Animals
All animal housing and use procedures were approved by the Institutional Animal Care and Use Committees of Peking University, an AAALAC accredited laboratory animal facility. All mice used in the experiments were bred on the C57BL/6J background. Surf4 fl/fl mice were generated and maintained as previously described27. Tmem41b fl/fl mice (GemPharmatech, T026750) were generated by GemPharmatech on the C57BL/6J background with loxP sites flanking exons 2 and 5, using CRISPR–Cas9 genome-editing technology. Cre-dependent spCas9 knock-in (mice were purchased from the Jackson Lab (026556)29. Clcc1-LKO mice were obtained by intravenous injection of spCas9 mice with AAV8 carrying TBG-cre and Clcc1-targeting sgRNA. Surf4fl/fl mice and Tmem41bfl/fl mice were bred with spCas9 knock-in mice to generate Surf4fl/flspCas9 mice and Tmem41bfl/flspCas9 mice, respectively. A list of the primers for the genotypes is provided in Supplementary Table 1. C57BL/6J mice were provided by Peking University at 6 weeks of age, and ob/ob mice (GemPharmatech, T001461) were purchased from GemPharmatech at 4 weeks of age. Mice were housed under standardized conditions, including a temperature of approximately 22 °C, a 12 h–12 h light–dark cycle and a humidity of 40–60%. Mice had free access to food and water unless otherwise stated. Male mice aged 6–16 weeks were used in all experiments. Mice were randomly assigned to different experimental groups. For fasting studies, all experiments were performed at 09:00 on the second day, with fasting starting at 17:00 on the first day. Liver samples of male macaque monkeys were acquired from the AAALAC accredited Nonhuman Primate Research Center, Peking University.
Cell culture
HEK293T cells (ATCC, CRL-3216), Huh7 cells (JCRB Cell Bank, JCRB0403) and HEK293F cells (Thermo Fisher Scientific, R79007) were obtained from ATCC, JCRB and Thermo Fisher Scientific, respectively. HEK293F suspension cells were cultured in SMM 293T-I medium (Sino Biological, M293T1), supplemented with 0.5% penicillin–streptomycin (Caisson, PSL01) and 1% FBS (VisTech, SE100-011) and maintained at 37 °C in a 5% CO2 environment, with an optimal spinner speed of approximately 100 rpm to 130 rpm. The other cell lines were cultured in Dulbecco’s modified Eagle’s medium (DMEM) (HyClone, SH30022.01B) supplemented with 1% penicillin–streptomycin and 10% FBS, under the same temperature and CO2 conditions. Transfections were performed with polyethyleneimine (PEI) (Polysciences, 23966-1) according to the manufacturer’s protocol.
DNA vector construction
The sgRNAs were designed using the Benchling platform (https://benchling.com/) to optimize editing efficiency and minimize the unintended off-target effects. The sgRNA sequences are listed in the Supplementary Table 2. Oligonucleotides were cloned into either the pX602-AAV-Cre sgRNA backbone for CRISPR–Cas9-induced acute gene knockout in mouse liver or the pLentiCRISPR V2 (Addgene, 52961) and pLentiGuide Blast for genome editing in cell lines, according to established protocols described previously48. The pX602-AAV-Cre sgRNA construct was derived from the pX602 vector (Addgene, 61593) in our previous study49. The pLentiGuide Blast construct was derived from the pLentiGuide Puro vector (Addgene, 52963), in which blasticidin S deaminase was cloned between the BsiWI and MluI restriction sites to replace puromycin N-acetyltransferase.
pAAV-TBG-mCLCC1-StrepTagII-Flag-StrepTagII was generated from pAAV-TBG-EGFP (Addgene, 105535) by replacing the GFP sequence with the mouse Clcc1 cDNA. For rescue experiments, the PAM region of mouse Clcc1 cDNA was synonymously mutated as annotated in Extended Data Fig. 3c.
For fluorescence imaging, mEGFP–PLIN2, mEGFP–CB5 (residues 90 to 124 at the C-terminal end of cytochrome b5 type A, CYB5A), mEGFP–Sec61β, IgH signal peptide–mEGFP–KDEL, APOE–mEGFP, ApoE–GFP1-10 and GFP11–mCLCC1 (GFP11 was inserted into the N terminus of mCLCC1, followed by the signal peptide) were cloned between the KpnI and HindIII restriction sites to substitute for enhanced GFP (eGFP) in the pAAV-CAG-EGFP vector (Addgene, 51502).
For protein purification, CLCC1–Flag, twin StrepTagII–TMEM41B and Flag–TMEM41B were cloned into pKH3 (Addgene, 12555) between the EcoRI and XbaI restriction sites.
Recombinant AAV production and delivery
AAV packaging and purification were performed as previously described49. For the mouse experiment, AAV shuttle plasmids, Rep/Cap (2/8) plasmids and helper plasmids were transfected into HEK293T cells using PEI. At 60 h after transfection, cells were collected and virus was purified, quantified by both Coomassie Blue R250 staining and quantitative PCR (qPCR). AAV-2/8 was administered by tail vein injection. To achieve acute inactivation of the hepatic Clcc1 gene in 6-week-old spCas9 knock-in mice, each mouse was administered with pX602-AAV-Cre sgRNA at a viral genome copy number of 4 × 1011. For rescue experiments, each mouse was simultaneously injected with 4 × 1011 viral genome copies of pX602-AAV-Cre-sgRNA and 5 × 1010 viral genome copies of AAV-TBG-mCLCC1-StrepTagII-Flag-StrepTagII.
For AAV delivery into a cultured cell line, pRep/Cap (2/8) was replaced by pAAV-DJ for AAV-DJ preparation. AAV-DJ with 1 × 1010 genome copies number was used per well in six-well plate to delivery indicated gene into cultured cells. Subsequent experiments were conducted 24 h after AAV-DJ infection, unless otherwise specified.
