitochondrion-targeting experiments involving the anti-CD71 nanobody, both unstimulated and activated CD8+ T cells were stained with monoclonal mouse anti-CD71-FITC antibodies (1:100, BioLegend, 334104) to identify the CD71+ population. For CD4+ T cell targeting experiments, anti-CD4-FITC antibodies (1:100, BioLegend, 344604) were used to identify the CD4+ population. As the transplanted mitochondria exhibited DsRed2 fluorescence, no additional staining was necessary.
In mitochondrial targeting experiments involving SNAP-tag strategy to target CD8 receptor in unstimulated CD8+ T cells, cells were stained initially with polyclonal rabbit anti-SNAP antibodies (1:100, NEB, P9310S), followed by washing and incubation with donkey anti-rabbit IgG (H+L) secondary antibody conjugated to Alexa Fluor 568 (Thermo Fisher Scientific, A10042). Data were acquired using the Cytoflex flow cytometer (Beckman Coulter) and analysed in FlowJo.
Respiration measurements of isolated mitochondria
Respiration analysis of freshly isolated mitochondria was performed using the Seahorse XF96 extracellular flux analyser (Seahorse Bioscience, Agilent). In total, 5 µg of mitochondria was resuspended in 25 µl of storage buffer containing 2 µM rotenone, 10 mM succinate and 10 mM ADP. Mitochondria were added into a prechilled 96-well plate and centrifuged for 20 min at 2,000g at 4 °C. The wells were then supplemented with 155 µl of pre-warmed storage buffer containing the substrates. For the control condition, succinate and ADP were not added. To stop mitochondrial respiration 4 µM antimycin A was injected.
Respiration measurements of LHON iHNeurons
Patient-derived LHON iHNeurons were induced from iPS cells as described previously. CD90+ sorted cells were seeded on a 96-well Seahorse well plate (Agilent, 103793-100) coated with Matrigel at 40,000–120,000 cells per well (ranged from induction batch). Seeded cells were allowed to recover for a week and then infected with a PHP.eB72 serotyped AAV encoding cell-surface anti-GFP nanobody (1 × 1011 genomic copies). The cells were incubated for 2 weeks before adding mitochondria and the oxygen consumption rate of the cells was measured 2 days after transplantation.
Respiration measurements were performed using the Seahorse XF96 extracellular flux analyzer (Seahorse Bioscience, Agilent). The day before the assay, the sensor cartridge was hydrated overnight in a calibrant in a CO2-free incubator at 37 °C. On the day of the assay, the cells were washed three times in unbuffered DMEM supplemented with glucose, pyruvate and glutamine (Agilent, 103680-100) and allowed to equilibrate for at least 30 min in a CO2-free incubator at 37 °C. During the assay, metabolic modulators—1 µM oligomycin (Sigma-Aldrich, 75351), 2 µM carbonyl cyanide 4-(trifluoromethoxy) phenylhydrazone (FCCP, Sigma-Aldrich, C2920), 1 µM rotenone (Sigma-Aldrich, R8875) and 1 µM antimycin A (Sigma-Aldrich, A8674)—were added sequentially and the oxygen consumption rates were measured in real time. Oligomycin, an ATP synthase inhibitor, was used to assess ATP-linked respiration. FCCP, an uncoupling compound, was used to assess maximal respiration. Rotenone and antimycin A (mitochondrial electron-transport chain complex I and III inhibitors, respectively) were used to block mitochondrial respiration.
Endosomal escape and lysosomal pH acidification experiments in primary endothelial cells
HUVECs were seeded in eight-well slides (10,000 cells per well) and were infected on the next day with an AAV expressing cell surface GFP or co-infected with the AAV (PHP.eB serotype, Ef1a promoter) and baculovirus expressing RAB5A–TagRFP for labelling early endosomes. After 2 days, cells were transplanted with 10 µg of mito-mTagBFP2 donor mitochondria displaying anti-GFP nanobody or without any binder. Cells were washed 2 h after transplantation with culture medium and live imaged after 6 h and 1 day. For assessment of lysosomal pH changes, a lysosomal acid pH detection kit was used according to the manufacturer’s instructions (Dojindo, L266-10).
Glycolysis inhibition experiments by glucose substitution with galactose in LHON iHNeurons
Patient-derived LHON iHNeurons were induced from iPS cells and seeded on a Matrigel-coated MatTek gridded coverslips dish or an eight-well Ibidi slide (50,000 cells per dish/well). The cells were allowed to recover for a week and then infected with AAVs to express cell-surface anti-GFP or control nanobody and cytosolic tdTomato (PHP.eB serotype, Ef1a promoter). After 3 weeks, cells were transplanted with 5 µg of mito-GFP or remained untreated. After 2 days, the cells were carefully washed with prewarmed PBS and the culture medium was substituted with glucose-free Neurobasal-A medium supplemented with D-galactose (50 mM) and 1% penicillin–streptomycin. Cells were live imaged and tracked over 2 days during starvation using grids in the plate. For cells treated with mitochondria, survival of tdTomato-positive cells positive for mito-GFP was assessed, and survival of tdTomato-positive cells was evaluated for control samples. Moreover, survival of tdTomato+ cells were assessed by using a cell viability calcein dye (Thermo Fisher Scientific, C1430). We restricted the starvation to 2 days due to cell detachment that occurs over a longer period.
Immune response profiling after mitochondria injection
Mitochondria either displaying anti-GFP nanobody or no binder were injected into the retina, systemically through the bloodstream or locally into the brain. For retinal injections, intravitreal injections were performed as previously described. A total of 2.5 µg of mitochondria in 1 µl of buffer was injected into the vitreous chamber. For systemic injections, 20 µg of mitochondria in 30 µl of buffer was injected retro-orbitally. Mice were anaesthetized with isoflurane and received a topical drop of anaesthetic on the eye (oxybuprocaine hydrochloride, 0.4%). The eye was gently protruded from the socket, and the injection was administered at the medial canthus into the retro-orbital sinus with a 28 G syringe. Carprofen (4 mg per kg) was administered before the procedure and provided in the drinking water (10 mg per kg) for 24 h after surgery. For brain injections, mice were anaesthetized with a combination of fentanyl (0.05 mg per kg), midazolam (5 mg per kg) and medetomidine (0.5 mg per kg) and placed onto a heating pad. The surgical area was sterilized with betadine and a midline scalp incision was made from lambda to bregma. A small craniotomy was performed to allow for intracortical injections. Mitochondria were delivered into the cortex of the brain with a nanoinjector (Nanoliter 2020, WPI). A total volume of 150 nl (1.5 µg μl−1) of mitochondria was injected into three different locations of the cortex in each mouse. The skin incision was sutured and the mice received a reversal cocktail of flumazenil (0.5 mg per kg) and atipamezol (2.5 mg per kg). Post-operative analgesia was maintained for 2 days. Blood samples were collected at three timepoints: the day of the injection, and at 7- and 28 days after injection. Tail-vein collection was performed under isoflurane anaesthesia. Tails were warmed to promote vasodilation and approximately 50 µl of blood was collected from the lateral tail vein using a needle. Haemostasis was achieved by applying a sterile swab to the puncture site. ELISAs on blood serum were performed to assess the presence of antibodies against mitochondrial proteins and described below in the ‘ELISA’ section.
Wound-healing assay
One day before the assay, HUVECs were seeded at a density of 25,000 cells per well (50,000 cells per insert) in Ibidi two-well silicone inserts with a cell-free gap (Ibidi, 80209), placed into 24-well plates and topped with endothelial cell growth medium (Bio-techne, CCM027). Cells were kept at 37 °C in a humidified incubator (5% CO2) and grown overnight to reach confluence. On the day of the assay, mitochondria were isolated from HEK293T cells transfected with pcDNA3.1-SNAP-OMP25, enabling SNAP-tag display on the surface of mitochondria. Isolated mitochondria were incubated for 1 h on ice in one of two conditions: (1) anti-CD31 monoclonal antibodies (130-108-038) or (2) mouse IgG1 isotype control (130-106-545), both conjugated to BG. Moreover, control BG-conjugated anti-CD31 antibodies without mitochondria were added to cells. Mitochondria were added to the HUVECs in inserts, 10 μg per insert side (20 μg per insert) suspended in the storage buffer provided with the Qproteome Mitochondria Isolation Kit (Qiagen, 37612). HUVECs were incubated with mitochondria for 3 h at 37 °C. Next, the inserts were gently and swiftly removed to create a defined wound gap. The HUVECs were washed with warm PBS to remove excess mitochondria and cell debris and fresh medium was added to the wells. Time-lapse imaging was initiated immediately using an Olympus confocal spinning-disc microscope capturing one image per hour for 10 h to monitor wound closure. The gap area was quantified over time using ImageJ.