Lentiviral packaging and knockout cell construction
HEK293T cells were used for lentivirus packaging. In brief, the lentivirus shuttle plasmids, psPAX2 (Addgene, 12260) and pMD2.G (Addgene, 12259) plasmids were introduced into the cells using PEI according to the protocol provided by the manufacturer. Then, 48 h after transfection, the medium containing the lentivirus was collected and subsequently added to the wild-type Huh7 cells. Transduced cells were selected by antibiotic at 72 h after infection.
For control, TMEM41B-KO and CLCC1-KO Huh7 cell construction, pLentiCRISPR V2 lentivirus containing LacZ-targeting sgRNA, human TMEM41B targeting sgRNA and human CLCC1 targeting sgRNA (Supplementary Table 2) were generated and infected into Huh7 cells, respectively. Then, 2 days after transfection, cells were selected with puromycin (Sigma-Aldrich, P8833). For TMEM41B and CLCC1 double-deficient Huh7 cells, TMEM41B-KO Huh7 cells were infected with pLentiGuide Blast lentivirus containing control or human CLCC1 targeting sgRNA, and further selected with blasticidin (Wako, 022-18713).
Plasma characterization and FPLC analysis
Blood samples were collected from the tail tips of mice fasted for 16 h using a heparinized capillary. Plasma was separated by centrifugation at 6,000 rpm for 10 min at 4 °C. Triglyceride, total cholesterol, ALT/GPT and AST/GOT levels were determined using specific commercial kits (Sigma-Aldrich, TR0100; 000180, 1000000010 and 1000000020 from Zhongsheng Beikong, respectively) according to the manufacturer’s instructions. Absorbance values were measured using a Thermo Fisher Scientific Varioskan LUX multimode microplate reader controlled by Thermo Fisher Scientific SkanIt Software 3.3 for Microplate Readers. For fast-protein liquid chromatography (FPLC) analysis, pooled plasma samples from the same treatment group were fractionated using Superose 6 increase columns. The fractions were collected at a flow rate of 0.5 ml min−1 for subsequent measurements of cholesterol and triglyceride levels.
VLDL secretion assay
Tyloxapol (Sigma-Aldrich, T8761) was administered to mice at a dose of 50 mg per kg body weight after a 16 h fast. Blood samples were collected at 1, 2 and 4 h after injection, and plasma triglyceride levels were analysed as previously described.
Histology
Tissue samples were collected and preserved in 4% paraformaldehyde (PFA) in PBS (Leagene, DF0135). Tissue embedding, sectioning and H&E staining were performed by the Pathology Center of Peking University or Beijing ZKWB-Bio Biotechnology. For Oil Red O staining, tissues were embedded in OCT compound and rapidly frozen. Cryosections at a thickness of 8 μm were cut and stained with Oil Red O according to the manufacturer’s instructions. Immunohistochemistry was performed on paraffin-embedded liver sections. Sections were deparaffinized and rehydrated, followed by antigen retrieval. To prevent non-specific binding, the sections were blocked with 10% goat serum for 1 h and then incubated overnight at 4 °C with primary antibodies diluted in blocking buffer. The sections were then exposed to secondary antibodies for 2 h at room temperature. Finally, they were visualized by DAB (3,3′-diaminobenzidine) staining.
Histological quantifications were performed according to the standard NAFLD activity scoring and fibrosis scoring system (https://tpis.upmc.com/), in a double-blinded manner. In brief, the histological features of metabolic-dysfunction-associated steatotic liver disease (MAFLD) and MASH were grouped into four broad categories: steatosis, lobular inflammation, hepatocyte ballooning and fibrosis. Oil Red O-stained histology images were used to score steatosis, H&E-stained histology images were used to score lobular inflammation and hepatocyte ballooning, and Masson-trichrome-stained histology images were used to score fibrosis. The MASH score represents the sum of scores for steatosis, lobular inflammation and hepatocyte ballooning.
Quantification of hepatic triglycerides, cholesterol and cholesterol esters
Liver samples were quantified and homogenized in PBS. Lipids were extracted from the homogenates according to the established protocols of the modified Bligh–Dyer method. In brief, the homogenates were vigorously mixed with a chloroform–methanol mixture (2:1). After centrifugation, the organic phase was carefully collected and concentrated using a rotary evaporator under vacuum. The lipid extract obtained was reconstituted in a solution of 15% Triton X-100 (Sigma-Aldrich) in double-distilled H2O. Triglycerides levels were measured using commercially available kits (Zhongsheng Beikong, 100000220, for triglycerides and free glycerol; Sigma-Aldrich, F6428, for free glycerol) according to the manufacturers’ protocols. The total cholesterol levels were assessed using a commercially available kit (Zhongsheng Beikong, 100000180) according to the manufacturer’s instructions. Quantification of cholesteryl esters was measured using the Amplex Red Cholesterol and Cholesteryl Ester Test Kit (Beyotime Biotechnology, S0211M).
LD isolation and identification
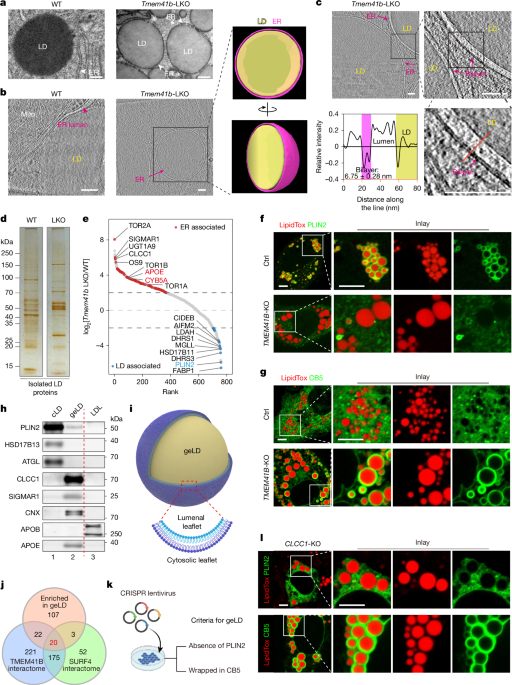
The procedure of LD isolation was modified from a previously published protocol50. Before LD dissection, mice were fasted for 4 h. After anaesthesia, the liver was perfused through the portal vein to remove blood. The liver was cut into approximately 1 mm3 pieces in buffer A (25 mM tricine pH 7.6, 250 mM sucrose) plus 0.2 mM PMSF and protease inhibitors (Roche, 4693132001) and homogenized ten times on ice with a Dounce type glass-Teflon homogenizer. The lysate was centrifuged twice at 500g for 10 min at 4 °C to remove debris. The postnuclear supernatant (PNS, 10 ml) was then transferred to a SW40 Ti centrifuge tube, and 2 ml buffer B (20 mM HEPES pH 7.4, 100 mM KCl and 2 mM MgCl2) was added to the PNS. The tubes were loaded into a Beckman SW40 rotor and centrifuged at 300,000g for 2 h at 4 °C. After the ultracentrifugation step, the white band of LD floating on the top of the solution was carefully collected into a new SW40 Ti centrifuge tube filled with 12 ml buffer B and centrifuged at 300,000g for 30 min to remove contaminating organelles. This was repeated for three times to wash the isolated LD until no more pellets could be observed. The size of purified lipid droplets was analysed using dynamic light scattering (DLS). For estimating the membrane integrity, the isolated LD samples were treated with 0.005% trypsin in the absence or presence of 1% Triton X-100 for 30 min. For isolation of ER membrane-encapsulated geLDs, purified geLDs were treated with a hypotonic solution (1 mM Tris-HCl, pH 8.8) for 30 min on ice, to induce osmotic shock and rupture the surrounding ER membrane. Subsequent centrifugation at 100,000g for 1 h at 4 °C was performed to obtain the top fraction as the pure droplets (D), the supernatant fraction as interspace and/or peripheral-associated contents (I) and the pellet fraction as membranes (M).
LD proteins were precipitated with acetone at −80 °C overnight, centrifuged at 15,000g for 10 min, washed with ice-cold acetone, dried with the lid open and then solubilized in RIPA buffer (50 mM Tris-HCl pH 7.4, 150 mM NaCl, 1% Nonidet P-40, 1% sodium deoxycholate, 0.1% SDS, 1 mM EDTA, 10% glycerol with protease inhibitor) supplemented with 2% SDS. Protein concentrations were measured using the BCA Protein Assay Kit (Thermo-Pierce, 23227). Silver staining of LD proteins was performed using a commercial kit according to the manufacturer’s protocol (Thermo Fisher Scientific, 24600). Proteomics analysis was performed on the Thermo Fisher Scientific Q-Exactive HF-X mass spectrometer, and raw data were processed using Proteome Discoverer software v.2.5 (Thermo Fisher Scientific), according to a previously published procedure49. The proteins with more than one unique peptide were further analysed. The criterion for identifying differentially expressed proteins was a fold change greater than 10. Enrichment analysis of upregulated or downregulated different expressed proteins was performed separately using Cluster Profiler R package (v.4.10.0). Venn analysis was performed using the VennDiagram R package (v.1.7.3). The significant GO terms of the cellular component were visualized using the ggplot2 R package (v.3.4.4).
Isolation of subcellular organelles
Mice were fasted for 16 h overnight. After anaesthesia, the liver was perfused and homogenized as described in the ‘LD isolation and identification’ section. The lysates were centrifuged twice at 3,000g for 10 min at 4 °C to remove debris and nuclei (PNS). For analysis of APOB distribution in the early secretory pathway, an eleven-step OptiPrep gradient of 10–30% (top to bottom, 2.5% increments) was used in a SW40 Ti centrifuge tube, as a previously decribed51. The PNS (1 ml, 1 mg ml−1) were then transferred to the top of the tube, followed by centrifugation at 100,000g for 4 h at 4 °C using a Beckman SW40 rotor. After ultracentrifugation, fractions (0.5 ml) were collected from top to bottom and immunoblotted for Golgi markers and ER markers. Fractions enriched with the Golgi or the ER were collected for further immunoblot analysis. For analysis of the lipid and protein composition of the ER, PNS were subjected to sequential centrifugation at 9,000g for 40 min, and 21,000g for 60 min. The 21,000g pellets were designated as the ER fractions for further analysis.
Immunoblot analysis
Liver lysates were prepared using RIPA buffer supplemented with protease inhibitors. Protein concentrations were measured using the BCA Protein Assay Kit. 1× SDS–PAGE sample buffer was added. Equal amounts of protein were loaded onto 3% to 15% Tris-acetate SDS–PAGE gels and transferred overnight at 4 °C to nitrocellulose membranes (Cytiva, 10600006). The membranes were then blocked with 5% milk in TBST (20 mM Tris pH 7.4, 150 mM NaCl and 0.1% Tween-20) for 1 h at room temperature. Primary antibodies were diluted in TBST containing 5% milk and 0.02% Na3N, incubated with membranes overnight at 4 °C and washed three times with TBST at room temperature, for 15 min each time. HRP-conjugated goat secondary antibodies were diluted in TBST containing 5% milk and incubated for 1 h at room temperature, followed by three washes. Membranes were visualized after exposure to ECL substrate (Thermo Fisher Scientific, 34578) using Amersham Imager 600/800 systems (Cytiva) and western blot images were acquired and analysed using Amersham Imager 600/800 Analysis Software (v.3.0, Cytiva).