ONC for disease modelling
ONC was performed in PV-cre/tdTomato mice previously transduced with the AAV-ProA5-DIO-αGFP nanobody (PhP.eB). We used the ProA5 promoter to exclude amacrine cells, which also express PV. Mice were deeply anaesthetized with isoflurane and buprenorphine (Temgesic, 0.05 mg per kg) injected subcutaneously. Oxybuprocaine hydrochloride (Novesin) drops were applied locally for analgesia. The eyeball was extracted from the orbit by wide opening of the eyelids, the optic nerve gripped and pressed for 10 s with the tip of surgical forceps and the eye was placed back into the orbit. The ONC was performed unilaterally with the second eye as a control. A suspension of isolated mitochondria was injected intravitreally 1 day after ONC. A similar volume of storage buffer was injected into control eyes without donor mitochondria. Then, 10 days after ONC, the retinas were fixed in 4% PFA in PBS for 1 h for further analysis.
To validate the model, C57BL6 mice were injected intravitreally with retrograde tracer cholera toxin subunit B-Alexa Fluor-555, 647 or 488 (Thermo Fisher Scientific, C34776, C34778 and C34775) in crushed and control eyes at 8 days after crush to label axons of retinal ganglion cells in the brain. Then, at 10 days after crush, mice were transcardially perfused with 4% PFA in PBS and brains were postfixed overnight in the same solution, embedded in 3% agarose in PBS and sliced at a thickness of 100 μm using a vibratome (Leica, VT1000S) for further analysis.
TEM analysis of freshly isolated mitochondria
A pellet of isolated mitochondria was fixed with 2.5% glutaraldehyde (Electron Microscopy Science; 16200) in 0.1 M sodium cacodylate buffer pH 7.4 (Sigma-Aldrich, C0250). The pellet was post-fixed in 2% aqueous osmium tetroxide (Electron Microscopy Science, 19160), dehydrated in gradual ethanol (30–100%), embedded in EMbed 812 epoxy resin (Electron Microscopy Science, 14120) and cured for 24 h at 60 °C. A region of interest was selected under light for TEM analysis. After trimming, silver/grey thin sections (50 nm) were collected on formvar-coated single-slot copper grids (Electron Microscopy Science, FF2010-CU-50). After post-staining with 1% uranyl acetate and lead citrate (5 min each), images were recorded using a FEI Tecnai Spirit (FEI Company) operated at 120 keV using a side-mounted 2 K × 2 K CCD camera (Veleta, Olympus).
SEM analysis of cultured iHNeurons
iHNeurons (15,000–20,000 cells) were cultured on a Matrigel-coated MatTek gridded coverslips dish (MatTek, P35G-1.5-14-C-GRD) for live-cell imaging and subsequent localization. After 3 h, coordinates of cells positive and negative for GFP-tagged donor mitochondria were recorded by both fluorescent and transmitted light microscopy. After imaging, the cells were fixed with 2.5% glutaraldehyde (Electron Microscopy Science, 16200) and 2% paraformaldehyde (Electron Microscopy Science; 15710) in 0.1 M cacodylate buffer pH 7.4 for 1 h. After rinsing with 0.1 M cacodylate buffer pH 7.4, the cells were dehydrated in a graded ethanol series and dried with hexamethyldisilazane (Sigma-Aldrich, 379212). The cells were further dried in an oven at 60 °C for 5 min, and mounted onto aluminium stubs by fixing the coverslip with double-sided carbon tape and sputter coated (Quorum, SC7620) with gold/palladium (5–8 nm). Cells were localized using the coordinates on the gridded coverslips and imaged at 3 kV and 200 pA with a scanning electron microscope (SEM Merlin, Zeiss) using the HE-SE2 detector.
Photooxidation of cultured cells and localization of miniSOG-labelled mitochondria using TEM
Photo-oxidations of cells and sample preparations were performed as previously described25. For HEK293T cells, 10,000–15,000 cells were seeded on a MatTek gridded dish and transfected with miniSOG-encoding plasmid DNA. The next day, cells expressing miniSOG in the matrix of mitochondria were localized by live imaging using an inverted Olympus confocal spinning-disc microscope and their coordinates were determined from the dish grid. The cells were fixed with 2% glutaraldehyde in 0.1 M cacodylate buffer (pH 7.4) for 1 h, rinsed several times with ice cold cacodylate buffer and blocked for 30 min in 50 mM glycine, 10 mM KCN and 5 mM aminotriazole. Diaminobenzidine (DAB) in 0.1 M cacodylate buffer (pH 7.4) solution (1 mg ml−1) was saturated with pure oxygen and poured onto the cells. Live recorded cells were identified and photooxidized for 10 min using the OBIS 488 nm LS 100 mW Laser (Coherent, 88-025) and the Olympus UPlanSApo ×20 objective (Olympus, N1480500). For iHNeurons, 50,000 cells per dish were seeded on the MatTek gridded dish coated with Matrigel and infected with AAVs to express the cell-surface anti-GFP nanobody. After 2 weeks, the cells were transplanted with mito-GFP (2.5 µg). The next day iHNeurons were live imaged to locate cells positive for mito-GFP, fixed with 2% glutaraldehyde in 0.1 M cacodylate buffer (pH 7.4) for 1 h and processed further as described above. Subsequently, dishes with cells were placed on ice and washed with ice-cold cacodylate buffer (5 × 2 min) to remove unpolymerized DAB. After washing, cells were post-fixed with 1% osmium tetroxide (Electron Microscopy Science, 19160) in 0.1 M cacodylate buffer (pH 7.4) for 30 min on ice. The cells were then washed three times with double-distilled H2O and stained with 1% uranyl acetate in double-distilled H2O for 20 min. After five washes in double-distilled H2O and dehydration steps in graded alcohol series, the cells were infiltrated with EMbed 812 epoxy resin, using the hard recipe (Electron Microscopy Science, 14120), for 12 h. The next day, the resin was replaced with freshly prepared resin and polymerized at 60 °C for 24 h. Subsequently, photooxidized regions of interest (ROIs) were identified by transmitted light, sawed out using a jeweller’s saw and mounted onto dummy acrylic blocks with cyanoacrylic adhesive. The glass coverslip was carefully removed, and ultrathin sections (50 nm) containing the photooxidized region of interest were collected on formvar-coated single-slot copper grids (EMS). Images were recorded using a FEI Tecnai Spirit (FEI Company) operated at 120 keV using a side-mounted 2 K × 2 K CCD camera (Veleta, Olympus).
AFM analysis
Cantilever coating for collection of an isolated mitochondrion was done as follows. Silica beads with 5 μm diameter (Kisker Biotech, PSI-5.0) were glued to the free end of a tipless triangular microcantilever (NP-O-D, Bruker Instruments, nominal spring constant, ~0.06 N m−1) using ultraviolet curable glue (Dymax, OP-29) and were cured with ultraviolet light for 30 min. Beaded cantilevers were then cleaned for 2 min using a plasma cleaner (Harrick Plasma)73. Thereafter, the beaded cantilevers were coated with 2 mg ml−1 concanavalin A (Sigma-Aldrich, C5275) for 30 min at 25 °C. Any extra concanavalin A was removed by washing the cantilevers with 1× PBS. Next, the cantilevers were coated with anti-TOMM22 antibody-conjugated microbeads (Miltenyi Biotech, 130-127-693) for 1 h at 25 °C. Any excess beads were removed by washing the cantilevers with 1× PBS.
Freshly isolated mitochondria were added to an empty well of the PDMS insert containing glass-bottomed fluodish (WPI, FD35) at a concentration of 2 µg ml−1. The mitochondria pick-up experiments were performed using an atomic force microscope (Nanowizard II with CellHesion Module, JPK Instruments) mounted onto an inverted epifluorescence microscope (Observer.Z1, Zeiss). The temperature was maintained at 37 °C throughout the experiment by a Petri dish heater (JPK Instruments). The AFM cantilever coated with 5-µm anti-TOMM22-antibody-conjugated microbeads was moved towards the surface at a speed of 5 µm s−1 and was softly pushed (set point force = 2 nN) onto the mitochondria for 60 s. After retraction of the cantilever, the successful pick-up of mitochondria was monitored using the epifluorescence signal.
Mitochondrion–cell unbinding forces were measured using an atomic force microscope (Nanowizard II with CellHesion Module, JPK Instruments) mounted onto an inverted epifluorescence microscope (Observer.Z1, Zeiss). The temperature was maintained at 37 °C throughout the experiment by a Petri dish heater (JPK Instruments). The cantilever-attached mitochondrion was moved towards a cell at a speed of 2 µm s−1 and pushed gently (set point force = 0.5 nN) onto the cell for ~50 ms (0 s contact time). Subsequently, the mitochondrion was retracted at 2 µm s−1 and allowed to recover for a time period equal to that of the contact time with the cell74. A minimum of three unbinding measurements was performed on each cell. Adhesion forces were determined after the retraction force-distance (F–D) curves were drift- and baseline-corrected using the JPK data-processing software (v.8.0.92, Bruker Nano). The maximum adhesive force of the F–D curve was determined by measuring the difference between the minimum force value (maximum cantilever deflection) and the corrected baseline. This is a measure of the force required to separate the mitochondria from a cell. Single events were counted as each force step between the maximum detachment force and the final force step before the F–D curve merges with the baseline and recorded using JPK data processing software (v.8.0.92, Bruker Nano). A cut-off of ~20 pN, which corresponds to twice the noise in our AFM experiments, was used before analysing single force steps75. This is a measure of the frequency with which receptor–ligand interactions occur between the mitochondria and a cell.
Confocal imaging of immunostained and live samples
Images of cultured cells and tissue were acquired using a spinning-disc confocal microscope (Olympus IXplore SpinSR). For fixed samples, objectives with ×10 (UPLXAPO10X, Olympus, N5701900), ×20 (UPlanSApo, Olympus, N1480500) and ×40 (UPLSAPO40XS, Olympus, N4274100) magnifications were used for the acquisitions. For live imaging, the microscope included a cellVivo incubation system (Olympus) to maintain samples at 37 °C and 5% CO2. The samples were imaged with an UPlanSApo ×20 objective and an extra ×3.2 optical zoom for 2 min at 1–2 s intervals.
Super-resolution imaging of immunostained samples
Three-dimensional structured illumination microscopy (3D-SIM) was performed on the DeltaVision OMX-Blaze V4 system (Leica) equipped with 405, 445, 488, 514, 568 and 642 nm solid-state lasers. Images were acquired using a Plan Apo N ×60/1.42 NA oil-immersion objective lens (Olympus) and four liquid-cooled sCMOS cameras (pco.edge 5.5, full frame 2,560 × 2,160; PCO). Exciting light was directed through a movable optical grating to generate a fine-striped interference pattern on the sample plane. The pattern was shifted laterally through five phases and three angular rotations of 60° for each z section. The 405, 488 568 and 642 nm laser lines were used during acquisition and the optical z sections were separated by 0.125 μm. The laser power was attenuated to 10%–50% and exposure times were typically between 5 and 100 ms. The power of each laser was adjusted to achieve optimal intensities of between 5,000 and 8,000 counts in a raw image of 15-bit dynamic range at the lowest laser power possible to minimize photobleaching. Multichannel imaging was achieved through sequential acquisition of wavelengths by separate cameras. Raw 3D-SIM images were processed and reconstructed using the DeltaVision OMX SoftWoRx software package (v.7.0.0 release RC 6, GE Healthcare). The resulting size of the reconstructed images was 1,024 × 1,024 pixels from an initial set of 512 × 512 raw images. The channels were aligned in the image plane and around the optical axis using predetermined shifts as measured using a target lens and the SoftWoRx alignment tool. The channels were then carefully aligned using an alignment parameter from control measurements with 0.5 µm diameter multi-spectral fluorescent beads (Invitrogen, Thermo Fisher Scientific).
Ex vivo two-photon calcium imaging of mouse retinal ganglion cells
For ex vivo calcium imaging, we expressed a Cre-dependent GCaMP8m calcium indicator (Addgene, 162378) specifically in retinal ganglion cells using ProA5 promoter using AAVs76. Two-photon live imaging of ganglion cells was performed as previously described77. In brief, retinas from light-adapted mice were isolated under red dim light in oxygenated Ringer’s medium. Isolated retinas were mounted with the ganglion-cell-side up on filter paper with an aperture of 1.5 × 3 mm positioned under the ventral side of the retina. The retina was perfused in a recording chamber with Ringer’s medium with a flow rate of 1.5 ml min−1 at 35 °C for the entire duration of the experiment. The ganglion cell bodies in the ventral retina were imaged using a custom-built two-photon imaging system equipped with a Mai Tai two-photon laser (Spectra Physics) tuned to 920 nm, a ×60 water-immersion objective (Fluor, 1.0 NA, Nikon) and a 510/20 nm band-pass emission filter. The laser power at the sample was kept lower than 20 mW. The system acquired images of 200 × 200 µm at 1 µm px−1 at a frame rate of 5.71 Hz. For measuring the light responses, light stimulation (LED 405 nm) of a spot (250 µm diameter) was delivered onto the retinal region inside of the paper filter aperture for 2 s. The light intensities for stimulation (100%) and background (0%) were 134 and 0.153 µW cm−2, corresponding to 2.31 × 105 and 2.64 × 102 R* s−1, respectively. The stimulation light was turned on during the fly-back periods of the horizontal scanning mirror to minimize PMT noise originating from the stimulation light. Each light stimulation was repeated three times.
The data were analysed with Python and MATLAB. Ganglion cell bodies were segmented manually. To compute ∆F/F of each region of interest, the background was first calculated as the mean of the 10% dimmest pixels in each frame, and this was subtracted from all pixels. Then, all of the pixels in each region of interest were averaged in each frame and ∆F/F was calculated. F was the baseline calculated as the mean fluorescence within the 2 s time window before the stimulation onset. Three repetitions were averaged to give the mean ∆F/F curve. The amplitude of the response was calculated as the maximum ∆F/F in the 2 s time window after the onset/offset of the light stimulation. Those peaks with signal-to-noise ratio (SNR) higher than 2 were considered as light responses, where SNR is the response amplitude/s.d. of the baseline.
Immunostaining of cultured cells, organoids and mouse and human retinas
The blocking solution used for the immunostaining contained 10% normal donkey serum (NDS, Sigma-Aldrich, S30-M), 1% BSA (Sigma-Aldrich, A4503), 0.5% Triton X-100 (Sigma-Aldrich, 93443) and 0.01% sodium azide (Sigma-Aldrich, S2002) in PBS. The staining solution consisted of PBS containing 3% NDS, 1% BSA, 0.5% Triton X-100 and 0.01% sodium azide in PBS, and was supplemented with primary antibodies.
Cultured cells were fixed for 30 min in 4% PFA, washed three times with PBS and blocked for 1 h in blocking solution. The blocking solution was exchanged with staining solution and the cells were stained with the primary antibodies at 4 °C overnight. The cells were then washed with PBS and stained for 1 h at room temperature with secondary antibodies diluted in the staining solution. The cells were stored and imaged in PBS; the images were acquired within several days. For long-term storage, 0.01% sodium azide was added to the PBS.