The following primary antibodies were used for IB: rabbit polyclonal anti-PLIN2 (Cell Signaling Technology, 45535, 1:1,000), rabbit polyclonal anti-HSD17B13 (ABclonal, A6256, 1:1,000), rabbit polyclonal anti-ATGL (Cell Signaling Technology, 2138, 1:1,000), rabbit polyclonal anti-SigmaR1 (Proteintech, 15168-1-AP, 1:1,000), rabbit polyclonal anti-calnexin (Proteintech, 10427-2-AP, 1:1,000), rabbit polyclonal anti-APOB (Proteintech, 20578-1-AP, 1:1,000), rabbit polyclonal anti-ApoE (Fitzgerald, 10R-10633, 1:1,000), mouse monoclonal anti APOE (Cell Signaling technology, 74417, 1:1,000), rabbit polyclonal anti-APOA1 (Fitzgerald, 70R-15769, 1:1,000), rabbit polyclonal anti-LIPIN1 (Proteintech, 27026-1-AP, 1:1,000), rabbit polyclonal anti-FASN (Proteintech, 10624-2-AP, 1:1,000), rabbit polyclonal anti-ACC1 (Proteintech, 21923-1-AP, 1:1,000), mouse monoclonal anti-MTP (Santa Cruz Biotechnology, sc-135994, 1:1,000), mouse monoclonal anti-PDI (BD transduction Laboratories, 610946, 1:1,000), rabbit polyclonal anti-BIP (Proteintech, 11587-1-AP, 1:1,000), rabbit polyclonal anti-tubulin (Proteintech, 11224-1-AP, 1:1,000), rabbit polyclonal anti-torsin A/DYT1 (Abcam, ab34540, 1:1,000), rabbit polyclonal anti-CES3 (Proteintech, 14587-1-AP, 1:1,000), rabbit polyclonal anti-REEP5 (Proteintech, 14643-1-AP, 1:1,000), mouse monoclonal anti-Flag (M2) (Sigma-Aldrich, F1804, 1:500). rabbit anti-GM130 antibody (Proteintech, 11308-1-AP, 1:1,000), rabbit polyclonal antibody against the N-terminal epitope of human TMEM41B (residues 1–109) were produced by the Proteintech Group (www.ptglab.com), the rabbit anti-serum was collected and purified by NHS beads conjugated with His-tagged human TMEM41B (residues 1–109), followed by washing with PBS containing 0.15% Triton X-100, eluted with 50 mM glycine (pH 2.5) and neutralized with Tris-HCl pH 8.0. Rabbit polyclonal antibodies against the C-terminal epitope of mouse CLCC1 (residues 355–539) was a gift from Y. Jia.
The following secondary antibodies were used: goat anti-mouse IgG (H+L) secondary antibody (Thermo Fisher Scientific, 31430, 1:10,000) and goat anti-rabbit IgG (H+L) secondary antibody (Thermo Fisher Scientific, 31460, 1:10,000).
Blue Native PAGE
Blue native PAGE analysis of CLCC1 was performed according to a protocol described previously52. In brief, livers from mice of the indicated genotype were disrupted in buffer C (25 mM Tris pH 7.4, 250 mM sucrose, 1 mM EDTA and protease inhibitors) and homogenized ten times on ice using a Dounce type glass-Teflon homogenizer. The lysates were centrifuged at 1,000g for 10 min at 4 °C to remove nuclei and unbroken cells. The supernatants were further centrifuged at 100,000g for 30 min to isolate membrane fractions. The membranes from the indicated mice and geLDs from Tmem41b-LKO mice were incubated with buffer D (25 mM Tris pH 7.4, 50 mM NaCl, 1% n-dodecyl-β-d-maltoside (DDM, Qisong biological, QS81007015) and protease) for 2 h at 4 °C, followed by centrifugation at 100,000g for 30 min. The concentrations of the supernatants were measured using the BCA Protein Assay Kit. 5% Coomassie Brilliant Blue G-250 dye was added at a final dye to detergent ratio of 1/8 (g/g). Equal amounts of protein were loaded on 4–16% blue native PAGE gel, transferred overnight at 4 °C to PVDF membranes (Cytiva, 10600023), followed by immunoblot analysis.
Immunoprecipitation analysis
For immunoprecipitation of endogenous TMEM41B, cells were washed twice with ice-cold PBS and incubated with lysis buffer (50 mM Tris-HCl pH 7.4, 150 mM NaCl, 1% MNG, 1 mM EDTA, 10% glycerol with protease inhibitor) for 20 min on ice. Liver samples from Rhesus monkeys were homogenized in buffer C (25 mM Tris pH 7.4, 250 mM sucrose, 1 mM EDTA and protease inhibitors) on ice using a Dounce type glass-Teflon homogenizer, followed by centrifugation at 1,000g for 10 min at 4 °C to remove nuclei and unbroken cells. The supernatants were further centrifuged at 100,000g for 30 min to isolate membrane fractions. The membrane fractions were lysed in buffer (50 mM Tris-HCl pH 7.4, 150 mM NaCl, 1% MNG, 1 mM EDTA, 10% glycerol with protease inhibitor) for 60 min on ice, followed by centrifugation at 100,000g for 30 min at 4 °C to collect the supernatants. Supernatant concentrations were adjusted based on measurements taken with the BCA Protein Assay Kit. Subsequently, the supernatants were incubated with anti-TMEM41B antibodies with end-over-end rotation for 2 h at 4 °C, followed by incubation with rProtein A beads 4FF (Smart-life sciences, SA015025), which were continuously rotated for 4 h at 4 °C. The beads were washed five times with wash buffer (50 mM Tris-HCl pH 7.4, 150 mM NaCl, 1% MNG and 10% glycerol). For immunoblot analysis, proteins were further eluted from the beads with elution buffer (50 mM Tris-HCl pH 7.4, 150 mM NaCl, 1% MNG, 1 mM EDTA, 10% glycerol, protease inhibitor with 0.4 mg ml−1 TMEM41B (residues 1–51) peptides). After gel separation, proteins were transferred to NC membrane for immunoblotting.