Organoids and mouse and human retinas were fixed for 1 h in 4% paraformaldehyde diluted in PBS. The tissues were washed in PBS and placed in 30% sucrose in PBS for cryopreservation. Before sectioning or staining, the tissues underwent three freeze–thaw cycles using dry ice. The sucrose was then removed by washing several times with PBS. For mouse and human whole-mount retinas, the tissue was blocked for 1 h in blocking solution. For organoids, they were embedded in 3% agarose (Lonza, 50110), cut into 80–100μm vibratome sections (Leica, VT1000S vibratome) and blocked for 1 h in blocking solution. The tissue was then stained for 3–5 days at room temperature with primary antibodies in the staining solution. After washing with PBS, the tissue was stained with secondary antibodies in the staining solution for 2 h at room temperature and mounted on Super-Frost Ultra Plus GOLD slides (Thermo Fisher Scientific, 11976299), embedded with ProLong Gold antifade mounting medium (Thermo Fisher Scientific, P36934).
The primary antibodies used in this study for immunostaining were as follows: polyclonal chicken anti-GFP (1:500, Thermo Fisher Scientific, A10262), polyclonal chicken anti-RFP (1:500, Rockland, 600-901-379), polyclonal rabbit anti-OLLAS (1:500, Genscript, A01658), polyclonal rabbit anti-TagRFP (1:250, Evrogen, AB233), polyclonal rabbit anti-TOMM20 (1:500, Sigma-Aldrich, HPA011562), polyclonal rabbit anti-CD71 (1:250, Thermo Fisher Scientific, PA5-85936), monoclonal mouse anti-Rhodopsin (1:500, Sigma-Aldrich, R5403), polyclonal rabbit anti-SNAP (1:200, NEB, P9310S), monoclonal mouse anti-MT-CO1 (1:250, Thermo Fisher Scientific, 459600), monoclonal mouse anti-CD31 (1:500, BD Biosciences, 550389), monoclonal mouse anti-CD90 (1:250, Thermo Fisher Scientific, 14-0909-82), monoclonal mouse anti-TUBB3 (1:250, BioLegend, 801202), polyclonal rabbit anti-RBPMS (1:500, phosphosolutions, 1830-RBPMS) and monoclonal rabbit anti-CD73 (1:250, Abcam, AB133582).
The secondary antibodies used in this study for immunostaining were as follows: Alexa Fluor 647 goat anti-alpaca IgG (against VHH domain, Jackson, 128-605-232), Alexa Fluor 405 donkey anti-rabbit IgG (H+L, Thermo Fisher Scientific, A48258), Alexa Fluor 488 donkey anti-rabbit IgG (H+L, Thermo Fisher Scientific, A21206), Alexa Fluor 568 donkey anti-rabbit IgG (H+L, Thermo Fisher Scientific, A10042), Alexa Fluor 647 donkey anti-rabbit IgG (H+L, Thermo Fisher Scientific, A31573), Alexa Fluor 488 donkey anti-mouse IgG (H+L, Thermo Fisher Scientific, A21202), Alexa Fluor 568 donkey anti-mouse IgG (H+L, Thermo Fisher Scientific, A10037), Alexa Fluor 647 donkey anti-mouse IgG (H+L, Thermo Fisher Scientific, A31571), Alexa Fluor 488 goat anti-chicken IgY (H+L, Thermo Fisher Scientific, A11039) and Alexa Fluor 568 goat anti-chicken IgY (H+L, Thermo Fisher Scientific, A11041). All secondary antibodies were used at a dilution of 1:500.
Antibodies used for pluripotency validation are described previously in the ‘Reprogramming and characterization of LHON iPS cells’ section. For mitochondria staining, MitoTracker Deep Red was used (50 nM, Thermo Fisher Scientific, M22426) For actin filament staining, Phalloidin conjugated to Alexa Fluor 488 was used (1:1,000, Abcam, ab176753). For lysosome labelling, pHLys Red (1:1,000, Dojindo, L266-10) and LysoTracker Deep Red (5 nM, Thermo Fisher Scientific, L12492) dyes were used. To label early endosomes, CellLight Early Endosomes-RFP baculovirus (Thermo Fisher Scientific, C10587) was used according to the manufacturer’s instructions.
ELISA
All assays were run in 384-well Maxisorp plates (Nunc). All reagents had a volume of 25 µl. Immobilization of capturing reagents or target proteins was performed at the following concentrations in PBS over night at 4 °C: neutravidin (1.25 µg ml−1, Invitrogen, A2666), anti-hFc antibody (1 µg ml−1, Southern Biotech, 2048-01), fluorescent proteins (eGFP, mCherry, dsRed and tdTomato all 5 μg ml−1, produced in house), anti-GFP VHH (2 µg ml−1, produced in house), TOMM70 (1 μg ml−1, produced in house), TOMM20 (1 µg ml−1, produced in house), purified mitochondria (5 µg ml−1, produced in house). Blocking was performed by adding 100 µl of PBS with 1% BSA for 1.5 h at room temperature. All of the following protein steps were performed in PBS with 0.05% Tween-20 and 1% BSA for >30 min at room temperature. After each step, the plates were washed three times with 70 µl PBS with 0.05% Tween-20 (six times after detection antibody). The following proteins were used in the proteins steps in different combinations: biotinylated TOMM70 (1.8 µg ml−1, produced in house, see above), hCD71-Fc (1.5 µg ml−1, Sino Biological, 11020-H01H), avi-tagged mCherry (2.5 µg ml−1, produced in house), MYC-tagged mCherry (2 µg ml−1, produced in house), VHHs and VHH bispecifics (50 nM or concentration series, produced in house, see above), mouse serums (dilution series starting at 1:200). Positive control antibodies were as follows: goat anti-alpaca IgG, VHH domain (Jackson IR, 128-005-230, 1:1,000), anti-TOMM70 antibody (SantaCruz, sc-390545, 1:1,000), anti-TOMM20 (SantaCruz, sc-17764, 1:2,000). Detection antibodies were as follows: anti-human IgG Fc antibody (HRP) (Genscript, A01854-40, 1:5,000), anti-His-tag antibody (HRP) (Genscript, A00612, 1:2,500), anti-MYC antibody (HRP) (Genscript, A00863, 1:5,000) and rabbit anti-goat IgG(H+L)-HRP (Southern Biotech, 6160-05, 1:5,000). 1-Step Ultra TMB-ELISA substrate (Thermo Fisher Scientific, 34024) or OBP substrate (Thermo Fisher Scientific, 34006) was added for 5–10 min and stopped with 2 M sulphuric acid. Absorbance was measured at 450 nm (TMB substrate) or 490 nm (OBP substrate) and plotting and fitting were performed in GraphPad Prism 10.
mtDNA sequencing of patient-derived iPS cell clones
Total DNA from iPS cells was isolated using the DNeasy Blood & Tissue Kit (Qiagen, 69504). Furthermore, mtDNA was amplified using the REPLI-g Mitochondrial DNA Kit (Qiagen, 151023). Primers for human mtDNA amplification were included in the kit. The amplified product was column purified using the DNA Clean & Concentrator-5 Kit (Zymo Research, D4014) and eluted in ultrapure water. All of the above-mentioned procedures were performed according to the manufacturer’s instructions. Whole-genome sequencing was performed at CeGaT using the NovaSeq 6000 (Illumina) system as 100-bp paired-end reads.
Genomic mapping, identification of genetic variations and variant annotation
For each sample, we aligned the raw genome-sequencing data to the human reference genome sequence (build hg19/GRCh37) using BWA-MEM (v.0.7.17)78. The alignment results in SAM format were converted to the BAM format using the SortSam tool from the Picard suite (v.2.14.0-SNAPSHOT), and samtools (v.1.10) for indexing. We identified duplicate reads in the BAM files using Picard’s MarkDuplicates module. On the basis of the GATK Best Practices workflow79, base quality score recalibration (BQSR) was performed (v.4.1.4.1), followed by variant calling using HaplotypeCaller in gVCF mode80. The gVCF files from each sequencing batch were first merged with CombineGVCFs. Subsequently, these merged gVCF files were converted into a single VCF file per batch using GenotypeGVCFs81. We used VariantRecalibrator and ApplyVQSR81 to recalibrate the quality of single-nucleotide polymorphisms and insertions–deletions (indels). The view and norm functions in bcftools (v.1.10.2) were applied to process each VCF file. Annotation of variants obtained from genome sequencing was performed by ANNOVAR82.