Metabolic labelling of nascently synthesized APOB
Mouse primary hepatocytes were isolated from male mice liver of the indicated mouse genotypes, and cultured in collagen-coated 10 cm culture plates in high-glucose DMEM containing 10% FBS and 1% penicillin–streptomycin. Cell were washed twice with PBS, and the medium was then switched to methionine-free DMEM (Thermo Fisher Scientific, 21013-024), and cultured for an additional hour. To start the metabolic labelling, cells were next cultured in methionine-free DMEM containing 1 mM l-azidohomoalanine hydrochloride (l-AHA) (MedChemExpress, 942518-29-8) for 30 min. The chase period was initiated by washing cells twice with PBS before being cultured in high-glucose DMEM for subsequent analysis. At 10, 60 and 120 min, 20% NP-40 was added to the medium to reach a final concentration of 1%. APOB IP was performed using a rabbit anti-mouse APOB antibody (Meridian, K23300R). The immunoprecipitated APOB proteins were then incubated with 20 μM alkyne-biotin (MedChemExpress, HY-138749), 200 μM TBTA (MedChemExpress, HY-116677), 1 mM CuSO4 and 1 mM TCEP (Thermo Fisher Scientific, 20491) in PBS at room temperature for 3 h for labelling biotin to l-AHA-pulsed APOB through the click reaction. The samples were subsequently used for SDS–PAGE analysis to evaluate the remaining newly synthesized APOB by detecting biotin through HRP-conjugated streptavidin (Proteintech, SA00001-0, 1:2,000).
Protein expression and purification
HEK293F cells were transfected with CLCC1-Flag, Flag-TMEM41B or CLCC1-Flag + twin StrepTagII-TMEM41B plasmids. Cells were grown for 48 h, and collected by centrifugation at 1,000g for 10 min at 4 °C, then washed with ice-cold TBS buffer. Cell pellets were resuspended in buffer E (50 mM Tris-HCl pH 7.4, 150 mM NaCl, 1% DDM, 1 mM EDTA, 10% glycerol with protease inhibitor). After incubation for 2 h, the cell lysates was centrifuged at 100,000g for 30 min, and the supernatant was incubated with prewashed anti-DYDDDDK affinity beads (Smart-lifesciences, SA042500) for 2 h at 4 °C, the beads were washed with buffer E (50 mM Tris-HCl pH 7.4, 150 mM NaCl, 0.025% DDM and 10% glycerol). The protein was eluted with buffer E containing 0.4 mg ml−1 Flag peptides. For tandem purification of the CLCC1–TMEM41B complex, the flag-peptide-eluted products were further incubated with Streptactin beads 4FF (Smart-lifesciences, SA053500) for 2 h at 4 °C, and the slurry was washed and eluted with buffer E containing 10 mM desthiobiotin (Sigma-Aldrich, 71610-M). Proteins were loaded onto Amicon 0.5 ml concentrators (10 kDa cut-offs, SEP, UFC501008), concentrated, buffer replaced with buffer E and quantified by Coomassie Blue R250 staining using BSA standards.
Immunofluorescence and confocal microscopy
Cells were plated into 35 mm glass-bottom dishes (Cellvis, 150680) for subsequent treatment and staining. For immunofluorescence, cells were fixed with 4% PFA in PBS for 10 min at room temperature. After fixation, cells were washed three times with PBS, then permeabilized with immunostaining permeabilization buffer with Saponin (Beyotime, P0095) for 20 min and washed three times with PBS. After blocking with 2% BSA in PBS for 1 h, the cells were incubated with primary antibodies in blocking solution overnight at 4 °C. The cells were then washed three times with PBS. The cells were then incubated with fluorescent-labelled secondary antibodies for 1 h at room temperature and washed three times with PBS. For staining, lipid droplets were visualized with HCS LipidTox Red (Thermo Fisher Scientific, H34476, 1:1,000) or HCS LipidTox Deep Red (Thermo Fisher Scientific, H34477, 1:1,000) for 20 min, and immersed in PBS or complete cell culture medium for imaging. For plasma membrane (PM) staining, PM were visualized with WGA–Alexa Fluor 488 (Thermo Fisher Scientific, W11261, 2 μg ml−1) for 10 min, and immersed in phenol-red-free DMEM (Gibco, 21063029) supplemented with 10% FBS for imaging.
The following primary antibodies were used for immunofluorescence: mouse monoclonal anti-Flag (M2) (Sigma-Aldrich, F1804, 1:500); rabbit polyclonal anti-PLIN2 (Proteintech, 15294-1-AP, 1:200); mouse monoclonal anti ApoE (Cell Signaling Technology, 74417, 1:800); rabbit polyclonal anti-SEC31A (Proteintech, 17913-1-AP, 1:300); rabbit polyclonal anti-ARF1 (Proteintech, 10790-1-AP, 1:300), rabbit polyclonal antibody against the C-terminal epitope of human CLCC1 (residues 355–539, 1:500; a gift from Y. Jia); rabbit polyclonal anti-TMEM41B (1:20), purified from rabbit serum against TMEM41B (residues 1–109) using the N-terminal 1–20 amino acid peptide; goat anti-rabbit IgG (H+L) cross-adsorbed secondary antibody, Alexa Fluor 488 (Thermo Fisher Scientific, A-11008, 1:1,000); goat anti-rabbit IgG (H+L) cross-adsorbed secondary antibody, Alexa Fluor 568 (Thermo Fisher Scientific, A-11011, 1:1,000); goat anti-rabbit IgG (H+L) cross-adsorbed secondary antibody, Alexa Fluor 568 (Thermo Fisher Scientific, A-11031, 1:1,000). Images were acquired on the ZEISS 900 confocal microscope with Airyscan2 (Carl Zeiss) controlled by ZEISS ZEN v.3.2 (blue edition) and processed with Fiji (ImageJ distribution, v.2.1.0; based on ImageJ, NIH; https://imagej.net/software/fiji/).