Proteomics for iHNeurons transplanted with mitochondria
Cells were lysed with buffer containing 100 mM TEAB pH 8.5, 5% SDS, 10 mM TCEP by heating for 10 min at 95 °C, followed by 10 min sonication (30 s on, 30 s off per cycle) on a Pixul system (Active Motif, 53130) using the following settings: Pulse[N]: 50; PRF[kHz]: 1; burst rate[Hz]: 20. Protein extracts were alkylated using 15 mM iodoacetamide at 25 °C in the dark for 30 min. For each sample, 100 µg of protein lysate was captured, digested and desalted using S-Trap cartridges (Protifi, CO2-micro). In brief, samples were acidified by addition of 10% TFA (1:10) and then S-trap buffer (90% methanol, 100 mM TEAB pH 7.1) was added to the samples (6:1). Samples were briefly vortexed and loaded onto S-trap micro spin-columns (Protifi, CO2-micro) and centrifuged for 1 min at 4,000g. The flow-through was discarded and spin-columns were then washed three times with 150 µl of S-trap buffer (each time samples were centrifuged for 1 min at 4,000g and flow-through was removed). S-trap columns were then moved to the clean tubes and 20 µl of digestion buffer (50 mM TEAB pH 8.0) and trypsin (at 1:25 enzyme to protein ratio) were added to the samples. Digestion was allowed to proceed for 1 h at 47 °C. Next, 40 µl of digestion buffer was added to the samples and the peptides were collected by centrifugation at 4,000g for 1 min. To increase recovery, S-trap columns were washed with 40 µl of 0.2% formic acid in water (400g, 1 min) and 35 µl of 0.2% formic acid in 50% acetonitrile. Eluted peptides were dried under vacuum and stored at −20 °C until further processing. Moreover, phosphopeptides were enriched using AssayMAP Fe(III)-NTA cartridges, but these enriched fractions were not included in the final analysis. The peptides were subjected to LC–MS/MS analysis using the Orbitrap Exploris 480 Mass Spectrometer fitted with an Vanquish Neo (both Thermo Fisher Scientific) and a custom-made column heater set to 60 °C. Peptides were resolved using a RP-HPLC column (75 μm × 30 cm) packed in-house with C18 resin (ReproSil-Pur C18–AQ, 1.9 μm resin; Dr. Maisch) at a flow rate of 0.2 μl min−1. Separation of peptides was achieved using the following gradient: from 2% buffer B to 10% buffer B over 5 min; to 35% buffer B over 45 min; to 50% buffer B over 10 min. The mass spectrometer was operated in data-independent acquisition (DIA) mode. Each MS1 scan was acquired at a resolution of 120,000 FWHM (at 200 m/z) and a scan mass range from 390 to 910 m/z. The maximum injection time was set to auto mode with a normalized AGC target set to 300%. Each survey scan was followed by a set of DIA scans acquired at a resolution of 15,000 FWHM. Precursor mass range was set to 400–900 m/z, the isolation windows size to 10 m/z and the HCD normalized collision energy to 28%. For MS2 scans, the maximum injection time was set to 22 ms with a normalized AGC target set to 3000%. The acquired raw files were searched using SpectroNaut (v.19.0, directDIA workflow, default settings) against a human database (downloaded from UniProt on 22 February 2022) using the following search criteria: full tryptic specificity was required (cleavage after lysine or arginine residues, unless followed by proline); three missed cleavages were allowed; carbamidomethylation (C) was set as a fixed modification; oxidation (M), N-acetylation (N-term) The identified transitions were exported as .tsv files and processed using the MSstats R package (v.4.0.1)83.
Proteomics analysis
Data were normalized by sample-wise equalizing medians and missing values were retained without imputation. Limma (v.3.62.2)84 was used to identify differentially abundant proteins for each pairwise comparison of treatments based on t-tests with empirical Bayes variance moderation. Pre-rank gene set enrichment analysis was performed for each comparison using clusterProfiler (v.4.14.0)85, incorporating gene ontologies for biological processes, molecular functionality and cellular components. Proteins were ranked based on the log-transformed fold change estimated by limma, with no log fold change threshold applied. Benjamini–Hochberg adjustment was performed to correct for multiple testing. The most significant terms in the anti-GFP nanobody treatment protein data versus untreated cells comparison were selected for visualization. Terms that were child terms of other selected terms were removed. To assess the degree of mitochondrial enrichment compared with other cellular compartments, gene set enrichment analysis was performed on custom protein lists of known marker proteins (Supplementary Table 2).
Single-cell FLASH-seq in transplanted LHON iHNeurons
GFP-positive LHON cells were collected 2 days after mitochondrial transplantation by single-cell picking, and full-length scRNA-seq was performed. Strip tubes containing 4.5 µl of guanidine lysis buffer were prepared for single-cell collection. The buffer consisted of 248 mM GuHCl, 6 mM dNTPs, 1.8 µM oligo-dT, 1.2 mM DTT, 1 M betaine and nuclease-free water. Then, 2 days after mitochondrial transplantation, cells were washed and perfused with Ringer solution (110 mM NaCl, 2.5 mM KCl, 1 mM CaCl2, 1.6 mM MgCl2, 22 mM NaHCO3 and 10 mM d-glucose) bubbled with 95% O2 and 5% CO2, at 37 °C. The flow rate was maintained at 1.5 ml min−1. The presence of GFP was assessed using a collimated LED (470 nm, 487 mW). Glass pipettes of diameter 10–20 µm were used to collect GFP+ cells by placing the pipette near the cell and applying negative pressure. For trypsin pretreatment experiments, 0.05% trypsin-EDTA (Thermo Fisher Scientific, 25300-054) was applied locally to a single cell through a glass micropipette for 1 min without perfusion. The application pipette was then withdrawn and the cell was collected with a second glass pipette. Cells were transferred to tubes containing lysis buffer and immediately stored at −70 °C. The samples were then processed according to the FLASH-seq protocol86,87, starting with sample denaturation. RNA from LHON cells transplanted with mito-GFP (LHON_mito_transplanted_2d) and control non-transplanted LHON cells (Control_LHON_no_mito_transplanted) was converted to cDNA using Superscript IV (Thermo Fisher Scientific, 18090200). RNA from LHON cells transplanted with mito-GFP and treated with trypsin-EDTA (LHON_mito_transplanted_2d_protease) and untreated controls (Control_LHON_no_mito_transplanted_protease), and LHON cells transplanted with mtDNA-free mito-GFP (LHON_mito_transplanted_2_empty_mito) was converted to cDNA using Maxima H Minus Reverse Transcriptase (Thermo Fisher Scientific). All cDNA was amplified using the KAPA HiFi HotStart (Roche, KK2602) and then cleaned using a 0.8× ratio of homebrew SeraMag beads in 18% PEG (Cytivia, GE24152105050250). cDNA concentrations and quality were measured using the Qubit 1× dsDNA High Sensitivity Assay Kit (Thermo Fisher Scientific, Q33231) and the Agilent Bioanalyzer (Agilent, 5067-4626). The cDNA was normalized to 200 pg μl−1 before tagmentation using 0.2 μM of homemade Tn586. The reaction was halted with 0.2% SDS. An indexing PCR was performed to add index adaptors carrying an 8-bp barcode (IDT Technologies) using KAPA HiFi reagents (Roche, KK2102). Libraries were pooled in equal volumes and a final 0.8× cleanup was performed with homebrew SeraMag beads before measuring sample concentration and quality. LHON_mito_transplanted_2d and Control_LHON_no_mito_transplanted libraries were sequenced on the NextSeq 550 (Illumina) system to either 75- or 100-bp single-end reads with 8-bp dual indexes. LHON_mito_transplanted_2d_protease were sequenced on either the NextSeq 550 (Illumina) or AVITI (Element Biosciences) system to 75-bp paired-end reads with 8 bp dual indexes. LHON_mito_transplanted_2d_empty_mito libraries were sequenced on the NextSeq550 (Illumina) system to 75-bp paired-end reads with 8-bp dual indexes.