Image analysis
To analyse the TMEM41B signal enriched around LDs, images were imported and processed using Fiji (ImageJ distribution, v.2.1.0; based on ImageJ, NIH; https://imagej.net/software/fiji/). LD regions of interest (ROIs) were detected using ‘Threshold-Analyse particle’ and the mean intensity of TMEM41B at LD ROIs was measured. The mean intensity of TMEM41B at five cytosol ROIs was then randomly measured. The ratio of mean intensity of LD ROIs to cytosol ROIs was defined as TMEM41B enrichment around LDs.
To quantify the percentages of LDs with PLIN2 or CB5 signals, images were imported into and processed using Fiji (ImageJ distribution, v.2.1.0; based on ImageJ, NIH; https://imagej.net/software/fiji/). LDs with PLIN2 or CB5 were detected and collected using ‘Threshold-Analyse particles (include holes)’ using the corresponding channel. Total LDs were detected using ‘Threshold-Analyse particles’ using the LD channel. The LD number or area with PLIN2 or CB5 signal divided by the total LD number or area is shown as the percentage of LDs with PLIN2 or CB5 signal.
Association analyses of SNPs and plasma lipid levels
GWAS summary statistics of the CLCC1 gene and plasma lipid levels were obtained from the GLGC dataset28 and performed using LocusZoom (http://locuszoom.org/). The allele frequency in humans of the rs149700491 single-nucleotide polymorphism (SNP) was obtained from 1000 Genome Phase 3.
In vitro lipid scrambling assay and NBD-glucose leakiness assay
POPC (Sigma-Aldrich, 42773), POPG (Sigma-Aldrich, 840457P) and NBD-PC (Sigma-Aldrich, 810144P-1MG) were solubilized in chloroform, at a molecular ratio 180:19:1, the lipids were dried under an argon or nitrogen stream and the flask was transfected to a vacuum desiccator at room temperature for at least 3 h. The dried lipid film was reconstituted in buffer F (50 mM HEPES pH 7.4, 200 mM NaCl,) at a concentration of 5.25 mM lipid. This lipid solution was incubated in a water bath at 37 °C for 10 min, processed for ten freeze–thaw cycles and extruded 30 times through a 400 nm polycarbonate filter (Avanti, 610020). Liposomes were destabilized with 7 mM DDM at room temperature for 2 h. The following samples were added: (1) detergent buffer control; (2) detergent-solubilized protein preparations containing 50 nM (low PPR) or 500 nM (high PPR) TMEM41B; or (3) 50 nM TMEM41B in a complex with CLCC1. Individually purified CLCC1 comparable to its concentration in the CLCC1–TMEM41B complex was used. After incubation for 1 h, Bio-beads (BioRad, 1528920) were used to remove detergent according to the manufacturer’s protocol, followed by 11 extrusions through a 200 nm Avanti polycarbonate filter to minimize multilamellar. The PPR in the 50 nM TMEM41B case was approximately 1:100,000 (~3–5 proteins per vesicle on average). Examination of protein incorporation was performed using a liposome flotation assay, followed by SDS–PAGE analysis of liposome-associated proteins. The scramblase assay was performed in a cuvette equipped with a magnetic stirrer at room temperature, with a 2 ml reaction volume (diluted 20 times, with proteoliposomes containing around 250 μM lipid and 2.5 nM TMEM41B in the case of low PPR). Fluorescence signals were monitored (excitation at 460 nm, emission at 538 nm) using the Synergy H1 Hybrid Multi-Mode Reader (BioTek) controlled by Gen5 software (v.3.10, BioTek) at 1 s intervals. To assess lipid scrambling, NBD fluorescence was monitored over time after the addition of dithionite (Sigma-Aldrich, 71699) to a final concentration of 10 mM. Subsequently, additional Triton X-100 (0.5%) was then added to disrupt the liposomes, allowing for a thorough reduction of all NBD fluorescence.
The NBD-glucose leakage assay was performed in a manner similar to the lipid scramblase assay described above, except that NBD-PC was omitted from the liposome composition. Instead, NBD-glucose (Abcam, ab146200, 20 μM) was added to the buffer during the destabilization step. Subsequently, the liposomes were dialyzed to remove unincorporated NBD-glucose, and were then subjected to the leakage assay.
Click labelling with alkyne-choline
CRISPR–Cas9-mediated control, TMEM41B-KO, CLCC1-KO and TMEM41B/CLCC1-DKO Huh7 cells were seeded onto glass coverslips in six-well plates at 25% density, and cultured with 100 μM alkyne-choline (CONFLUORE, BCP-44) for 18 h. Cells were fixed with 4% PFA in PBS for 10 min at room temperature and washed three times with PBS. The cells were then permeabilized with 25 μg ml−1 digitonin for 15 min and washed three times with PBS. Cells were incubated with 10 μM 5-TAMRA azide (Confluore, BDR-22), BTTAA (Confluore, BDJ-4) CuSO4 complex (50 μM CuSO4, BTTAA/CuSO4 6:1, mol/mol) and 2.5 mM sodium ascorbate (Aladdin, S105024) in PBS at room temperature for 1 h, and washed three times with PBS. After the click reaction, the coverslips were mounted onto glass slides and examined under the ZEISS 900 confocal microscope with the Airyscan2 module (Carl Zeiss). The fluorescence intensity was quantified using ImageJ Fiji (NIH) and analysed using Prism (GraphPad).
For alkyne choline uptake analysis, cells in 10 cm dishes at 80–90% density were incubated with 100 μM alkyne-choline for 10 min. Cellular alkyne-choline was extracted by methanol. For total alkyne PC analysis, cells in 10 cm dishes at 80–90% density were incubated with 100 μM alkyne-choline for 12 h. Lipids were extracted by chloroform:methanol (2:1) as previously described11. The extracts were dried under a stream of nitrogen gas and dissolved in organic solvents, followed by a CuBF4-catalysed click reaction with 3-azido-7-hydroxycoumarin (Jena Bioscience, CLK-FA047-1) in ethanol at 55 °C for 4 h, as previously described53. Then, 30 μl of this mixture was added to the lipids and incubated at 55 °C for 4 h. After centrifugation, the mixture was loaded onto 20 cm silica TLC plates and developed in chloroform:methanol:water:acetic acid (65:25:4:1) for 4–5 cm. The plates were treated with 4% N,N-diisopropylethylamine (Sigma-Aldrich, D125806) in hexanes, dried and imaged for fluorescence (excitation, 460–490 nm; emission, 518–546 nm) using ChemiDoc MP imager (BioRad), with analysis performed in Fiji ImageJ.
EM samples preparation and images
Mice were euthanized and perfused systematically with 0.1 M sodium phosphate buffer (pH 7.4), followed by a pre-fixation solution containing 2.5% glutaraldehyde and 0.8% PFA. The liver tissues were then surgically excised, fixed in the pre-fixation solution for 2 h at room temperature and further dissected into smaller sections (0.2 × 0.3 × 0.5 mm3). These samples underwent an additional overnight fixation at 4 °C in the same pre-fixing solution. After rinsing in phosphate buffer, the tissues were immersed in 0.1 M imidazole in 0.1 M sodium phosphate buffer for 30 min and then post-fixed in 2% osmium tetroxide in 0.1 M sodium phosphate buffer. After thorough rinsing with ultrapure water, the tissues were stained with 1% uranyl acetate at 4 °C overnight. The samples were dehydrated through a gradient acetone series, then embedded in epoxy resin and polymerized at 60 °C for 24 h. The prepared samples were sectioned at a thickness of approximately 60 nm using the Leica EM UC7 system, placed onto copper grids and imaged using the FEI Tecnai G2 20 Twin electron microscope equipped with a CMOS camera (EMSIS) controlled by Radius satellite software v.2.7. Examination of liver tissue structure was performed using a double-blinded method.
Cryo-ET sample preparation
High-pressure freezing
The mouse liver samples were vitrified using a previous established protocol17, with some modifications. In brief, the abdomen of each mouse was dissected under anaesthesia. A small piece of liver tissue was meticulously excised using a scalpel and placed onto an EM grid (Beijing XXBR, Cu microgrid, T10012, 200 mesh). The grid was coated with an additional carbon layer (around 10 nm thick) and freshly glow-discharged using a Model 950 Advanced Plasma System (Gatan) before use. The grid was previously placed into a 6 mm aluminium carrier (200 nm in depth). After filling the carrier with 2-methylpentane (Sigma-Aldrich, M65807), a 6 mm sapphire disc was immediately placed on top of the carrier. The assembly of the sandwich carrier was promptly inserted into the high-pressure cryostat (Leica EM ICE) for vitrification. The vitrified sandwich assembly was then transferred to a liquid ethane and propane mixture (ethane:propane = 36.9%:63.1%) at −170 °C, allowing 2-methylpentane to dissolve. The resulting grid was then transferred to liquid nitrogen for storage until milling.
The cryo-lift-out and lamella thinning
A cryo-FIB/SEM (Aquilos 2, Thermo Fisher Scientific) was used to prepare lamella, using an adapted serial lift-out method18,54. In brief, the grid was clipped into an Autogrid (Thermo Fisher Scientific) before being loaded into the FIB chamber through the sample transfer rod. To minimize ion-beam damage and enhance the electrical conductivity, the sample was coated with a layer of Pt using a gas-injection system (GIS) for 2 min, followed by sputtering at 30 mA for 15 s. Next, four trenches were milled around the region of interest with the ion beam to expose the sample block. This block was subsequently attached to the cryo-needle and transferred to the receiver grid (copper, 100/400 mesh, Beijing XXBR, G100/400), where it was serially sectioned to several lamellae. These lamellae were securely attached to the grid in turn by redeposition method. Finally, each lamella was finely milled to a target thickness of approximal 200 nm, which was then used for subsequent tomographic data acquisition.
Cryo-ET data acquisition
Lamellae from wild-type mice were loaded onto the 300 kV cryo-transmission electron microscope (Titan Krios G3, Thermo Fisher Scientific) equipped with a K3 camera and a Gatan energy filter. Automatic tomographic tilt series acquisition was performed using SerialEM software55. Images were acquired at a magnification of ×64,000 (pixel size, 1.37 Å) in TIFF format using super-resolution mode, resulting in ten frames per image. The defocus was set from −4.0 to −6.0 μm. The acquisition was performed from −50° to +50° (with respect to the pre-tilt angle) in 2° increments, using the dose symmetric scheme56. The dose rate was set to 13 e− px−1 s−1 and total dose was limited to 120 e− Å−2 per tilt series and the total dose was limited to 120 e− Å−2 per tilt series.
Lamellae from Tmem41b-LKO mice were loaded onto the 300 kV cryo-transmission electron microscope (Titan Krios G4, Thermo Fisher Scientific) with a Falcon 4i camera and a Thermo Fisher Selectris X energy filter. Automatic tomographic tilt-series acquisition was performed using Tomography v.5.12.0 (Thermo Fisher Scientific). Images were acquired at a magnification of ×42,000 (pixel size, 3.00 Å) in EER format. The defocus was set from −2.5 to −5.0 μm. The acquisition was performed from −50° to +50° (with respect to the pre-tilt angle) in 2° increments, using the dose symmetric scheme56. The dose rate was set to 8 e− px−1 s−1.