Single-cell transcriptome analysis
Sequencing reads were processed to obtain gene counts using Snakemake (v.7.21.0)88, a robust workflow management system. The workflow comprised three key steps. The first step is quality control. The tool BBDuk from BBMap (v.39.01) was used for removing remaining sequencing adapters, while Trimmomatic (v.2.6.0)89 was used for cropping potentially low-quality bases from the 3′-end and removing short reads such that reads had at most 75 bp and at least 45 bp. In a second step, reads were aligned to the GRCh38 reference genome (Ensembl release 110) using STAR (v.2.7.10b)90. This yielded two essential outputs: alignments translated into transcript coordinates (–quantMode TranscriptomeSAM) and the quantification of read counts per gene (–quantMode GeneCounts). Gene counts per cell were subsequently aggregated using custom Python scripts. The third step includes downstream quality control and counting of nucleotide polymorphisms within the MT-ND4 gene. Before variant counting, duplicate reads were marked with Samtools markdup (v.1.17) to avoid inflation of nucleotide counts. Quality assurance measures were implemented by calculating, for each sequencing run, the mean and s.d. of per-sample read depth at the MT-ND4 gene and flagging samples with mean depth below one s.d. from the group mean, or—when this threshold was non-positive—below the fifth percentile of the group distribution. Moreover, we retained only samples with a read depth in MT-ND4 of greater than 30. For the MT-ND4 gene, nucleotide counts were calculated while excluding marked duplicates and spliced alignments spanning the MT-ND4 region to retain only variant-supporting reads. We then estimated the fraction of donor mitochondria using the frequency of MT11778G relative to the total of MT11778G and MT11778A.
Single-cell annotation followed the strategy outlined in a previously published study33, with initial quality-control filtering to remove cells with fewer than 200 expressed genes and genes expressed in fewer than three cells. Using the Leiden algorithm (leidenalg, v.0.10.1), we clustered single cells based on a neighbourhood graph constructed from highly variable genes. Highly variable genes were called using a batch-aware procedure, with batches defined based on the experimental batch. We ran Scanpy’s (v.1.9.5) highly variable genes routine with the default settings, except for 40 mean-variance bins, and selected the top 200 genes per batch; a gene was retained as a highly variable gene if it was called variable in ≥2 batches. During the embedding and clustering stages, transcripts from mitochondrial and ribosomal protein-coding genes—commonly characterized by high expression and dispersion, independent of cell type identity—were excluded. Cells were then manually annotated into major types using established markers33.
For downstream analyses, we focused on neurons and retained cells with ≥1,000 expressed genes, which led to the removal of 24 low-quality cells (5 control cells, 0 transplanted with donor mitochondria, 15 transplanted with donor mitochondria and then trypsin-treated, and 4 transplanted with mtDNA-depleted, ρ0, donor mitochondria). We used DESeq2 (v.1.38)91 to identify genes that were differentially expressed dependent on the frequency of donor mitochondria. This analysis used a log2-transformed fold change threshold of 1 and a 5% significance level after the Benjamini–Hochberg correction for multiple-hypothesis testing. Next, the GSEApy (v.1.0.6)92 pre-rank functionality was used for gene set enrichment analysis, incorporating gene ontologies for biological processes, molecular functionality and cellular components. In particular, the genes were ranked based on the Wald test statistic from DESeq2.
Measurement of an individual mitochondria area in TEM images
iHNeurons with transplanted mitochondria (miniSOG-mito-GFP) were localized, photooxidized and imaged under EM as described above. After photo-oxidation by blue light and oxygen, miniSOG generated a strong dark stain with high contrast visible under light and electron microscopes; thus, mitochondria were classified as miniSOG-positive and miniSOG-negative based on contrast levels. Mitochondria area in TEM images was measured in ImageJ by manually selecting ROIs in the photooxidized and neighbouring cells.
Quantification of LHON iHNeuron survival after glycolysis inhibition
The survival rate of LHON iHNeurons after glycolysis inhibition was evaluated by quantifying tdTomato+ cells in a gridded dish. The imaging set-up included one channel to detect tdTomato+ cells, a second bright-field channel to locate cells within the gridded dish for easier tracking over time, and a third channel to monitor the fluorescence of donor mitochondria (mito-GFP) in transplanted iHNeurons. For each biological replicate, images were captured from three different locations on the gridded dish. At the outset (day 0), control samples with tdTomato+ cells were defined as ROIs, with 30–50 cells per dish, and monitored for changes over a 2-day period. Similarly, in the day 0 mito-GFP transplanted samples, ROIs were established for tdTomato-and mito-GFP-positive cells and tracked for the same duration. The leak of tdTomato fluorescence from the cells served as an indicator of cell death. The cell survival rate was defined as the ratio between number of ROIs at day 2 and number of ROIs at day 0.
Quantification of retinal ganglion cell survival after optic nerve injury
The survival rate of retinal ganglion cells following optic nerve injury (ONC) was assessed by quantifying the density of tdTomato-labelled cells in the retina positive for RBPMS, a retinal ganglion cell marker. Quantification was conducted manually using the Cell Counter plugin in ImageJ. For each whole-mounted retina, images were captured at eight locations using a ×20 objective, encompassing both central and peripheral regions. In each image, five ROIs (100 µm × 100 µm) were selected to ensure comprehensive coverage of the entire retinal area. The numbers of counted cells were averaged per retina and shown as cell density (cells per mm2).
Quantification of RGC axonal projections to the dLGN after optic nerve injury
To assess the absence of axonal projections from retinal ganglion cells to the dorsal lateral geniculate nucleus (dLGN) after ONC, the fluorescence intensity of retinal ganglion cell axon terminals in the dLGN was quantified using ImageJ. For ONC samples, mice with retinas correctly injected with cholera toxin were selected prior to brain analysis. Images of the dLGN were captured in three different brain slices from both control and ONC mice using a ×10 objective. The dLGN was outlined as a ROI based on cell density, while another ROI was selected outside the dLGN for background measurements. Mean grey values were measured within both the dLGN and the background ROIs. A ratio was calculated by dividing the mean grey value obtained from the dLGN ROI by that of the background ROI. This ratio reflects the normalized fluorescence intensity within the dLGN region, accounting for background fluorescence or noise. The three values obtained from each subject were averaged to represent a sample.
Mitochondrion enrichment in HEK293T cells, iHNeurons, blood vessel organoids, and mouse and human retina
Mitochondrion enrichment in cells was determined using custom-written routines in MATLAB (MathWorks). Per experiment, several (2–5) images were acquired at different spatial locations on the biological sample. Each image contained a channel representing cell-type-specific surface marker fluorescence (further referred to as marker-positive and marker-negative), a second channel representing donor mitochondria fluorescence and the last channel labelling all cells in the image.
First, for each acquired image, marker-positive and marker-negative cells were manually chosen as ROIs in ImageJ (https://imagej.net). Then, all channels as well as ROIs were loaded into MATLAB. The mean mitochondria fluorescence was computed for each ROI. To correct for non-specific background fluorescence in the acquired mitochondria images, as is commonly done for neuropil correction in calcium imaging93, the median fluorescence within a 10-pixel-wide ring at 10-pixel distance from the ROI edge was subtracted from the computed mean mitochondria fluorescence for each ROI. To ensure that the ring does not contain other marker-positive cells, only the quarter of pixels with the lowest marker fluorescence was included. After obtaining a single corrected mitochondria fluorescence value for each marker-positive and marker-negative cell, the mitochondrial enrichment of the whole image was defined as the ratio between the mean of the corrected mitochondria fluorescence in marker-positive cells and the mean of the corrected mitochondria fluorescence in marker-negative cells. Moreover, the percentage of marker-positive cells significantly enriched in mitochondria was defined as the fraction of marker-positive cells with a corrected mitochondria fluorescence of more than 2 s.d. above the mean of the corrected mitochondria fluorescence of marker-negative cells.