Correlative cryo-light microscopy and cryo-EM
The ERs in Huh7 cells were labelled by stably transfecting meGFP–CB5, while LDs were stained using LipidTox Red. Cells were cultured on gold grids (Quantifoil R2/1, 200 mesh) at 37 °C in a 5% CO2 environment before vitrification. These grids were precoated with an additional carbon layer (~10 nm thick) and glow-discharged for 60 s using the Model 950 Advanced Plasma System (GATAN) before cell seeding.
Vitrification was performed when the cell confluency reached approximately 50%. The culture medium was replaced with a medium containing 10% glycerol before being plunge-frozen into a mixture of liquid ethane and propane (ethane:propane = 36.9%:63.1%) using Vitrobot Mark IV (Thermo Scientific) at a chamber temperature of 37°C. The vitrified grids were stored in liquid nitrogen until cryo-CLEM imaging and cryo-FIB milling.
Grids were clipped into Autogrids (Thermo Fisher Scientific) before being loaded onto the cryo-stage of the Thunder Imager EM Cryo-CLEM (Leica) equipped with a ×50/0.9 NA objective. To locate ERs and LDs, overview images, GFP signals and Texas Red signals on the grid were acquired through the reflection channels and the corresponding fluorescence channel, respectively. The overlay images were then exported into FEI MAPS software to correlate fluorescence microscopy images with scanning EM images, which guided subsequent FIB milling.
For FIB milling, the grid was transferred to the FIB chamber, where it was first coated with a Pt layer using GIS for 30 s, followed by sputtering at 30 mA for 15 s to minimize ion beam damage and enhance electrical conductivity. Alignment of fluorescence and scanning EM images was performed before milling. Regions containing fluorescence signals were milled using an 1 nA ion beam to produce lamellae with an initial thickness of 1.5 μm. The current was then progressively reduced, and a 30 pA ion beam was used for final polishing, resulting in lamellae with a final thickness of 150 nm. The prepared lamellae were stored in liquid nitrogen for subsequent data acquisition. The lipid droplets and ER network occupy a large volume within the Huh7 cells, making it highly probable that the structures of interest were retained within the thinned lamella.
Tomogram reconstruction and membrane segmentation
TOMOMAN57 and TOM toolbox58 were used as general tools in image processing. Initially, for TIFF format files, all frames of each tilt were motion corrected using MotionCor2 software59. While for EER format files, 20 or 23 frames (depend on the specific EER frames) with a dose of approximal 0.10 e/Å2 per frame were rendered before motion correction. Subsequently, each tilt series was aligned using patch-tracking method in IMOD software60, and then reconstructed using back projection method to obtain a tomogram. For visualization, all tomograms were rescaled before further processing. Specifically, the wild-type tomograms were binned by a factor of 8, while tomograms from Tmem41b-KO sample used a binning factor of 4 or 6 depending on the desired resolution. Then a deconvolution filter was applied to all tomograms to further improve the contrast61.
Membrane segmentation was firstly performed using MemBrain-Seg62, and then manually polished with Amira 3D 2022.2 (Thermo Fisher Scientific). Lipid droplets were manually segmented with Amira. For the distance between the ER membrane and lipid droplet, each pixel of the lipid droplet edge segmentation was selected and its closest distance to ER membrane segmentation was defined as the distance. ChimeraX (v.1.7.1)63 was used for the final rendering.
Cryo-EM sample preparation
For observation of purified LDs, cryo-EM holey carbon grids (Quantifoil, R2/1, Au, 200 mesh) were glow-discharged before plunge freezing using Vitrobot Mark IV (Thermo Scientific) with 100% humidity and a temperature of 6 °C. Aliquots of around 4 μl of the purified LD samples were applied to the grids, and blotted with a force of 0 and time of 5 s. All of the grids were observed using the Talos Arctica or Glacios2 microscope operated at an accelerating voltage of 200 kV.
Calculating the number imbalance between the cytoplasmic and lumenal leaflets
The number of phospholipid molecules composing a membrane layer was calculated as the ratio of the membrane surface area to the cross-sectional area of a phospholipid molecule’s phosphate group (alipid). The number of phospholipid molecules in the cytosolic leaflet of the ER (Ni) is 4πro2/alipid; and, the number in the lumenal leaflet of the ER (No) is 4πri2/alipid, where ri is the inside radius of the encapsulating LD and the outer radius (ro) is the inside radius + t (the thickness of the ER bilayer membrane). As an assumption, t and alipid are typically 5 nm and 0.7 nm2 in the eukaryotic ER membrane, respectively. The geometric model derivation shows that the difference in the number of phospholipids between the inner and outer membranes (No − Ni) is 4πt × (2ri +t)/alipid. As t is much smaller than the ri of geLDs, the formula of No − Ni can be simplified to 8πtri2/alipid ≈ 180ri. The imbalance percentage (No − Ni)/No = 2t/ri, ~ 10/ri (assume t = 5 nm).
CLCC1 structure prediction
The amino acid sequence 91–360 of human CLCC1 was used to predict the monomer and hexamer structure using AlphaFold333.
Statistics and reproducibility
All experimental results are presented as mean ± s.e.m. unless otherwise noted in the figure legends. Sample sizes were not predetermined by statistical methods. Statistical analysis was performed with GraphPad Prism 9. Statistical significance was determined using Student’s t-tests or one-way ANOVA with Tukey’s post hoc test, as indicated in the figure legends. Results were considered significant if P < 0.05. Statistical test P values are labelled in the relevant figures. Experimental results shown are representative of at least three biologically independent experiments. For Fig. 1b,c and Extended Data Fig. 1c,e, a total of 40 and 197 tomograms was collected from 2 control mice and 3 Tmem41b-LKO mice, respectively, with at least two independent experiments performed per mouse, all yielding consistent results. Mouse experiments were randomized. Imaging and histology were evaluated in a blinded manner.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.