For mitochondrial targeting experiment involving cytosolic tdTomato cells in HEK293T cells (Extended Data Fig. 1) additional analysis was performed. Owing to strong expression of tdTomato, it led to substantial bleedthrough in the green mitochondria channel; therefore, a linear regression correction was applied. The contribution of tdTomato fluorescence to the green signal was estimated by considering pixels which simultaneously had more than median intensity in the red channel (to only consider tdTomato-positive locations) and less than the 99th-percentile intensity in the green channel (to ignore a true mitochondria signal). The green intensity contributed by tdTomato fluorescence was then determined by a linear fit with offset and removed from the green channel. Negative values were set to 0. Finally, the normal enrichment algorithm comparing mitochondria fluorescence in tdTomato+ and tdTomato− cells was implemented as usual.
Mitochondrial enrichment in human cardiac cells, fibroblasts and HUVECs
Mitochondrial enrichment in the cells was computed slightly differently than previously described, given that there were no marker-negative cells. As before, several (2–5) images were acquired per experiment at different spatial locations on the biological sample. Each image contained a channel representing cell-type-specific surface marker fluorescence and a second channel representing donor mitochondria fluorescence.
For each acquired image, marker-positive cells were manually chosen as ROIs in ImageJ (https://imagej.net). Given that no marker-negative cells could be chosen that way, they were instead randomly simulated. For each marker-positive ROI in a given image, a marker-negative ROI was defined by randomly rotating and repositioning the marker-positive ROI mask in the original image. It was ensured that simulated ROIs did not overlap each other or the original marker-positive ROIs. Moreover, for each simulated ROI, it was verified that they were marker-negative by ensuring that their mean fluorescence was below an intensity-based threshold dependent on the mean marker fluorescence of the marker-positive ROIs in that image. For human cardiomyocytes, the threshold was defined as one fifth of the median fluorescence intensity across all marker-positive ROIs. For samples of endothelial cells in which the background fluorescence was higher, the threshold was defined as half of the median fluorescence intensity across marker-positive ROIs. The random simulation algorithm was repeated until it converged and produced the right number of marker-negative ROIs satisfying all defined criteria. The mitochondrial enrichment in marker-positive and marker-negative ROIs was then computed as described above.
Given that marker-negative ROIs were, by design, randomly computed, the randomization procedure was repeated ten times independently for each image. The obtained values of mitochondrial enrichment and percentage of significantly enriched cells were then averaged across repeats from a single image before being combined across images from the same experiment as before.
Mitochondrial enrichment in retinal organoids
Mitochondrial enrichment in retinal organoids was computed differently. For each organoid, several (2–3) organoid sections were acquired for analysis. Each image contained a channel representing photoreceptor layer, a second channel representing all cells and a third channel representing mitochondria fluorescence. For this dataset, the total enrichment of mitochondria fluorescence in the photoreceptor layer was computed. For each image, photoreceptor-positive and photoreceptor-negative regions were selected manually in ImageJ. Mean fluorescence of the channel representing mitochondria for each ROI was computed. The mitochondrial enrichment of the whole image was defined as the ratio between the mean of the nanobody fluorescence in photoreceptor-positive ROI and the mean of the nanobody fluorescence in photoreceptor− ROI. Obtained enrichment values were averaged across several images of a given sample.
Mitochondrial enrichment in T cells
Data for targeting T cells were acquired by flow cytometry, not confocal imaging. Thus, the mitochondrial enrichment in these cells were quantified differently. The targeting was performed on non-activated CD8+ T cells, activated CD8+ T cells overexpressing CD71, and non-activated CD4+ cells. Depending on the experiment, the CD8+, CD71+ or CD4+ populations were first identified and then the fraction of cells positive for mito-DsRed2 within this population was defined. Gating strategies to define the cell populations are described in the ‘Flow cytometry’ section.
TagBFP (destabilized nanobody fused to TagBFP) enrichment in iHNeurons expressing either GFP or TdTomato in fixed cells
Per experiment, at least three images were acquired at different spatial locations of the biological sample. Each image contained a channel representing GFP fluorescence, a second channel representing destabilized nanobody fused to TagBFP and the last channel labelling all neurons in the image. Enrichment of destabilized nanobody was computed as the previously described mitochondrial enrichment quantifying TagBFP fluorescence enrichment in GFP-positive versus GFP-negative cells.
TagBFP enrichment in iHNeurons expressing either GFP or tdTomato in live cells
Enrichment of destabilized nanobody during live imaging of GFP-positive and tdTomato-positive cells was computed in a different way to the fixed samples, as it was not possible to trivially identify GFP- or tdTomato-negative cells. Thus, an approach was used based solely on an automated image segmentation procedure, subdividing the GFP or tdTomato fluorescence image into three distinct regions (GFP positive or tdTomato positive, intermediate and GFP negative or tdTomato negative) depending on the relative local fluorescence in the image, taking into account possible heterogeneity in background fluorescence.
To determine GFP- or tdTomato-positive regions, the GFP or tdTomato fluorescence image was first smoothed by a relatively large 2D-Gaussian filter of 30-pixel s.d. The filtered image was then subtracted from the original image to estimate local fluorescence above the background. The resulting image underwent a combination of small morphological filtering operations (closing followed by opening with a circular 3-pixel mask) to remove small noise in the processed image. The outcome was then thresholded based on intensity (40 in the case of neurons and 20 in the case of HEK293T cells) and finally dilated by a morphological circular filter of pixel size 2.
To estimate conservative GFP- or tdTomato-negative regions, a similar method was used by first determining an intermediate region computed by finding all pixels above a lower intensity threshold than before (10 in the case of neurons and 5 in the case of HEK cells). Smaller morphological filters (circular 1 pixel mask) were used to remove less noise. Moreover, the intermediate region was dilated by a large circular filter of pixel size 20. The GFP- or tdTomato-negative region was defined as any part of the image that is not part of the intermediate region or the GFP-positive or tdTomato-positive region.
Enrichment of destabilized nanobodies was finally computed as the ratio between the mean nanobody fluorescence in the GFP- or tdTomato-positive regions and the GFP- or tdTomato-negative regions. As before, to obtain a single value of this quantity per experimental repetition, the nanobody enrichment was averaged across 2–5 acquired images of the same sample.
TagBFP enrichment in iHNeurons transplanted with mito-GFP
Per experiment, at least three images were acquired at different spatial locations of the biological sample. Each image contained a channel representing cell-surface nanobody fluorescence, a second channel representing destabilized nanobody fused to TagBFP and the last channel labelling all neurons in the image. Enrichment of destabilized nanobodies was computed in the same way as the previously described mitochondrial enrichment quantifying TagBFP fluorescence enrichment in nanobody-positive versus nanobody-negative cells.
Mitochondrial targeting specificity
We defined the specificity of mitochondrial targeting as the ratio between the percentage of donor-mitochondrion-positive cells targeted with a specific binder and the sum of the percentages of donor-mitochondrion-positive cells targeted by both specific and control binders. To normalize this index, the ratio was first subtracted by a value of 0.5 and then multiplied by 2 to have values ranging from −1 to 1. A value of 1 indicated maximal specificity, 0 indicated no specificity and −1 indicated maximal negative specificity.
Co-localization of donor mitochondria with a mitochondrial marker
The co-localization of donor mitochondria with a mitochondrial marker (TOMM20 or MT-CO1) was determined using custom-written routines in MATLAB (MathWorks). First, TOMM20- or MTCO1-positive cells were manually chosen as ROIs in ImageJ (https://imagej.net) before loading the donor mitochondria and mitochondrial marker images, as well as the ROIs, into MATLAB. Then, both the donor mitochondria and mitochondrial marker images were thresholded based on their intensity: 1 s.d. above the mean of the donor mitochondria fluorescence for HEK293T cells, 0.5 s.d. above the mean of the donor mitochondria fluorescence for iHNeurons and heart cells; 3 s.d. above the mean of the mitochondrial marker fluorescence for HEK293T cells and 1 s.d. above the mean of the mitochondrial marker fluorescence for iHNeurons and heart cells, to obtain binary images. For each ROI, the fraction of donor-mitochondrion-positive pixels among TOMM20- or MT-CO1-positive pixels was computed to assess the amount of colocalization. TOMM20- or MT-CO1-negative pixels, or pixels outside of ROIs were ignored in the analysis.
To check for unspecific co-localization values that would increase with increasing amounts of donor mitochondria within ROIs, a spatial randomization method was applied. For any given ROI, the donor-mitochondrion-positive pixels were spatially randomized within the ROI, while the location of TOMM20- or MT-CO1-positive pixels was maintained constant. The colocalization value was then computed as before. The randomization process was repeated 1,000 times for each ROI and the final unspecific co-localization value defined as the mean across the 1,000 iterations.
Co-localization of donor mitochondria displaying SNAP-tags with BG–anti-CD31 or BG–IgG1
The co-localization of SNAP-tags with BG-conjugated anti-CD31 antibodies or IgG1 isotype control was performed as discussed above. Images contained a channel representing SNAP-tag fluorescence and a channel representing either anti-CD31 or IgG1 fluorescence. The values were averaged across two to five acquired images of the same sample.
Co-localization of donor mitochondria with endosomes and lysosomes
To determine endosome/lysosome-free mitochondria, a procedure similar to the colocalization analysis of donor mitochondria with mitochondrial markers described previously was employed. Per field of view, one cell was selected for analysis and a background region was designated for threshold determination. Within the selected cell, pixels were classified as mitochondrion-positive if their fluorescence intensity exceeded the mean background fluorescence by more than 5 s.d. in the mitochondrion-containing channel. Similarly, pixels were classified as endosome or lysosome positive if their fluorescence intensity exceeded the mean background fluorescence by more than 5 s.d. in the endosome/lysosome-containing channel. To assess the colocalization rate, the fraction of endosome- or lysosome-positive pixels among mitochondrion-positive pixels was computed. The proportion of endosome/lysosome-free mitochondria was calculated as 1 minus the colocalization rate. The abundance of endosome/lysosome-free mitochondria was then calculated by multiplying this proportion by the total surface area covered by mitochondria in the selected cell.
Quantification of transplanted mito-GFP accessibility in iHNeurons
Mito-GFP accessibility was evaluated in fixed iHNeurons expressing the anti-GFP nanobody and destabilized nanobodies fused to TagBFP (dGBP1–TagBFP). Mitochondrial accessibility in cells was determined using custom-written routines in Python. The proportion of transplanted mitochondria labelled with GFP (mito-GFP) positive for TagBFP was computed. Images of individual neurons expressing the anti-GFP nanobody and positive for mito-GFP were used for the analysis. Each image contained a channel representing anti-GFP nanobody fluorescence, a second channel representing donor mitochondria fluorescence (mito-GFP) and the last channel representing TagBFP fluorescence (destabilized nanobody). For each neuron, the cell area was annotated as an ROI in ImageJ and used for mito-GFP segmentation. Mito-GFP within a cell were segmented using a custom-trained model in Cellpose94. Segmented objects from the mito-GFP channel were mapped onto the TagBFP channel. Segmented objects of less than 3 pixels were excluded from the analysis. For each segmented object, a 5-pixel-wide ring was used to compute the median background fluorescence (TagBFP). Overlapping segmented objects were removed from the background fluorescence measurement. The ratio between the median TagBFP fluorescence in segmented objects and TagBFP fluorescence in the defined background area was used as a contrast measurement. The segmented objects with a contrast value higher than 1 were considered positive for TagBFP.
Quantification of TagBFP co-localization with mito-GFP in live-imaged iHNeurons
TagBFP colocalization with mito-GFP was evaluated in live iHNeurons expressing the anti-GFP nanobody and destabilized nanobodies fused to TagBFP (dGBP1–TagBFP). Individual neurons transplanted with mito-GFP were imaged at 1 day after transplantation for 2 min at 2 s intervals. Each recording contained a channel representing donor mitochondria fluorescence (mito-GFP) and a channel representing TagBFP fluorescence. TagBFP colocalization with mito-GFP in cells was determined using custom-written routines in Python. Due to a weak signal to noise ratio in the TagBFP channel, a Noise2Void tool was used for image denoising95. A subset of images from a recorded dataset was used for the model training. Training parameters were as follows: 192 × 192 patch size, batch size 128, neighbourhood radius 2 and learning rate 0.001, 120 epochs. A Gaussian filter was applied to all images with TagBFP fluorescence before applying the Noise2Void model. To generate kymographs for each recorded neuron, denoised images with TagBFP fluorescence and corresponding images with GFP fluorescence were projected onto the xT and yT planes. In the generated kymographs, the TagBFP and GFP tracks were annotated using the Labelme tool (http://labelme.csail.mit.edu/). For each annotated TagBFP track, the fraction of TagBFP-positive pixels positive for GFP-positive pixels was computed by assessing the distance of each individual TagBFP-positive pixel to neighbouring pixels from annotated GFP tracks. Pixels with a distance of less than 15 pixels were considered overlapping. To calculate the overlapping fraction in a given TagBFP track, the number of overlapping pixels was divided by the total number of TagBFP-positive pixels. For any given recording, GFP-positive tracks were spatially randomized within the cell area, while the location of BFP tracks was maintained constant. The colocalization value was then computed as before. The randomization process was repeated 100 times for each recording and the final unspecific colocalization value defined as the mean across the 100 iterations.
Tracking of mitochondria and lysosomes
Tracking of donor and native mitochondria in iHNeurons and donor mitochondria and lysosomes in HUVECs was performed manually in live-imaged cells using Trackmate96,97. Median and maximum speeds of generated tracks in Trackmate were used for quantification and illustration.
Quantification of axonal beading in retinal ganglion cells axons after ONC
Axonal beading in retinal ganglion cell axons after optical nerve crush experiments was quantified using a semi-automatic full image segmentation method. This method, based on the Segment Anything Model (SAM)98 in MATLAB (R2025a, MathWorks), involved first labelling the total surface of RGC axons in two-photon mean projection images of ex-vivo retinas with the SAM algorithm. Subsequently, axonal beads were manually labelled based on their distinctive shape and size. The axonal beading ratio for each image was calculated by dividing the total area covered by axonal beads by the sum of the areas covered by axonal beads and labelled axons. For each retina, three to four projection images were independently analysed, and their mean value was reported.
Statistical analyses
All quantification and statistical analyses were performed using Python, MATLAB, ImageJ or GraphPad Prism. The cumulative distributions of two independent groups were compared using the Kolmogorov–Smirnov test. Normality was assessed using the Shapiro–Wilk test for datasets with more than four samples. For unpaired datasets with multiple groups, each group was tested individually for normality; for paired datasets, normality was tested on the differences between pairs. If the null hypothesis of normality was rejected (P < 0.05), nonparametric tests were applied. For unpaired datasets with two independent groups, parametric comparisons were performed using an unpaired two-tailed t-test with Welch’s correction; for nonparametric comparisons, we used the Mann–Whitney U-test. When the sample size was fewer than five, an unpaired two-tailed t-test with Welch’s correction was applied. For unpaired datasets with more than two groups (n > 4) and a single independent variable, ordinary one-way ANOVA (unequal variance) or Welch’s ANOVA (equal variance) was used for parametric analyses, with Bartlett’s test applied to assess homogeneity of variance. The Kruskal–Wallis test was used for nonparametric analyses. For groups with sample size fewer than five, Welch’s ANOVA was used. For datasets with two independent variables, two-way ANOVA was used. For paired datasets, parametric and nonparametric comparisons were performed using the paired t-test and Wilcoxon signed-rank test, respectively. Data are mean ± s.e.m., unless otherwise stated.
For cultured cells in mitochondrial transplantation experiments, each n value refers to the number of wells or dishes of cells used per condition from independent cultures. For induced organoids, it refers to the number of organoids used per condition. For ex vivo and in vivo experiments, each n value refers to the number of explants or retinas used per condition. In some cases, n values refer to the number of human donors used in this study.
Biological materials availability
All unique biological materials used are readily available from the authors.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.